PID TUboovarian Complex
Diunggah oleh
Alvin Germo PasuquinDeskripsi Asli:
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
PID TUboovarian Complex
Diunggah oleh
Alvin Germo PasuquinHak Cipta:
Format Tersedia
1
A Case of Pelvic Inflammatory Disease
Silliman University Medical
School
Submitted by: Pasuquin, Alvin
01/26/2016
Submitted to: Dr. Gem Austria
Gynecology Worksheet
REPRESENTATIVE CASE
This is a case of LHG, a 25-year-old female, single, Filipino, Roman Catholic, an Online Job
woker from Magbanua, Bantayan, Dumaguete City, Negros Oriental who came in at Silliman
University Medical Center on January 14, 2016 due to persistent hypogastric pain.
The source of information is the patient herself with 90%.
HISTORY OF PRESENT ILLNESS
The patient was apparently well until 6 hours prior to admission, the patient had
an onset of generalized abdominal pain described as cramping in character, rated
as 7/10 in a pain scale. No other symptoms were noted such as fever, nausea and
vomiting. No medications were taken. Pain resolved spontaneously, 2 hours after.
4 hours prior to admission, the patient felt another discomfort, localized at the
hypogastric area described as pulsating and cramping, rated at 10/10 in a pain
scale. No other symptoms were noted. Patient took Mefenamic acid which
afforded temporary relief. Persistence of hypogastric pain prompted consultation,
and was subsequently admitted.
OB-GYNE HISTORY
The patient had her menarche at 11 years old which lasted for 3-4 days,
consuming <3 pads a day, moderately soaked, associated with occasional
dysmenorrhea. Subsequent menstruations were of regular interval of 30-daycycle lasting for 3-4 days, consuming 3 pads a day, moderately soaked, associated
with occasional dysmenorrhea.
Coitarche at 18 years of age. Patient claims to be sexually active with currently 1
sexual partner but had multiple sexual partners in the past. No history of sexually
transmitted diseases, as claimed. No history of oral contraceptive use.
Her OB score is G0. Her LMP was on 12/12/15. Her PMP was on 11/16/15
PAST MEDICAL HISTORY
Her first hospitalization was in October 2013 due to Acute Sinusitis. No other
known conditions such as hypertension, diabetes mellitus, bronchial asthma,
tuberculosis and heart diseases. No previous hospitalizations. She has no known
food and drug sensitivities. No history of trauma, injury or accidents. No previous
surgeries.
FAMILY HISTORY
A Case of Pelvic Inflammatory Disease
Maternal side: (+) HPN, (-) DM, (-) BA, (-) Malignancy
Paternal side: (+) HPN, (-) DM, (-) BA, (-) Malignancy
PERSONAL and SOCIAL HISTORY
She is not a cigarette smoker but an alcoholic beverage drinker. She consumes to
bottles of beer per week. Her last alcohol intake was on January 1, 2016. No
history of illicit drug use.
REVIEW OF SYSTEMS
General: no weight loss, no fatigue, no fever, no weakness, no difficulty sleeping,
Skin: no rashes, no lumps, no itching, no dryness
HEENT: no headache, no head injury, no neck pain, no earache, no drainage, no
vision changes, no eye pain, no redness, no blurring of vision, no stuffiness, no
discharge, no itching, no nosebleed, no sinus tenderness, no bleeding, no dry
mouth, no sore throat, no hoarseness, no thrush, no sores
Neck: no pain, no stiffness
Respiratory: no cough, no sputum, no hemoptysis, no shortness of breath
Cardio: no chest pain, no chest discomfort, no chest tightness, no palpitations
Gastrointestinal: no swallowing difficulties, no heart burn, no change in appetite,
with occasional nausea, no change in bowel habits, no rectal bleeding, no
diarrhea, no constipation,
Urinary: no frequency, no urgency, no dysuria, no hematuria, no incontinence,
Vascular: no calf pain with walking, no leg cramping
Musculoskeletal: no muscle or joint pain, no stiffness, no back pain, no tingling,
no tremor
Hematologic: no easy bruisability, no easy bleeding
Endocrine: no heat or cold intolerance, no sweating, no frequent urination, no
thirst, no change in appetite.
PHYSICAL EXAMINATION
General: Examined awake, conscious, coherent, responsive to questions, not in
respiratory distress
Vital Signs: BP: 90/60 mmHg; RR: 24/min; HR = 88/min; Temp: 36.9 C ; O 2
saturation: 98%
Skin: no pallor, no jaundice, no pigmentation, no active lesions, warm, good skin
turgor and mobility, CRT < 2 seconds
HEENT: anicteric sclerae, pink palpebral conjunctivae, no eye discharge, no sinus
tenderness, moist lips and oral mucosae, no tonsillopharyngeal congestion
Neck: supple neck, no neck vein engorgement, no lymphadenopathies, no thyroid
enlargement
Chest and Lungs: Symmetric chest expansion, no retractions, resonant lungs,
clear breath sounds
Heart: adynamic precordium, distinct heart sounds, normal rate and regular
rhythm, no murmurs
Abdomen: flat, nondistended, normoactive bowel sounds, no organomegaly, direct
tenderness in the hypogastric area
BPE: closed cervix, (+) cervical motion tenderness upon lateral wiggling of
examining finger, minimal mucupurulent slightly malodorous discharge
GUT: Negative kidney punch sign, bilateral; no discharge;
Extremities: No gross deformities, pink nail beds, strong peripheral pulses, full
range of motion
A Case of Pelvic Inflammatory Disease
PRIMARY WORKING IMPRESSION: Acute Pelvic Inflammatory Disease (Tubo-ovarian
Complex), G0
Pelvic inflammatory disease (PID) comprises a spectrum of inflammatory disorders of the upper female genital
tract, including any combination of endometritis, salpingitis, tubo-ovarian abscess, and pelvic peritonitis. Sexually
transmitted organisms, especially Neisseria gonorrhoeae and Chlamydia trachomatis, are implicated in many
cases; however, microorganisms that comprise the vaginal flora (e.g., anaerobes, Gardnerella vaginalis,
Haemophilus influenzae, enteric Gram-negative rods, and Streptococcus agalactiae) also have been associated with
PID. In addition, cytomegalovirus (CMV), Mycoplasma hominis, and Ureaplasma urealyticum may be the etiologic
agents in some cases of PID.
CLINICAL DIAGNOSTIC CRITERIA FOR PID
One or more of the following minimum criteria must be present on pelvic
examination to diagnose PID:
Cervical motion tenderness
Uterine tenderness
Adnexal tenderness
The following criteria can improve the specificity of the diagnosis:
Oral temperature > 101F (> 38.3C)
Abnormal cervical or vaginal mucopurulent discharge
Presence of abundant numbers of white blood cells on saline microscopy of vaginal
fluid
Elevated erythrocyte sedimentation rate
Elevated C-reactive protein level
Laboratory documentation of cervical infection with gonorrhea or chlamydia
The following test results are the most specific criteria for diagnosing PID
Endometrial biopsy with histopathologic evidence of endometritis
Transvaginal sonography or magnetic resonance imaging techniques showing
thickened, fluid-filled tubes with or without free pelvic fluid or tubo-ovarian complex,
or Doppler studies suggesting pelvic infection (e.g., tubal hyperemia)
Laparoscopic abnormalities consistent with PID
Risk Factors
Risk factors for PID which are present in this particular patient include age younger than 25
years; young age at first sexual encounter; new, multiple sex partners; Patients vaginal
overdouching may also be a risk factor.
DIFFERENTIAL DIAGNOSIS
The differential diagnosis of lower abdominal pain in a young woman includes:
1.
2.
3.
4.
5.
6.
ectopic pregnancy
acute appendicitis
endometriosis
irritable bowel syndrome
complications of an ovarian cyst i.e. rupture, torsion
functional pain (pain of unknown physical origin)
DIAGNOSTIC WORK-UP
1. Testing for gonorrhoea and chlamydia in the lower genital tract is recommended
since a positive result supports the diagnosis of PID. However the absence of
infection from the endocervix or urethra does not exclude PID
2. The absence of endocervical or vaginal pus cells has a good negative predictive value
(95%) for a diagnosis of PID but their presence is non-specific (poor positive
predictive value 17%)
3. Human immunodeficiency virus (HIV) testing (e.g., enzyme-linked immunosorbent
A Case of Pelvic Inflammatory Disease
assay [ELISA]
or Western blot) and counseling should be offered to all patients with suspected PID
or sexually
transmitted disease (STD)
4. An elevated ESR or C reactive protein supports the diagnosis 11 but is non-specific
and often normal in mild/moderate PID
5. CBC- Elevation of the white cell count (WBC) supports the diagnosis but can be
normal in mild cases.
6. Laparoscopy is the criterion standard for the diagnosis of PID. It is significantly more
specific and sensitive than are clinical criteria alone. The minimum criteria for
diagnosing PID laparoscopically include tubal wall edema, visible hyperemia of the
tubal surface, and the presence of exudate on the tubal surfaces and fimbriae.
However, it is not justified routinely on the basis of associated morbidity, cost and the
potential difficulty in identifying mild intra-tubal inflammation or endometritis
7. Endometrial biopsy may also be helpful when there is diagnostic difficulty
8. Urinalysis to rule out urinary tract infection (UTI)
9. A pregnancy test should be performed to help exclude an ectopic pregnancy
10. Transvaginal Sonography -Although ultrasonography is neither specific nor sensitive
in distinguishing the cause of a pelvic mass, findings of dilated and fluid-filled tubes,
free peritoneal fluid, and adnexal masses may be confirmatory of symptoms and
physical signs. Thus, vaginal ultrasound has a high positive predictive value when
used in a high-risk population.
Patients result
Uterus: 3.54 x 4.45 x 3.67 cm, retroverted
Endometrium: 1.43 cm, hyperechoic
Cervix: 2.17 x 2.06 without nabothian cyst
R Ovary: 3.03 x 2.58 CM, lateral
L Ovary: 3.04 x 1.99 cm, posterior
Findings:
Endometrial and myometrial echoes appear homogenous and intact
Endometrium thick and hypoechoic with intact subendometrial halo
Left ovary unremarkable
Within R Ovary is a thin walled, unilocular, cystic structure measuring 2.71 x
2.21 cm with homogenous low level echoes within
Medial to R Ovary is a biloculated, tubular cystic structure 5.33 x 2.64 cm
filled with homogenous low level echoes
Minimal free fluid at the posterior cul de sac, left adnexal area with thin
adhesions
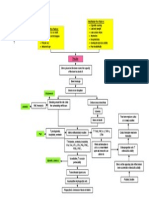
PATHOPHYSIOLOGY DIAGRAM
Risk Factors
Younger age
Multiple sex
partners
No condom use
PELVIC
INFLAMMATORY
DISEASE
Vaginal Inflammation and
hormonal change during
ovulation or menstruation
Salphinx and Ovaries
Cervix
Salphingitis
Mucopurulent
Cervicitis
Sterilitty
A Case of Pelvic Inflammatory Disease
Efficacy of the
functional barrier
provided by the cervical
Acquisition of vaginal or cervical
infection through sexual contact
(infected exudates and secretions)
Gonococcus and other bacteria
adhering to the Epithelium may
Gonococcus
Pili- attach to mucosal surface and prevents ingestion by
neutrophils
OPA proteins- adherence between gonococcus and
phagocytes, promoting invasion into the host cell,
Microorganisms ascend from the lower genital tract
through rhythmic uterine contractions occurring during
orgasm or bacteria may also be carried along with sperm
Inflammation
Hyperthermia
Pain
Erythema
Heat and Burning sensation
Swelling
Retrograde spread of the organisms
Urethra
Unrethrit
is
Dysuria
Vagina
Vaginitis
Formation
of Small
and larger
Abscess
Tubo-Ovarian
Complex
Salphingooophoritis
PATHOPHYSIOLOGY
Bilateral
Abdominal
Acute PID is usually a polymicrobial infection caused by organisms
ascending
from the
vagina and cervix along the mucosa of the endometrium to infect the mucosa of the oviduct.
In many cases, no causative organism is found. The two classic sexually transmitted
organisms associated with PID, N. gonorrhoeae
and C. trachomatis, cause acute PID in many cases.
A Case of Pelvic Inflammatory Disease
Risk Factors
Adolescence is a risk factor because of increased age-related chlamydia and gonorrhea
rates and the presence of cervical ectopy, which allows for increased adherence of infectious
organisms.
History of PID: damaged fallopian tube mucosa may be more susceptible to recurrent
infection.
History of gonorrhea or chlamydia: increased likelihood of recurrent gonorrhea or
chlamydia.
Male partners with gonorrhea or chlamydia
Multiple sex partners
Current douching: contributes to vaginal flora changes, epithelial damage, and disruption
of cervical mucous barrier.
Insertion of IUD within the first 21 days of placement (this risk is greatly reduced if a
woman is tested and, if necessary, treated for STDs before an IUD is inserted); after 21 days,
risk returns to baseline.
Bacterial vaginosis has been associated with PID.
Oral contraceptive use: may increase the risk of cervical chlamydial infection because of
cervical ectopy, but decreases the risk of clinically apparent symptomatic PID (mechanisms
unclear). Oral contraceptives also cause thickening of cervical mucous which may be
protective against lower genital tract organisms ascending
into the upper genital tract.
Demographics (socioeconomic status): may be related to access to care.
INFORMATION, EXPLANATION AND ADVICE
1. Patients should be advised to avoid unprotected intercourse until they, and their
partner(s),
have completed treatment and follow-up
2. A detailed explanation of their condition with particular emphasis on the long-term
implications for the health of themselves and their partner(s) should be provided,
reinforced with clear and accurate written information. Appropriate information
should include:
a. fertility is usually well preserved in women with first episode PID who receive
prompt appropriate anti-microbial therapy
b. the risk of impaired fertility increases significantly with each subsequent
episode of PID
c. the risk of impaired fertility is increased in clinically more severe PID
d. chronic pelvic pain of varying severity affects around 30% of women following
PID
e. PID increases the relative risk of a subsequent pregnancy being an ectopic,
but the absolute risk of ectopic pregnancy remains low at around 1%
3. Partner Notification
a. Current male partners of women with PID should be contacted and offered
health advice and screening for gonorrhea and chlamydia. Other recent sexual
partners may also be offered screening - tracing of contacts within a 6 month
period of onset of symptoms is recommended but this time period is not
evidence based and may be influenced by the sexual history, available
resources or local practice.
b. Partners should be advised to avoid unprotected intercourse until they and
their partner have completed the treatment course.
A Case of Pelvic Inflammatory Disease
c. Gonorrhea diagnosed in the male partner should be treated appropriately
SUGGESTED CRITERIA FOR HOSPITALIZATION OF PATIENTS WITH PELVIC
INFLAMMATORY DISEASE
Inability to follow or tolerate an outpatient oral medication regimen
No clinical response to oral antimicrobial therapy
Pregnancy
Severe illness, nausea and vomiting, or high fever
Surgical emergencies (e.g., appendicitis) cannot be excluded
Tubo-ovarian abscess
IN-PATIENT REGIMEN
1. Patient should be admitted for parenteral antibiotic therapy
2. Start intravenous fluid of PNSS 1 L @ 30 gtts/min for fluid replacement
3. Monitor the intake and output of the patient.
4. Advise the patient to eat low fat-low protein diet due to its liver involvement.
5. Monitor the VS every four hours.
6. Patient should be nursed in a semi-fowler position with frequent monitoring of
general and pelvic signs
7. Medications:
Antibiotics : IV Cefoxitin 2g four times daily (or i.v. cefotetan 2g twice daily or i.v./i.m.
ceftriaxone 1g once daily) + IV Doxycycline 100mg twice daily followed by Oral
doxycycline 100mg twice daily + Oral metronidazole 400mg twice daily to complete
14 days
Pain Reliever: Demerol 25 mg + Promethazine (Phenergan) 25 mg cocktail slow IVTT
DRUG STUDY
Ceftriaxone
1g once daily
Oral
metronidazole
400mg twice
daily to
complete 14
days
IV Doxycycline
100mg twice
daily
Inhibits the mucopeptide synthesis
in the bacterial cell wall. The betalactam moiety of Ceftriaxone binds
to carboxypeptidases,
endopeptidases, and
transpeptidases in the bacterial
cytoplasmic membrane. These
enzymes are involved in cell-wall
synthesis and cell division. By
binding to these enzymes,
Ceftriaxone results in the formation
of of defective cell walls and cell
death.
Inhibits nucleic acid synthesis by
disrupting DNA and inhibits protein
synthesis causing cell death;
amebicidal, bactrericidal,
trichomonacidal.
Doxycycline reversibly binds to the
30 S ribosomal subunits and possibly
the 50S ribosomal subunit(s),
blocking the binding of
Contraindicated to patients with
hypersensitivity to cephalosporins;
neonates. Do not use calcium or
calcium-containing solutions or
products with or within 48 hours of
ceftriaxone administration due to
risk of calcium-ceftriaxone
precipitate formation in neonates.
Hypersensitivity to the drug is a
contraindication. Caution in patients
with CNS disease, severe hepatic
impairment, history with blood
dycrasias. Avoid alcohol while
taking medication. Adverse
reactions include appetite loss,
candidiasis, diarrhea, dizziness,
headache, nausea, and vomiting.
Contraindicated in pregnancy,
lactation; porphyria;
hypersensitivity to tetracyclines;
severe hepatic dysfunction;
A Case of Pelvic Inflammatory Disease
aminoacyltRNA to the mRNA and
inhibiting bacterial protein synthesis.
Doxycycline prevents the normal
function of the apicoplast of
Plasmodium falciparum, a malaria
causing organism.
prolonged exposure to sunlight or
tanning
FOLLOW-UP
1. Review at 72 hours is recommended, particularly for those with a moderate or severe
clinical presentation, and should show a substantial improvement in clinical
symptoms and signs. Failure to do so suggests the need for further investigation,
parenteral therapy and/or surgical intervention.
2. Further review 4 weeks after therapy may be useful to ensure:
a. adequate clinical response to treatment
b. compliance with oral antibiotics
c. screening and treatment of sexual contacts
d. advice on future use of condoms to prevent recurrent PID
3. Repeat testing for gonorrhoea or chlamydia is appropriate:
a. in those with persistent symptoms
b. where antibiotic sensitivities are unknown or resistance is present
(gonorrhoea only)
c. history of poor compliance with antibiotics
d. inadequate tracing of sexual contacts where there is a possibility of persisting
or recurrent infection.
Anda mungkin juga menyukai
- Chronic Diseases - Lymes, Hpv, Hsv Mis-Diagnosis and Mistreatment: A New Approach to the EpidemicDari EverandChronic Diseases - Lymes, Hpv, Hsv Mis-Diagnosis and Mistreatment: A New Approach to the EpidemicPenilaian: 5 dari 5 bintang5/5 (1)
- Pelvic Inflammatory DiseaseDokumen6 halamanPelvic Inflammatory DiseaseLembah BarokahBelum ada peringkat
- Ob-Gyn CR 1Dokumen11 halamanOb-Gyn CR 1Tresor MbuyiBelum ada peringkat
- Pelvic Inflammatory DiseaseDokumen8 halamanPelvic Inflammatory DiseaseKhairunNisaBelum ada peringkat
- PID & Ectopic PregnancyDokumen23 halamanPID & Ectopic Pregnancy會 Ṧwahsa 會Belum ada peringkat
- A Patient With Wasting and Oral Thrush FinalDokumen43 halamanA Patient With Wasting and Oral Thrush FinalWasswaBelum ada peringkat
- Pelvic Inflammatory DiseaseDokumen71 halamanPelvic Inflammatory DiseaseGraceline Margaretha Marsintauly SianiparBelum ada peringkat
- Pelvic Inflammatory Disease: Bibo Yuan M.D.,PH.DDokumen51 halamanPelvic Inflammatory Disease: Bibo Yuan M.D.,PH.DNorman AjxBelum ada peringkat
- Pelvic Inflammatory Disease (PID)Dokumen16 halamanPelvic Inflammatory Disease (PID)CHRISTIAN RAY ALPAS PASILIAOBelum ada peringkat
- What Is Causing This Girl S Abdominal Pain .10Dokumen2 halamanWhat Is Causing This Girl S Abdominal Pain .10LuisBelum ada peringkat
- 1101201903.pelvic Inflammatory DiseaseDokumen34 halaman1101201903.pelvic Inflammatory Diseaseraed faisalBelum ada peringkat
- Case Report n0 31Dokumen19 halamanCase Report n0 31EkoApriandhiBelum ada peringkat
- I Think I Am Pregnant?: Hawler Medical University Collage of Medicine OBGYN Department DR Trifa Y. MuttalibDokumen18 halamanI Think I Am Pregnant?: Hawler Medical University Collage of Medicine OBGYN Department DR Trifa Y. MuttalibZahraa HasanBelum ada peringkat
- PID LectureDokumen41 halamanPID LectureHarold Gabriel Sanchez DelgadoBelum ada peringkat
- Pelvic Inflammatory DiseaseDokumen10 halamanPelvic Inflammatory DiseaseMuhammad IqbalBelum ada peringkat
- Pid BSTDokumen17 halamanPid BSTPutri Senna RahayuBelum ada peringkat
- Patient ProfileDokumen19 halamanPatient Profilebangus08Belum ada peringkat
- Hematemesis Melena Due To Helicobacter Pylori Infection in Duodenal Ulcer: A Case Report and Literature ReviewDokumen6 halamanHematemesis Melena Due To Helicobacter Pylori Infection in Duodenal Ulcer: A Case Report and Literature ReviewodiBelum ada peringkat
- Hematemesis Melena Due To Helicobacter Pylori Infection in Duodenal Ulcer: A Case Report and Literature ReviewDokumen6 halamanHematemesis Melena Due To Helicobacter Pylori Infection in Duodenal Ulcer: A Case Report and Literature ReviewWahyu Agung PribadiBelum ada peringkat
- Case Presentation en Dome Trios IsDokumen36 halamanCase Presentation en Dome Trios IsVindhya SubramanyamBelum ada peringkat
- ENDOMETRITISDokumen6 halamanENDOMETRITISKhrisna ParamaarthaBelum ada peringkat
- Pelvic Inflammatory Disease: CDC 24/7: Saving Lives. Protecting PeopleDokumen9 halamanPelvic Inflammatory Disease: CDC 24/7: Saving Lives. Protecting PeopleAmanda GABelum ada peringkat
- Appendicitis Database 3Dokumen8 halamanAppendicitis Database 3khushrooBelum ada peringkat
- Ob-Gyn CR2Dokumen10 halamanOb-Gyn CR2Tresor MbuyiBelum ada peringkat
- Cell Carcinoma, But Also To Other Cancers As Well (E.g., Thyroid Carcinoma, Melanoma)Dokumen4 halamanCell Carcinoma, But Also To Other Cancers As Well (E.g., Thyroid Carcinoma, Melanoma)Erick EchualBelum ada peringkat
- PPT-Acute PyeloDokumen117 halamanPPT-Acute PyeloJudy UrciaBelum ada peringkat
- Abortion, Myoma, H Mole, EctopicDokumen93 halamanAbortion, Myoma, H Mole, Ectopicdhainey100% (4)
- BM18110019 Shrley Chee See Yin (R2 Obstetric Cwu)Dokumen12 halamanBM18110019 Shrley Chee See Yin (R2 Obstetric Cwu)RUDOLF YUSON BM20110052Belum ada peringkat
- Gynecology Notes Legal Issues and EthicsDokumen19 halamanGynecology Notes Legal Issues and EthicsUsm LeeBelum ada peringkat
- Differential Diagnoses APDokumen3 halamanDifferential Diagnoses APrae_ramirez25Belum ada peringkat
- Womens Health - Soap Note 1Dokumen11 halamanWomens Health - Soap Note 1api-494643478100% (2)
- Case Discussion PD: Pelvic Inflammatory Disease/Tubo-ovarian AbscessDokumen7 halamanCase Discussion PD: Pelvic Inflammatory Disease/Tubo-ovarian AbscessJASMEEN RAVALBelum ada peringkat
- Colorectal CancerDokumen50 halamanColorectal CancerHans Chester Rirao Capinding100% (2)
- Hum. Reprod.-2013-Romosan-1569-79Dokumen11 halamanHum. Reprod.-2013-Romosan-1569-79Ahmad Arbi AninditoBelum ada peringkat
- CASE WRITE UP-obstetric CaseDokumen7 halamanCASE WRITE UP-obstetric CaseAmbhi GanaBelum ada peringkat
- Portfolio 1 - Pre-EclampsiaDokumen13 halamanPortfolio 1 - Pre-EclampsiaJojo LiBelum ada peringkat
- Pelvic Inflammatory DiseaseDokumen34 halamanPelvic Inflammatory DiseaseOhunakin AfolabiBelum ada peringkat
- Pelvic Inflammatory Disease - Clinical Manifestations and DiagnosisDokumen13 halamanPelvic Inflammatory Disease - Clinical Manifestations and DiagnosisJavier Manuel Escobedo CalderónBelum ada peringkat
- Midwifery 102 Module 1Dokumen10 halamanMidwifery 102 Module 1WynJoy NebresBelum ada peringkat
- Chronic Pelvic Pain Women's Health Case ReportDokumen6 halamanChronic Pelvic Pain Women's Health Case ReportSharon Yunn Li ChenBelum ada peringkat
- SENTE Pharma Lab UTIDokumen13 halamanSENTE Pharma Lab UTIAnissa SenteBelum ada peringkat
- IBD-Rough Road - Case Study in IBD From Presentation To ManagementDokumen4 halamanIBD-Rough Road - Case Study in IBD From Presentation To ManagementBejan CorinaBelum ada peringkat
- OB FNP Case PresentationDokumen13 halamanOB FNP Case PresentationAdamBelum ada peringkat
- Pid Casestudy 2014 PDFDokumen4 halamanPid Casestudy 2014 PDFNur Syamsiah MBelum ada peringkat
- HIVAIDS and PregnancyDokumen44 halamanHIVAIDS and PregnancyAnastasiafynnBelum ada peringkat
- Quizz 1 Review Advanced IIDokumen66 halamanQuizz 1 Review Advanced IIamelyBelum ada peringkat
- Ectopic PregnancyDokumen54 halamanEctopic Pregnancypatriciaatan1497Belum ada peringkat
- CASE STUDY (Gastroenteritis)Dokumen7 halamanCASE STUDY (Gastroenteritis)Bible EvergardenBelum ada peringkat
- Focused History AssignmentDokumen4 halamanFocused History AssignmentEzioBelum ada peringkat
- Pelvic in Ammatory DiseaseDokumen5 halamanPelvic in Ammatory DiseaseGina Eliana Custodio GonzalesBelum ada peringkat
- Alumna: Código:: - Guivin Pereyra SharonDokumen42 halamanAlumna: Código:: - Guivin Pereyra SharonSharon Guivin PereyraBelum ada peringkat
- Obstetrics and GynaecologyDokumen31 halamanObstetrics and GynaecologyLana LocoBelum ada peringkat
- Puerperal SepsisDokumen24 halamanPuerperal SepsisWindelyn Gamaro60% (5)
- Pelvic Inflammatory DiseaseDokumen25 halamanPelvic Inflammatory DiseasedeepaBelum ada peringkat
- Case Protocol UtiDokumen4 halamanCase Protocol UtiRommel IrabagonBelum ada peringkat
- Judul 1Dokumen8 halamanJudul 1Ahmad Harissul IbadBelum ada peringkat
- CCD Obstetric GynecologyDokumen43 halamanCCD Obstetric GynecologyHanif GandohBelum ada peringkat
- OHSSDokumen4 halamanOHSSNur AnindyaBelum ada peringkat
- Tasha's Gynae Case WriteDokumen6 halamanTasha's Gynae Case WriteMelissa Aina Mohd YusofBelum ada peringkat
- 2018.pelvic Inflammatory DiseaseDokumen4 halaman2018.pelvic Inflammatory DiseaseObgynism 2019Belum ada peringkat
- May ReflectionDokumen4 halamanMay ReflectionAlvin Germo PasuquinBelum ada peringkat
- Seizure - Transient Occurrence of Signs or Symptoms Due To Abnormal Excessive or SynchronousDokumen2 halamanSeizure - Transient Occurrence of Signs or Symptoms Due To Abnormal Excessive or SynchronousAlvin Germo PasuquinBelum ada peringkat
- Fever and Rash: Alvin Germo PasuquinDokumen70 halamanFever and Rash: Alvin Germo PasuquinAlvin Germo PasuquinBelum ada peringkat
- Reflection May-AugDokumen4 halamanReflection May-AugAlvin Germo PasuquinBelum ada peringkat
- Journal Appraisal FinslDokumen26 halamanJournal Appraisal FinslAlvin Germo PasuquinBelum ada peringkat
- Clinical Case - 2019: Alvin Germo PasuquinDokumen49 halamanClinical Case - 2019: Alvin Germo PasuquinAlvin Germo PasuquinBelum ada peringkat
- Case Management: Leira Dith R. Castro, MD Presentor 1 Year Im ResidentDokumen64 halamanCase Management: Leira Dith R. Castro, MD Presentor 1 Year Im ResidentAlvin Germo PasuquinBelum ada peringkat
- PEDIA - TachypneaDokumen12 halamanPEDIA - TachypneaAlvin Germo PasuquinBelum ada peringkat
- Pathophysiology: Modifiable Risk FactorsDokumen1 halamanPathophysiology: Modifiable Risk FactorsAlvin Germo PasuquinBelum ada peringkat
- Advice and Information: ManagementDokumen4 halamanAdvice and Information: ManagementAlvin Germo PasuquinBelum ada peringkat
- Pathophysiology of Secondary CHFDokumen1 halamanPathophysiology of Secondary CHFAlvin Germo PasuquinBelum ada peringkat
- Advice and Information: ManagementDokumen4 halamanAdvice and Information: ManagementAlvin Germo PasuquinBelum ada peringkat
- CROHNSDokumen2 halamanCROHNSAlvin Germo Pasuquin100% (1)
- Acute PancreatitisDokumen9 halamanAcute PancreatitisAlvin Germo PasuquinBelum ada peringkat
- Carbuncle, Incision, Drainage, DebridementDokumen11 halamanCarbuncle, Incision, Drainage, DebridementAlvin Germo PasuquinBelum ada peringkat
- Proceso Marc F. Udarbe: Representative CaseDokumen6 halamanProceso Marc F. Udarbe: Representative CaseAlvin Germo PasuquinBelum ada peringkat
- Pathophysiology of Colonic Diverticulosis: Risk Factors Other Risk FactorsDokumen1 halamanPathophysiology of Colonic Diverticulosis: Risk Factors Other Risk FactorsAlvin Germo PasuquinBelum ada peringkat
- These Are The Documents Needed in Filing YourDokumen2 halamanThese Are The Documents Needed in Filing YourSuzanne Matuba SaetBelum ada peringkat
- Placenta Previa Differential DiagnosesDokumen4 halamanPlacenta Previa Differential DiagnosesikaoktaviasaktiBelum ada peringkat
- American Life League Report On Planned Parenthood Federation of AmericaDokumen48 halamanAmerican Life League Report On Planned Parenthood Federation of AmericaShane Vander HartBelum ada peringkat
- Childrens Sexual Behaviours 2015Dokumen11 halamanChildrens Sexual Behaviours 2015ninogallardo7Belum ada peringkat
- UGANDA SCOUTS ASSOCIATION SVoice01Dokumen1 halamanUGANDA SCOUTS ASSOCIATION SVoice01Conrad NatseBelum ada peringkat
- OBGYN Vaginal Delivery DikaDokumen10 halamanOBGYN Vaginal Delivery DikaOnyedika EgbujoBelum ada peringkat
- Worksheet - 2 - 10th - EnglishDokumen3 halamanWorksheet - 2 - 10th - EnglishsebastianBelum ada peringkat
- Heavy Petting, by Peter SingerDokumen3 halamanHeavy Petting, by Peter SingerTheDocAnonBelum ada peringkat
- OBSTETRIC NURSING Hand Out UPANG INHOUSE 2022Dokumen7 halamanOBSTETRIC NURSING Hand Out UPANG INHOUSE 2022Mary Ann G. CorsanesBelum ada peringkat
- The RH Bill (Pros & Cons)Dokumen1 halamanThe RH Bill (Pros & Cons)Ocariza Christy0% (1)
- Knowledge Attitude and Practice of ContraceptiveDokumen5 halamanKnowledge Attitude and Practice of ContraceptiveSam AdeBelum ada peringkat
- 4 Unit End eDokumen47 halaman4 Unit End eFiona TamBelum ada peringkat
- 2010 Lesson Plan Grade 9Dokumen11 halaman2010 Lesson Plan Grade 9809650577Belum ada peringkat
- OrgasmDokumen20 halamanOrgasmnathanBelum ada peringkat
- Penis Enlargement Exercises-The Natural Penis Elargment Guide-EducationalDokumen30 halamanPenis Enlargement Exercises-The Natural Penis Elargment Guide-Educationalmesagerdivin17Belum ada peringkat
- Vaginal InfectionsDokumen2 halamanVaginal InfectionsIndhumathiBelum ada peringkat
- Complete Androgen Insensitivity Syndrome: A Case Repport and Review of LiteratureDokumen4 halamanComplete Androgen Insensitivity Syndrome: A Case Repport and Review of LiteratureIJAR JOURNALBelum ada peringkat
- The Birth Process (Reading About Birth)Dokumen4 halamanThe Birth Process (Reading About Birth)BAYUFIJRIEBelum ada peringkat
- Forcep DeliveryDokumen32 halamanForcep DeliverybbaltazarjaniceBelum ada peringkat
- Project Topics - Family Law I, Div A-EDokumen30 halamanProject Topics - Family Law I, Div A-EHarsh GargBelum ada peringkat
- Polyhydramnios: SymptomsDokumen4 halamanPolyhydramnios: Symptomsbambem aevanBelum ada peringkat
- Postpartum Care PDFDokumen8 halamanPostpartum Care PDF2C- VILLACARLOS, LEONA ROSE M.Belum ada peringkat
- PDFDokumen24 halamanPDFGoldi RaghavBelum ada peringkat
- HyaDokumen3 halamanHyamae_hyacinth_cuteBelum ada peringkat
- Groups: The Heart of Interaction Social Aggregates: 2. Secondary GroupDokumen5 halamanGroups: The Heart of Interaction Social Aggregates: 2. Secondary GroupKenneth BoquingBelum ada peringkat
- Pelvic Organ ProlapseDokumen3 halamanPelvic Organ ProlapseMelissa Aina Mohd YusofBelum ada peringkat
- NSTDokumen5 halamanNSTMarlisa YanuartiBelum ada peringkat
- Premature BirthsDokumen3 halamanPremature BirthsZeynep YılmaztürkBelum ada peringkat
- Cephalopelvic DisproportionDokumen2 halamanCephalopelvic DisproportionRuru AlharthyBelum ada peringkat
- FP MethodDokumen138 halamanFP MethodSonu SubbaBelum ada peringkat
- Love Life: How to Raise Your Standards, Find Your Person, and Live Happily (No Matter What)Dari EverandLove Life: How to Raise Your Standards, Find Your Person, and Live Happily (No Matter What)Penilaian: 3 dari 5 bintang3/5 (1)
- Summary: The Psychology of Money: Timeless Lessons on Wealth, Greed, and Happiness by Morgan Housel: Key Takeaways, Summary & Analysis IncludedDari EverandSummary: The Psychology of Money: Timeless Lessons on Wealth, Greed, and Happiness by Morgan Housel: Key Takeaways, Summary & Analysis IncludedPenilaian: 4.5 dari 5 bintang4.5/5 (82)
- The Age of Magical Overthinking: Notes on Modern IrrationalityDari EverandThe Age of Magical Overthinking: Notes on Modern IrrationalityPenilaian: 4 dari 5 bintang4/5 (32)
- LIT: Life Ignition Tools: Use Nature's Playbook to Energize Your Brain, Spark Ideas, and Ignite ActionDari EverandLIT: Life Ignition Tools: Use Nature's Playbook to Energize Your Brain, Spark Ideas, and Ignite ActionPenilaian: 4 dari 5 bintang4/5 (404)
- ADHD is Awesome: A Guide to (Mostly) Thriving with ADHDDari EverandADHD is Awesome: A Guide to (Mostly) Thriving with ADHDPenilaian: 5 dari 5 bintang5/5 (3)
- By the Time You Read This: The Space between Cheslie's Smile and Mental Illness—Her Story in Her Own WordsDari EverandBy the Time You Read This: The Space between Cheslie's Smile and Mental Illness—Her Story in Her Own WordsBelum ada peringkat
- Raising Mentally Strong Kids: How to Combine the Power of Neuroscience with Love and Logic to Grow Confident, Kind, Responsible, and Resilient Children and Young AdultsDari EverandRaising Mentally Strong Kids: How to Combine the Power of Neuroscience with Love and Logic to Grow Confident, Kind, Responsible, and Resilient Children and Young AdultsPenilaian: 5 dari 5 bintang5/5 (1)
- The Comfort of Crows: A Backyard YearDari EverandThe Comfort of Crows: A Backyard YearPenilaian: 4.5 dari 5 bintang4.5/5 (23)
- The Ritual Effect: From Habit to Ritual, Harness the Surprising Power of Everyday ActionsDari EverandThe Ritual Effect: From Habit to Ritual, Harness the Surprising Power of Everyday ActionsPenilaian: 4 dari 5 bintang4/5 (4)
- When the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisDari EverandWhen the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisPenilaian: 3.5 dari 5 bintang3.5/5 (2)
- Think This, Not That: 12 Mindshifts to Breakthrough Limiting Beliefs and Become Who You Were Born to BeDari EverandThink This, Not That: 12 Mindshifts to Breakthrough Limiting Beliefs and Become Who You Were Born to BePenilaian: 2 dari 5 bintang2/5 (1)
- Summary: Outlive: The Science and Art of Longevity by Peter Attia MD, With Bill Gifford: Key Takeaways, Summary & AnalysisDari EverandSummary: Outlive: The Science and Art of Longevity by Peter Attia MD, With Bill Gifford: Key Takeaways, Summary & AnalysisPenilaian: 4.5 dari 5 bintang4.5/5 (42)
- Dark Psychology & Manipulation: Discover How To Analyze People and Master Human Behaviour Using Emotional Influence Techniques, Body Language Secrets, Covert NLP, Speed Reading, and Hypnosis.Dari EverandDark Psychology & Manipulation: Discover How To Analyze People and Master Human Behaviour Using Emotional Influence Techniques, Body Language Secrets, Covert NLP, Speed Reading, and Hypnosis.Penilaian: 4.5 dari 5 bintang4.5/5 (110)
- The Courage Habit: How to Accept Your Fears, Release the Past, and Live Your Courageous LifeDari EverandThe Courage Habit: How to Accept Your Fears, Release the Past, and Live Your Courageous LifePenilaian: 4.5 dari 5 bintang4.5/5 (254)
- Raising Good Humans: A Mindful Guide to Breaking the Cycle of Reactive Parenting and Raising Kind, Confident KidsDari EverandRaising Good Humans: A Mindful Guide to Breaking the Cycle of Reactive Parenting and Raising Kind, Confident KidsPenilaian: 4.5 dari 5 bintang4.5/5 (170)
- Manipulation: The Ultimate Guide To Influence People with Persuasion, Mind Control and NLP With Highly Effective Manipulation TechniquesDari EverandManipulation: The Ultimate Guide To Influence People with Persuasion, Mind Control and NLP With Highly Effective Manipulation TechniquesPenilaian: 4.5 dari 5 bintang4.5/5 (1412)
- The Body Keeps the Score by Bessel Van der Kolk, M.D. - Book Summary: Brain, Mind, and Body in the Healing of TraumaDari EverandThe Body Keeps the Score by Bessel Van der Kolk, M.D. - Book Summary: Brain, Mind, and Body in the Healing of TraumaPenilaian: 4.5 dari 5 bintang4.5/5 (266)
- To Explain the World: The Discovery of Modern ScienceDari EverandTo Explain the World: The Discovery of Modern SciencePenilaian: 3.5 dari 5 bintang3.5/5 (51)
- The Obesity Code: Unlocking the Secrets of Weight LossDari EverandThe Obesity Code: Unlocking the Secrets of Weight LossPenilaian: 4 dari 5 bintang4/5 (6)
- Cult, A Love Story: Ten Years Inside a Canadian Cult and the Subsequent Long Road of RecoveryDari EverandCult, A Love Story: Ten Years Inside a Canadian Cult and the Subsequent Long Road of RecoveryPenilaian: 4 dari 5 bintang4/5 (46)
- The Marshmallow Test: Mastering Self-ControlDari EverandThe Marshmallow Test: Mastering Self-ControlPenilaian: 4.5 dari 5 bintang4.5/5 (60)
- Mindset by Carol S. Dweck - Book Summary: The New Psychology of SuccessDari EverandMindset by Carol S. Dweck - Book Summary: The New Psychology of SuccessPenilaian: 4.5 dari 5 bintang4.5/5 (328)
- Why We Die: The New Science of Aging and the Quest for ImmortalityDari EverandWhy We Die: The New Science of Aging and the Quest for ImmortalityPenilaian: 4.5 dari 5 bintang4.5/5 (6)
- How to ADHD: The Ultimate Guide and Strategies for Productivity and Well-BeingDari EverandHow to ADHD: The Ultimate Guide and Strategies for Productivity and Well-BeingPenilaian: 1 dari 5 bintang1/5 (1)