Kimmelstiel-Wilson Syndrome B
Diunggah oleh
Ranela Kwinkee Pastor SalazarDeskripsi Asli:
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Kimmelstiel-Wilson Syndrome B
Diunggah oleh
Ranela Kwinkee Pastor SalazarHak Cipta:
Format Tersedia
Kimmelstiel-Wilson Syndrome Group O
KIMMELSTIEL WILSON SYNDROME GENERAL OBJECTIVE To comprehensively understand Kimmelstiel-Wilson Syndrome. Specific Objectives To correlate the patients overall history with the disease process. To identify pertinent physical examination results evident in the patient. To analyze the laboratory and diagnostic studies which will justify the diagnosis. To review the anatomy and physiology of the systems involved. To thoroughly explain the pathophysiology of the disorder including risk factors and clinical manifestations. 6. To evaluate the nursing and medical management rendered to the patient through the Nursing Care Plan. 7. To discuss the prognosis of the patient and the recommendations to combat or decelerate the progression of the disease. INTRODUCTION 1. 2. 3. 4. 5. History Diabetic nephropathy (nephropatia diabetica), also known as Kimmelstiel- Wilson syndrome and intercapillary glomerulonephritis, is a progressive kidney disease caused by angiopathy of capillaries in the kidney glomeruli. It is characterized by nephrotic syndrome and nodular glomerulosclerosis. It is due to longstanding diabetes mellitus, and is a prime cause for dialysis in many Western countries. Discovered by British physician Clifford Wilson (1906-1997) and Germanborn American physician Paul Kimmelstiel (1900-1970) and was published for the first time in 1936. Causes The exact cause of diabetic nephropathy is unknown, but it is believed that uncontrolled high blood sugar leads to the development of kidney damage. In some cases, your genes or family history may also play a role. Not all persons with diabetes develop this condition. Each kidney is made of hundreds of thousands of filtering units called nephrons. Each nephron has a cluster of tiny blood vessels called a glomerulus. Together these structures help remove waste from the body. Too much blood sugar can damage these structures, causing them to thicken and become scarred. Slowly, over time, more and more blood vessels are destroyed. The kidney structures begin to leak and protein (albumin) begins to pass into the urine. Persons with diabetes who have the following risk factors are more likely to develop this condition: African American, Hispanic, American Indian or Asian origin Family history of kidney disease or high blood pressure, poor control of blood pressure, poor control of blood sugars, Type 1 diabetes before age 20, smoking. Diabetic nephropathy generally goes along with other diabetes complications including high blood pressure, retinopathy, and blood vessel changes. Prevalence and Incidence Rate Diabetes has become the primary cause of end-stage renal disease (ESRD), and the incidence of type 2 diabetes mellitus continues to grow worldwide. Approximately 44% of new patients entering dialysis are diabetics. Early diagnosis of diabetes and early intervention are critical in preventing the normal progression to renal failure seen in many type 1 and a significant percentage of type 2 diabetics. The prevalence of diabetes is higher in certain racial and ethnic groups, affecting approximately 13% of African Americans, 9.5% of Hispanics, 15% of Native Americans and 15% of Asians, primarily with type 2 diabetes. Approximately 20% to 30% of all diabetics will develop evidence of nephropathy, although a higher percentage of type 1 patients progress to ESRD. Epidemiology The syndrome can be seen in patients with chronic diabetes (15 years or more after onset), so patients are usually of older age (between 50 and 70 years old). The disease is progressive and may cause death two or three years after the initial lesions, and is more frequent in men. Diabetic nephropathy is the most common cause of chronic kidney failure and end-stage kidney disease. People with both type 1 and type 2 diabetes are at risk. The risk is higher if blood-glucose levels are poorly controlled. Further, once nephropathy develops, the greatest rate of progression is seen in patients with poor control of their blood pressure. Also people with high cholesterol level in their blood have much more risk than others. Signs & Symptoms Early signs and symptoms of kidney disease in patients with diabetes are typically unusual. However, a vast array of signs and symptoms listed may manifest when kidney disease has progressed: Swelling, usually around the eyes in the mornings; later, general body swelling may result, such as swelling of the legs; Foamy appearance or excessive frothing of the urine; Unintentional weight gain (from fluid accumulation); Fatigue; Frequent hiccups; General ill feeling; Generalized itching; Headache; Nausea and vomiting; Poor appetite; Weakness, paleness, and anemia; Ankle and leg swelling, leg cramps; Going to the bathroom more often at night High blood pressure Tests and diagnosis Laboratory tests that may be done include: BUN (Blood Urea Nitrogen) Serum creatinine The levels of these tests will increase as kidney damage gets worse. Other laboratory tests that may be done include: 24-hour urine protein Blood levels of phosphorus, calcium, bicarbonate, and potassium Hemoglobin Hematocrit Protein electrophoresis - urine Red blood cell (RBC) count. Complications Possible complications include: hypoglycemia (from decreased excretion of insulin) rapidly progressing chronic kidney failure, end-stage kidney disease, hyperkalemia, severe hypertension, complications of hemodialysis, complications of kidney transplant, coexistence of other diabetes complications, peritonitis (if peritoneal dialysis used), increased infections. About 20% of clients with type II are found to have diabetes nephropathy 5-10 yrs after diagnosis. A consequence of microangiopathy, nephropathy involves damage to and eventual obliteration of the capillaries that supply the glomeruli of the kidney. This damage leads in turn to a complex a pathologic changes and manifestations which includes intercapillary glomerulosclerosis, nephrosis, gross albuminuria and hypertension. Risk factors include poor glycemic control, duration of disease and hypertension. With worsening of the nephrosis, chronic renal failure ensues. Unless the client can be maintained with hemodialysis or receives a renal transplant, uremia eventually causes death. Diabetic nephropathy cannot be cured. However prompt and adequate intervention for renal and bladder infection can prevent these causes of renal failure. Unsuccessfully treated nephropathy progresses to end stage renal disease. Treatment at this point includes hemodialysis, peritoneal dialysis or kidney transplantation. Treatments and drugs
Kimmelstiel-Wilson Syndrome Group O
The goals of treatment are to keep the kidney disease from getting worse and prevent complications. This involves keeping the blood pressure under control (under 130/80). Controlling high blood pressure is the most effective way of slowing kidney damage from diabetic nephropathy. The doctor may prescribe the following medicines to lower the blood pressure: Angiotensin-converting enzyme (ACE) inhibitors and Angiotensin receptor blockers (ARBs). These drugs help reduce the amount of protein in the urine. Many studies have suggested that a combination of these two types of drugs may be best. It is also very important to control lipid levels, maintain a healthy weight, and engage in regular physical activity. Closely monitor the blood sugar levels. Doing so may help slow down kidney damage, especially in the very early stages of the disease. Change diet to help control blood sugar. The doctor may also prescribe medications to help control the blood sugar. Dosage of medicine may need to be adjusted from time to time. As kidney failure gets worse, our body removes less insulin, so smaller doses may be needed to control glucose levels. Urinary tract and other infections are common and can be treated with appropriate antibiotics. Dialysis may be necessary once end-stage renal disease develops. At this stage, a kidney transplant must be considered. Another option for patients with type 1 diabetes is a combined kidney-pancreas transplant. COMPREHENSIVE HEALTH HISTORY Patients Profile NAME: Patient Love AGE: 60 SEX: Female BIRTHDAY: July 23, 1949 CIVIL STATUS: Widowed BIRTHPLACE: Balangiga, Eastern Samar PERMANENT ADDRESS: Brgy. 59, Picas, Sagkahan, Tacloban City NATIONALITY: Filipino RELIGION: Roman Catholic OCCUPATION: Housewife DATE OF ADMISSION: January 7, 2010 ATTENDING PHYSICIAN: Dr. Aloha Saballe CHIEF COMPLAINT: Bipedal edema, pallor, easy fatigability DIAGNOSIS: Diabetic Nephropathy with anemia History of Present Illness On January 2, 2010, patient noticed that her feet were swelling (about 4mm). No other associated symptoms were noted. She took Diamicron as her maintenance medication for Diabetes. No medical consultation was done. On January 7, 2010, patient was scheduled for a regular check-up with Dr. Aloha Saballe and when checked, the result of her blood glucose level was 220 mg/dl and her blood pressure reading was 220/140mmHg. Dr. Saballe advised the patient to stay and be confined at the EVRMC for further evaluation and management. Medical History Patient was diagnosed to be hypertensive when she was 40 years old. She was prescribed with Olmezar 20mg OD as a maintenance drug. On December 2002, as recalled by the patient, she was confined at the EVRMC due to excessive urination and body weakness. She was diagnosed as having Diabetes Mellitus Type 2. Diamicron was given as her medication for her everyday maintenance to which she was compliant. She was advised measures to control her blood glucose level and a scheduled monthly check-up.
Childhood Illnesses: Measles, mumps, chickenpox. Allergies: Patient had no known allergies. Alcohol: Patient drinks 1-2 medium-sized glasses of tuba once every month. Smoking: Patient is a non-smoker. Family History Patient love is the 8th child among 10 siblings. To date, only 3 of them are still alive. Her father was hypertensive and died with unknown cause. Her mother was diagnosed to have DM type II. Four of her siblings were known diabetics which were not properly managed. All of her siblings were hypertensive. Heredofamilial Diseases: Paternal Side: (+) Hypertension, DM Maternal Side: (+) Hypertension, DM, Bronchial asthma Personal/Psychosocial History Patient Love was born and raised in Balangiga, Eastern Samar. She is an elementary graduate. She got married at age 22 to a PUJ driver from Tacloban and since their marriage, they have resided in the city already. They were blessed with four children, 2 boys and 2 girls all of whom have families of their own already. Her husband passed away in 2006 at the age of 56 due to a heart attack. Patient love is a housewife and has a small sari-sari store which helps augment the familys income. At present, her youngest daughter and her family lives with Patient love. Activities: Typical day: Arises at 5 AM, prepares breakfast, cleans the house, tends her store, watches over 2 grandchildren, prepares lunch, takes afternoon naps, gardens, prepares dinner, watches TV, sleeps at 8 PM. Diet: Eats 3 times per day and usual diet consists of 2 cups of rice, fish, sometimes meat, vegetables and 8 glasses of water in one day. Before diagnosed to have diabetes, she consumes 500ml of Coke everyday and is fond of eating sweets such as kalamay on fried banana or camote. She puts a minimum of 2 tbsp of sugar in her coffee everyday. Her day was
Kimmelstiel-Wilson Syndrome Group O
not complete without something sweet in her meal. She modified her diet after she was diagnosed to have DM. Hobbies: Her sari-sari store keeps her busy and she takes care of 2 grandchildren who are living with her. These keep her busy. They sometimes spend weekends by the beach. Interpersonal Relationship: She has not encountered any problem in dealing with other people. She and her children, in-laws, and grandchildren have very good relationships and they share an open line of communication and has not encountered any major argument. Health Perception: She expressed that she has lived with her condition for a long time now. She tries her best to comply with her medication regimen and to follow what her doctor has prescribed her such as diet modification, exercise, and regular check-up. She is still saddened though because she does not live a normal life anymore. There are a lot of restrictions to her which she has to stick by religiously so as to extend her life. She still hopes that her condition gets better. She just prays hard and lifts everything to God. GORDONS HEALTH FUNCTIONING PATTERN Sleep and rest Health perception and management Subjective: Patient verbalized, Waray problema tak pagkaturog, unom ngadto pito ka-oras. Subjective: Patient verbalized, Han bata pa ako, waray ako kaospital bisan makausa. Yana naman la ini nga tikalagas na ako. 40 years old ako han nasabtan nga high blood ngayan ak. Gintagan ak hin Olmezar 20 mg nga tutumarun kada adlaw pampahamubo hin BP. Han December 2002, gindara ak ha ospital kay duro na an ak pag-inihi ngan nanluluya ak. Yakan han ak doctor hataas daw kuno ak sugar ha dugo, may diabetes daw ako. Gintagan liwat ak hin Diamicron 80mg nga nakakaduha pagtumar ha usa ka adlaw. Diri na gihap ak ginpapakaon hin magtam-is ngan duro nga karne. Ngatanan nga ak ginkakaon, sinukol. Ginpapadamu pagkaon hin utan nga linaw-agan. Ginpapaglakat-lakat gihap ako para exercise. Maupay nala liwat nga adi tak mga apo kay nakakagi-os ako kay ako man it nag-aalaga ha ira. Gintatalinguha ko nga masunod ini ngatanan nga ginyakan han ak doctor para man liwat ini hit akon kaupayan ngan para mag-iha pa it ak kinabuhi. Nababaraka gad gihapon ako kay diri ako maaram kun ano it mahihinabo ha akon tungod hini nga ak sakit. Pero kakayanon ko pa kutob hit ak akos ngan sige la it ak pangadi nga kaluuyan ak hit Ginoo. Objective: Poor eye contact, glancing about Activity and exercise Subjective: Patient verbalized, Maupay gad tak pagkita hit ak kalugaringon. Bisan diri kami riko, bisan waray ak magtapos hin college, ok la gihap kami, basta buhi la. Masisiring ko nga maupay an ak pagpatubo han ak mga anak. Ini nga akon kasakit, tungod na gihap ini han ak kawaray Cognition and perception Subjective: Patient verbalized, Nakagraduate ak hin elementary pero waray na ak magapadayon pagskwela kay waray man kami kwarta. Maaram ak bumasa ngan sumurat. Nakakaintindi gihap ak hin tagalog ngan guti ng English. Yana, baga hin malabo naman it ak pagkita, pero maupay pa it ak pagbati. Waray pa liwat ak magpacheck-up ha doktor, diri ak maaram kun kinhanglan ko hin antiyuhos. Objective: Patient is responsive, has intact remote and recent memory, coherent, oriented to time, place, person. Self-perception and self-concept Nutrition and metabolism Subjective: Patient verbalized, Han una mahilig gud ak hin bisan ano nga magtam-is. Nakakaimod ak hin 500ml nga Coke adlaw-adlaw. Diri kumpleto tak adlaw kun diri ak nakakaon hin matam-is. Nakakatulo ak pagkaon ha usa ka adlaw: kan-on nga may sura ngan utan. Mahilig ak hin karne pero nakaon gihap ak hin isda. Nainom ak hin 8 ngadto 10 ka baso hin tubig ha usa ka adlaw. Kasabti nga mayda ko na diabetes, ginyaknan man ak han ak doktor nga kinahanglan magbag-o tak pagkaon, gintalinguha ko nga bagohon. Yana diri na ak nakaon hin duro nga magtam-is ngan duro nga magtambok nga pagkaon. Kun nakaon man ako, talagudti la. Kinhanglan ko disiplinahon tak kalugaringon. Napansin ko gihap nga baga la ako hin pirmi nauuhaw amu tagdaragmu na tak pag-inom hin tubig, mga 15-20 nga baso hin tubig. Elimination Subjective: Patient verbalized, Waray problema tak pagbabawas, nakakausa ha usa ka adlaw. Tak pag-ihi lugod kay tagdaragmu, nakakaunom ha usa ka adlaw, bagan duro ka yellow it kolor tak ihi. Subjective: Patient verbalized, Ako man gud it nagi-os ha balay hit ngatanan nga trabahuun kay it ak anak ngan it iya asawa natrabaho man. Ako it naglilimpyo, nagluluto, may tindahan pa ako, tapos ako gihap it nag-aalaga hit ak mga apo. Maupay na ini nga exercise para ha akon.
Kimmelstiel-Wilson Syndrome Group O
disiplina han burubata pa ako, per okay adi naman ini, maguupay nala ak kutob hit ak kaya. Magaampo la ako ha Ginoo nga kaluuyan ako. Roles and relationships Subjective: Patient verbalized, Waray ako problema tak pakikitungo ha iba nga tawo. Waray ko man kaaway. Maupay tak relasyon hit ak mga anak, hit ira mga asawa, ngan hit akon mga apo ngan kasangkayan. Coping and stress management Subjective: Patient verbalized, Nakakalipay ha akon it ak mga apo. Kun ginkakapoy ako, mabatian ko la nga natawa hira, baga hin nawawara tak kapoy. Nalilibang liwat ako hit ak turutindahanay. Kun may oras it ak anak, nakadto kami ha beach kun sabado di ngani domingo para marelax. Sexuality and reproduction Subjective: Patient verbalized, Namatay naman an ak asawa han 2006 kay na heart attack. Buutan adto nga ak asawa, bisan nagkukuri kami nananalinguha gud la gihap hiya nga mayda kami kaunon ngan mayda isustento han am upat nga anak. Nakakamingaw gad usahay labis na kun nahinumdum ak han am mga mag-upay nga ginbubuhat hadto. Values and beliefs Subjective: Patient verbalized, Nasimba ako kada Domingo, nangangadi liwat ako adlaw-adlaw. Makusog man tak pagtuo hit Ginoo. Yana nga may sakit ako, duro tak pangadi nga buligan ako niya para makaya ko ini nga akon ginaagian yana. Nagdadahum pa gad ako nga matambal ako para humilaba pa tak kinabuhi ngan makabulig pa ha ak mga anak ngan mga apo. PHYSICAL EXAMINATION General survey: Patient Love is a 60 - year old female, anxious and weak but articulates clearly, has poor eye contact and was not in distress. Patient can ambulate but chooses not to because of her perceived weakness. She is fairly groomed and prefers lying in bed with head elevated to 45 degrees. Patient was examined conscious, coherent, oriented as to time, date, person and place. Weight: 140 lbs (64kg), Height: 54 (162.6m) BMI: 24 Vital signs: Blood pressure: 160/90 mmHg (left arm, lying) Temp: 37.5C, Pulse: 66 bpm Skin: Inspection Palpation With dry skin Senile skin turgor Medium brown skin complexion. Pale No active lesions. Respiration: 20 cpm
Nails: Inspection Palpation Hair: Inspection Head: Inspection Eyes: Inspection Normocephalic No lesions, masses, injury, scars, lumps, scaling Symmetric facial features with no weakness, no involuntary movements observed. Black and white, evenly distributed hair. No lice, no nits. Smooth with convex curvature Pale With a slight delay in capillary refill >3 seconds. Dirty untrimmed nails No clubbing of fingers.
Kimmelstiel-Wilson Syndrome Group O
Ears: Inspection Inspection Palpation Trachea at midline. Thyroid nonpalpable, non-tender. No nodules noted. No neck vein engorgement Muscles supple equal in size with full ROM; head centered. No masses, tenderness, lymphadenopathy. Symmetrical With symmetrically aligned eyebrows. Eyelashes are equally distributed. Visual fields full by confrontation. EOMs intact, no nystagmus. No ptosis, lid lag, discharges, or crusting. Corneal light reflex symmetric, no strabismus. Pale palpebral conjunctivae. No periorbital edema. Sclera white, no active lesions or redness; no hemorrhage or exudates. Pupils are round and reactive to light and accommodation or PERRLA. Neck: Pale, dry lips. Pale mucosa and gingivae. With dental caries. With halitosis. Tongue protudes midline, no tremors. Pharynx without exudates. Uvula rises midline on phonation. Tonsils 1+. Gag reflex present. Dysphagia not noted. Voice hoarseness not noted.
Palpation
Symmetrical, equal sized auricles. Color is the same with facial skin. Auricle aligned to outer canthus of the eye. Tympanic membrane pearly gray, landmarks intact, no perforation. No hemorrhage, crusting or swelling. Cerumen noted on ear canals. Pinna has no mass, swelling, scaling, discharge.
Spine and back: No lumps nor tenderness. Mobile and firm, pinna recoil after it is folded. Inspection Palpation Normal spinal profile. Vertebral column no tenderness, no deformity or curvature. No lateral spine deviation, no scoliosis, kyphosis, lordosis noted.
Nose:
Inspection
Palpation
Pale nasal mucosa. Uniform with facial color. Nares patent. Nasal septum intact and at midline. Nose bridge not deviated. No perforation noted. No nasal discharges, no hemorrhage noted.
No tenderness over spines. No CVA tenderness.
Percussion (Back) Positive Kidney Punch
Thorax and Lungs: No sinus tenderness. No lesions, no lumps or masses palpated. Inspection Palpation Symmetric chest expansion.
Mouth:
Inspection
Kimmelstiel-Wilson Syndrome Group O
Symmetrical chest expansion. Percussion Tactile fremitus equal bilaterally. Resonant in all lung fields. Diaphragmatic excursion bilaterally equal. Auscultation Breast: Inspection Bronchovesicular breath sounds in all regions, no crackles, no wheeze, no rhonchi. Tympanic in all quadrants. Palpation Soft, no organomegaly, no masses
Extremities: Inspection Palpation Have minimal ROM Unable to maintain flexion against resistance. (Muscle strength: 3/5) All peripheral pulses present, 2+ and = bilaterally. No calf tenderness noted. Tan in color, no active lesions noted. With Grade 2 pitting bipedal edema. Sole and nails are pale.
Palpation
Generally unequal in size. Uniform skin color and intact. Rounded, everted, equal in size and areola is dark brown in color, and points to the same direction without discharges. Generally symmetric; no retractions or dimpling, no rashes, active lesions or discharge. No active lesions noted.
Neurologic: Alert and oriented to person, place, time. Appearance, behavior, speech is appropriate. Remote and recent memories intact. Cranial nerves II through XII intact. Sensory: pinprick, light touch, vibration intact. Stereognosis, able to identify key.
Heart:
Soft and smooth. Contour and consistency firm and homogenous. No tenderness, masses or nodules noted.
REVIEW OF SYSTEMS GENERAL SURVEY: Patient is pale and weak. Integumentary: HEENT: Pale. Dry skin with Senile skin turgor. With edema No history of skin disease. No itchiness, no active skin lesions.
Auscultation PMI in 5th left ICS at the midclavicular line; no thrills. No extra murmurs heard.
Abdomen: Inspection Skin with no scars or any active lesions. No visible veins. Auscultation Normoactive bowel sounds 16 per minute. No bruits heard. Percussion
Head: No unusually frequent or severe headaches, dizziness, syncope, or vertigo. Eyes:
Kimmelstiel-Wilson Syndrome Group O
Slightly blurred vision. 20/50 OD, 20/70 OS No eyeglasses. No eye pain or exudates. No redness. Ears: No hearing difficulty. No earaches, infections now or as child, no discharge, tinnitus, or vertigo. Nose: No discharges; had at least one cold per year. No sinus pain, nasal obstruction, epistaxis, or allergies to food and drugs. Mouth and throat: Neck: No pain, lumps or swollen glands. No stiffness. With pale , dry lips. Pale mucosa and gingivae. With dental caries. With halitosis. No sores, no lesions. No gum bleeding. No dysphagia, hoarseness, nor sore throat. Voids yellow-colored urine approximately 1 glass (240ml) per void (6-10 times per day) No pain during urination. No hematuria.
Peripheral Vascular: With Grade 2 pitting bipedal edema. No varicosities. No leg cramps.
Musculoskeletal: With muscle weakness (Muscle strength: 3/5) With limitation of movement No joint pain, stiffness, muscle pain. No deformities.
Hematologic: No easy bruising or bleeding.
Respiratory: Eupneic.
Endocrine: With excessive thirst. No heat/cold intolerance. No excessive sweat.
Cardiovascular: With palpitations. Hypertensive. With easy fatigability.
Psychiatric: No flight of ideas, no shifting of mood
Gastrointestinal: Neurologic: No vomiting. No diarrhea. Moves bowel everyday to a formed brown-colored stool. No pain nor rectal bleeding. No heartburn, no indigestion, no abdominal pain. No problems with coordination, difficulty speaking. No memory problems.
Genitourinary:
Kimmelstiel-Wilson Syndrome Group O
LABORATORY STUDIES Clinical Chemistry: January 8, 2010 Serum creatinine Normal 53-106 umol/L Result 111.17 Significance : decreased glumerular filtration rate indicating deteriorating renal function : DM Nephropathy : Diabetes : decreased glumerular filtration/ decreased tubular excretion of urate : decreased Na reabsorption in tubule/ decreased glomerular filtration : hyperkalemia; loss of excretory renal function Hematology: January 7, 2010 Hemoglobin Hematocrit WBC Erythrocytes Leukocytes Platelet Lymphocytes Normal 12 16 g/dL 37% - 47% 5 10 x 109 /L 4.6-6.2 x 1012/L 4.5-10 x 109/L 150-450 x 109/L 0.15-0.35 Result 9.7 28 9.1 x 109 /L 5.43 x 1012/L 6.2 x 109/L 140 x 109/L 0.10 Significance : anemia from failure to produce erythropoietin : Low Hct leads to anemia Normal Normal Normal Slightly decreased; partly due to uremia : anemia
BUN Hgt Uric Acid
1.7 8.3 mmol/L 80 120 mg/dl 140 360 mol/L 135 145 mEq/L 3.5 5 mEq/L
16.3 200 mg/dl 447.1
Sodium
126.9
Potassium
5.7
24-Hour Urine Collection: January 8, 2010 Normal 24-hour urine total volume Urine creatinine 800 1800 mL
Result 2800
Significance : polyuria
Urinalysis: January 7, 2010 Normal Glucose Albumin Negative Negative Result Trace ++ Significance High blood glucose level Albuminuria, may indicate damage to kidneys normal Presence of crystals, RBCs, epithelial cells. normal normal normal : indicates tubular damage/ injury to the kidneys. : Proteinuria normal
500 2000 mg/dl
182
: creatinine is reabsorbed in kidney damage : inability to excrete creatinine in kidney damage :increased protein excretion in impaired kidney function
Creatinine clearance 24-hour urine protein
87 107 mL/min
48
Color Transparency pH Specific gravity WBC Epithelial Cells
Yellow Clear 5.0-9.0 1.010-1.035 0-5/hpf Few
Yellow Turbid 5.0 1.010 3/hpf Moderate
150 mg/24hours
300
DIAGNOSTIC STUDIES X-Ray: January 7, 2010 X-Ray Report: Impression: No significant chest findings.
RBC Bacteria
0 1/hpf Few
3/hpf Few
Ultrasound: January 9, 2010 Impression: Glomerulonephritis
Kimmelstiel-Wilson Syndrome Group O
I. ANATOMY AND PHYSIOLOGY Anatomy and Physiology of the Endocrine Pancreas the pancreas weighs approximately 80 grams (about 3 ounces) and is shaped like a pear. located in the upper abdomen, with the head lying immediately adjacent to the duodenum and the body and tail extending across the midline nearly to the spleen. Only 1 to 2 percent of pancreatic tissue has endocrine function. consists of the islets of Langerhans (approximately one million islets that weigh about one gram in total) The principal function of the endocrine pancreas is the secretion of insulin and other polypeptide hormones necessary for the cellular storage or mobilization of glucose, amino acids, and triglycerides. Glucagon also appears to have a minor effect of enhancing lipolysis of triglyceride in adipose tissue, which could be viewed as an addition means of conserving blood glucose by providing fatty acid fuel to most cells. Control of Glucagon Secretion Two other conditions are known to trigger glucagon secretion:
Elevated blood levels of amino acids, as would be seen after consumption of a protein-rich meal: In this situation, glucagon would foster conversion of excess amino acids to glucose by enhancing gluconeogenesis. Since high blood levels of amino acids also stimulate insulin release, this would be a situation in which both insulin and glucagon are active. Exercise: In this case, it is not clear whether the actual stimulus is exercise per se, or the accompanying exercise-induced depletion of glucose.
Pancreatic islets house three major cell types, each of which produces a different endocrine product:
B. Insulin Structure of Insulin Insulin is a rather small protein, with a molecular weight of about 6000 Daltons. It is composed of two chains held together by disulfide bonds. Biosynthesis of Insulin Insulin is synthesized in significant quantities only in beta cells in the pancreas. The insulin mRNA is translated as a single chain precursor called preproinsulin, and removal of its signal peptide during insertion into the endoplasmic reticulum generates proinsulin. Proinsulin consists of three domains: an amino-terminal B chain, a carboxy-terminal A chain and a connecting peptide in the middle known as the C peptide. Within the endoplasmic reticulum, proinsulin is exposed to several specific endopeptidases which excise the C peptide, thereby generating the mature form of insulin. Insulin and free C peptide are packaged in the Golgi into secretory granules which accumulate in the cytoplasm. When the beta cell is appropriately stimulated, insulin is secreted from the cell by exocytosis and diffuses into islet capillary blood. C peptide is also secreted into blood, but has no known biological activity. Control of Insulin Secretion
Alpha cells (A cells) secrete the hormone glucagon. Beta cells (B cells) produce insulin and are the most abundant of the islet cells. Delta cells (D cells) secrete the hormone somatostatin, which is also produced by a number of other endocrine cells in the body.
A. Glucagon major role in maintaining normal concentrations of glucose in blood has the effect of increasing blood glucose levels. linear peptide of 29 amino acids. Glucagon is synthesized as proglucagon and proteolytically processed to yield glucagon within alpha cells of the pancreatic islets. Proglucagon is also expressed within the intestinal tract, where it is processed not into glucagon, but to a family of glucagon-like peptides (enteroglucagon). Physiologic Effects of Glucagon Glucagon exerts control over two pivotal metabolic pathways within the liver, leading that organ to dispense glucose to the rest of the body:
Glucagon stimulates breakdown of glycogen stored in the liver. When blood glucose levels are high, large amounts of glucose are taken up by the liver. Under the influence of insulin, much of this glucose is stored in the form of glycogen. Later, when blood glucose levels begin to fall, glucagon is secreted and acts on hepatocytes to activate the enzymes that depolymerize glycogen and release glucose. Glucagon activates hepatic gluconeogenesis. Gluconeogenesis is the pathway by which non-hexose substrates such as amino acids are converted to glucose. As such, it provides another source of glucose for blood. This is especially important in animals like cats and sheep that don't absorb much if any glucose from the intestine - in these species, activation of gluconeogenic enzymes is the chief mechanism by which glucagon does its job.
Glucose is transported into the beta cell by facilitated diffusion through a glucose transporter; elevated concentrations of glucose in extracellular fluid lead to elevated concentrations of glucose within the beta cell. Elevated concentrations of glucose within the beta cell ultimately leads to membrane depolarization and an influx of extracellular calcium. The resulting increase in intracellular calcium is thought to be one of the primary triggers for exocytosis of insulin-containing secretory granules. The mechanisms by which elevated glucose
Kimmelstiel-Wilson Syndrome Group O
levels within the beta cell cause depolarization is not clearly established, but seems to result from metabolism of glucose and other fuel molecules within the cell, perhaps sensed as an alteration of ATP:ADP ratio and transduced into alterations in membrane conductance. Increased levels of glucose within beta cells also appears to activate calciumindependent pathways that participate in insulin secretion. of gastric acid and pepsin, lowers the rate of gastric emptying, and reduces smooth muscle contractions and blood flow within the intestine. Effects on the Nervous System Somatostatin is often referred to as having neuromodulatory activity within the central nervous sytem, and appears to have a variety of complex effects on neural transmission. Renal System I. The Kidneys 1. Functions Structure and Synthesis Two forms of somatostatin are synthesized: SS-14 -more potent in inhibiting glucagon release. predominant form produced in the nervous system the sole form secreted from pancreas SS-28 - roughly ten-fold more potent in inhibition of growth hormone secretion intestine secretes mostly SS-28 A. Excretion of metabolic waste products B. Regulation of the volume and composition of extracellular fluids 2. Produces Urine A. Formed from blood B. Varies in composition
C. Somatostatin first discovered in hypothalamic extracts and identified as a hormone that inhibited secretion of growth hormone.
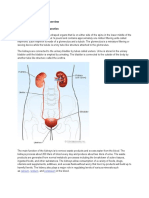
II. Anatomy of the Renal System Both forms of somatostatin are generated by proteolytic cleavage of prosomatostatin, which itself is derived from preprosomatostatin. Two cysteine residules in SS-14 allow the peptide to form an internal disulfide bond. Physiologic Effects "somatostatin inhibits the secretion of many other hormones". Effects on the Pituitary Gland Somatostatin was named for its effect of inhibiting secretion of growth hormone from the pituitary gland. Effects on the Pancreas Somatostatin appears to act primarily in a paracrine manner to inhibit the secretion of both insulin and glucagon. It also has the effect in suppressing pancreatic exocrine secretions, by inhibiting cholecystokinin-stimulated enzyme secretion and secretin-stimulated bicarbonate secretion. Effects on the Gastrointestinal Tract It has been shown to inhibit secretion of many of the other GI hormones, including gastrin, cholecystokinin, secretin and vasoactive intestinal peptide. Somatostatin suppresses secretion 1. Kidney A. Pair of Organs a. Bean-Shaped--Most Species b. Heart-Shaped--Horses c. Lobulated--Cattle B. Retroperitoneal a. Located outside of abdominal cavity suspended by peritoneum C. Blood Supply a. Renal Artery i. Brings blood to Kidney ii. Arises directly from aorta b. Renal Vein i. Brings blood away from Kidney ii. Empties into the Caudal Vena Cava D. Divisions a. Cortex i. Outer Portion ii. Contains upper parts of nephrons --Glomeruli and Tubules b. Medulla i. Inner Portion ii. Contains lower parts of nephrons --Loops of Henle and Collecting Ducts c. Renal Pelvis i. Drains kidney and connects ureters d. Renal Hilus
Kimmelstiel-Wilson Syndrome Group O
i. Indented area on concave edge ii. Ureter, Blood Vessels, Nerves and Lymphatics enter or leave 2. Ureter A. Smooth muscle lined tube B. Conveys urine from renal pelvis to bladder C. Enter bladder through ureterovesicular junction a. Acts as a valve b. Prevents backflow 3. Urinary Bladder A. Hollow muscular organ (Smooth Muscle) B. Stores Urine a. Stretches C. Lined with Transitional Epithelium a. Flatten when filling occurs 4. Urethra A. Caudal continuation of the neck of the bladder B. Conveys urine from bladder to exterior C. Urine release into urethra controlled by external sphincter a. Skeletal muscle III. Microstructure of the Kidney: The Nephron A. The functional unit of the kidney a. Numbers range from 250,000 (cat) to 4 million (cow) per kidney b. Components i. Glomerulus ii. Bowman's Capsule iii. Proximal Tubules iv. Loop of Henle v. Distal Tubules B. Glomerulus a. Tufts of Capillaries b. Afferent arterioles i. Conduct blood to glomerulus c. Efferent arterioles i. Conduct blood away from glomerulus ii. Divide into peritubular capillaries --Surround nephron tubules --Vasa Recta: Peritubular capillaries surrounding loops of Henle d. Function: Filtration of Blood C. Bowman's Capsule a. Collect Glomerular Filtrate b. Conduct filtrate into proximal tubules D. Proximal Tubules a. Conduct filtrate into Loop of Henle b. Very convoluted (Surrounded by Peritubular Capillaries) c. Reabsorb, Secrete, & Excrete --Simple cuboidal or columnar epithelium E. Loop of Henle a. Play important role in concentration of urine b. Divisions i. Descending limb --thin segment --simple squamous epithelium ii. Ascending limb --thick segments --simple cuboidal epithelium --returns to sight near glomerulus of origin c. Surrounded by Vasa Recta F. Distal tubules a. Convey urine from Loops of Henle b. Also convoluted (Surrounded by peritubular capillaries) c. Reabsorb, Secrete, and Excrete --Simple d. Feed Collecting tubules and ducts G. Juxtaglomerular Apparatus a. Junction between distal tube and glomerulus b. Contain unique cell types i. Macula Densa in tubules ii. Juxtaglomerular cells in arterioles c. Function: i. Regulate renal blood flow ii. Regulate glomerular filtration iii. Secrete Renin --Involved in production of angiotensin II H. Two Types of Nephrons a. Cortical i. Short loops of Henle ii. Only go into Outer medulla b. Juxtamedullary i. Long loops of Henle ii. Can go clear to renal pelvis PATHOPHYSIOLOGY
Kimmelstiel-Wilson Syndrome Group O
PROGNOSIS Our patient has a poor prognosis. This is mainly caused by the complications especially macrovascular diseases. In diabetic patients with proteinuria, a decline in the glomerular filtration rate, and hypertension, which collectively constitute the major clinical manifestations of Kimmelstiel-Wilson Syndrome, the mortality risk is 11-fold higher for diabetic men and 18-fold higher for diabetic women when compared with diabetics without nephropathy (our patient is a 60-year old female). In type 1 diabetic patients with end-stage renal disease the patient survival rates could dramatically be improved mainly due to continuous hemodialysis and successful kidney transplantations. The prognosis of type 1 diabetic patients treated with hemodialysis is furthermore significantly poorer than that of kidney transplanted patients. During the last decade simultaneous pancreas and kidney transplantation has become the method of choice for these patients. In type 2 diabetic patients (our patient) with microalbuminuria and overt proteinuria mortality is significantly increased, mainly due to cardiovascular disease. Several arteriosclerosis-related risk factors are more frequently seen in albuminuric patients, especially lipid abnormalities, and hemostatic disturbances. In type 2 diabetic patients with end-stage renal disease the patient survival rate is furthermore extremely low. The prognosis could be improved an inch by hemodialysis or kidney transplantation, however, during the last decade only 8% of type 2 diabetic patients who received either hemodialysis or kidney transplant survived a maximum of 3 years due to ESRD.
RECOMMENDATION The principles of prevention and treatment of Kimmelstiel-Wilson Syndrome are the same. However, the role of each factor could be different in each stage of disease. It is important to define the stage that is the target of intervention (microalbuminuria, proteinuria or GFR) and the outcome of interest. Two recent meta-analyses have demonstrated different results when evaluating different outcomes, such as proteinuria, GFR decline or progression to ESRD. Probably the best treatment is a multiple risk factor interventional approach, but due to a practical point of view each aspect will be addressed individually. The goal to be pursued is retarding the development or progression of DN and to decrease the patients cardiovascular risk and mortality. 1. Keep blood glucose levels as close to normal as possible. The American Diabetes Association recommends that you keep your blood sugar levels at: 90 mg/dL to 130 mg/dL before meals 110 mg/dL to 150 mg/dL at bedtime. Less than 180 mg/dL 1 to 2 hours after meals.
Kimmelstiel-Wilson Syndrome Group O
Three keys to success in monitoring your blood sugar anywhere are: Keeping your meter and supplies with you at all times so that you always have them when you need them. Making it a habit to check your blood sugar level by building it into your routine. Checking your blood sugar meter's accuracy when you visit your doctor by comparing your results with your doctor's results.
2. Maintain blood pressure at less than 130/80 mm Hg with medicine, diet, and exercise. 3. Follow the nutrition guidelines Eating fewer processed foods, such as snack items, luncheon meats, and canned soups, will reduce the amount of sodium in your diet. A diet high in calcium, potassium, and magnesium may lower your blood pressure. A diet high in sodium may cause high blood pressure. To increase the potassium in your diet, fruits and vegetables are excellent sources of this nutrient. Dairy products are high in calcium and magnesium. DASH recommends that you eat 8 to 10 servings of fruits and vegetables and 3 servings of low-fat dairy products each day. Eating a diet low in both saturated fat and total fat will also help lower your blood pressure. Only 30% of your total calories should be from fat, with only 7% to 10% of your fat calories from saturated fat. Saturated fat is found in meats, cheeses, butter, poultry, snack foods, and other processed foods. In general, vegetarian diets reduce blood pressure. The DASH diet could easily be a vegetarian diet if legumes were substituted for meat. Vegetarian diets tend to be higher in potassium, magnesium, and calcium, as is the DASH diet. Vegetarian diets also are higher in fiber and unsaturated fat than other diets. 4. Do not smoke or use other tobacco products. 5. Avoid dehydration by promptly treating other conditionssuch as diarrhea, vomiting, or feverthat can cause it. 6. Treat other conditions that may block the normal flow of urine out of the kidneys, such as kidney stones, an enlarged prostate, or bladder problems. 7. Avoid the use of unprescribed medicines that may be harmful to our kidneys, especially nonsteroidal anti-inflammatory drugs (NSAIDs).
Kimmelstiel-Wilson Syndrome is a chronic complication of DM with a growing incidence. Therefore it is essential to have a better understanding of it, especially in relation to prevention and aggressive management to avoid progression to ESRD. Besides, its direct association with cardiovascular complications makes it imperative to perform intensive, early management of the risk factors. The study of DN has evolved a lot as regards its pathophysiology, stages of renal involvement and, especially, the therapeutic instruments available. Early detection of DN, and the multi-factorial approach targeting the main risk factors may delay progression of kidney disease in DM.
Anda mungkin juga menyukai
- Diabetic Nephropathy, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsDari EverandDiabetic Nephropathy, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsBelum ada peringkat
- Hepatorenal Syndrome, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsDari EverandHepatorenal Syndrome, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsBelum ada peringkat
- Diabetic NephropathyDokumen5 halamanDiabetic NephropathyZiedTriki100% (1)
- Case Analysis Diabetic NephropathyDokumen6 halamanCase Analysis Diabetic NephropathyJassel EscobarBelum ada peringkat
- Avi Suarez Case StudyDokumen7 halamanAvi Suarez Case StudyIva Abigail EspinoBelum ada peringkat
- Diabetic Nephropathy BaruDokumen24 halamanDiabetic Nephropathy BaruRobiyanti Nur Chalifah HattaBelum ada peringkat
- CKD - Case PresDokumen29 halamanCKD - Case PresChristine Joy Ilao PasnoBelum ada peringkat
- Chronic Kidney Disease Stage 5Dokumen21 halamanChronic Kidney Disease Stage 5Kristine Anne Soriano100% (1)
- Case Report On CRF PrtatikDokumen7 halamanCase Report On CRF PrtatikOm PalekarBelum ada peringkat
- Hyperglycemic H-WPS OfficeDokumen4 halamanHyperglycemic H-WPS OfficeShiehan Mae ForroBelum ada peringkat
- Chronic Kidney Disease Case StudyDokumen52 halamanChronic Kidney Disease Case StudyGi100% (1)
- Case Study For Chronic Renal FailureDokumen6 halamanCase Study For Chronic Renal FailureGabbii CincoBelum ada peringkat
- DM NephroDokumen6 halamanDM NephroJulienne Sanchez-SalazarBelum ada peringkat
- Assessing Nutrition in Patients With Chronic Kidney Disease by Kimberly Thompson, MS, RD, LDNDokumen23 halamanAssessing Nutrition in Patients With Chronic Kidney Disease by Kimberly Thompson, MS, RD, LDNCarlos RaiolBelum ada peringkat
- CKD CaseDokumen2 halamanCKD CasePatricia Kaye Tayag CastroBelum ada peringkat
- ST - Lukes Casepress.Dokumen31 halamanST - Lukes Casepress.Justin EduardoBelum ada peringkat
- Diabetes Mellitus HospitalDokumen29 halamanDiabetes Mellitus HospitalgegeBelum ada peringkat
- End Stage Renal DiseaseDokumen62 halamanEnd Stage Renal DiseaseMae Navidas DigdiganBelum ada peringkat
- Diabetic NephropathyDokumen6 halamanDiabetic NephropathyZulfadliZulfadliBelum ada peringkat
- Chronic Kidney DiseaseDokumen54 halamanChronic Kidney DiseaseJosh Matthew RosalesBelum ada peringkat
- Chronic Kidney DiseaseDokumen20 halamanChronic Kidney DiseasePappu HSD VenkateshBelum ada peringkat
- Chronic Kidney DiseaseDokumen7 halamanChronic Kidney DiseasejprakashjjBelum ada peringkat
- Power PointDokumen196 halamanPower PointGrace Dela Rosa MelecioBelum ada peringkat
- The Treatment and Prognosis of Nephrotic Syndrome in The ElderlyDokumen5 halamanThe Treatment and Prognosis of Nephrotic Syndrome in The ElderlysulemanbankerBelum ada peringkat
- Chronic Kidney Diseases (CKD) - 2Dokumen43 halamanChronic Kidney Diseases (CKD) - 2Victoria DadaBelum ada peringkat
- ACR Albumin Creatinine Ratio To Be PRESENTED BY HMN TO THE NEW MED TECH TRAINEES IN JAHRA HEALTH CENTER Apr 23 2014Dokumen22 halamanACR Albumin Creatinine Ratio To Be PRESENTED BY HMN TO THE NEW MED TECH TRAINEES IN JAHRA HEALTH CENTER Apr 23 2014honorinanuguidBelum ada peringkat
- Diabetes Mellitus CommunityDokumen23 halamanDiabetes Mellitus CommunityJoychukzBelum ada peringkat
- Chronic Kidney Disease OverviewDokumen15 halamanChronic Kidney Disease Overviewjames100% (1)
- ImpulogDokumen31 halamanImpulogNimesh ModiBelum ada peringkat
- Renal Case StudyDokumen4 halamanRenal Case Studydsaitta108Belum ada peringkat
- Chronic Kidney DiseaseDokumen55 halamanChronic Kidney DiseaseBentoys Street100% (1)
- Chronic Renal FailureDokumen7 halamanChronic Renal FailureMary Jane Tiangson100% (1)
- Diabetes Mellitus IIDokumen20 halamanDiabetes Mellitus IIMa R Dy100% (1)
- Diabetes Mellitus - ICD-10 CodingDokumen14 halamanDiabetes Mellitus - ICD-10 CodingTim T VargheseBelum ada peringkat
- CHF Secondary To COPD and DM Type 2Dokumen3 halamanCHF Secondary To COPD and DM Type 2Ma Regina Ocio PatanaBelum ada peringkat
- Case Pres 1Dokumen7 halamanCase Pres 1Bright SunshinenBelum ada peringkat
- Chronic Kidney DiseaseDokumen9 halamanChronic Kidney Diseaseluvgrace100% (1)
- By3 Safaa Mohammed Abd-Elghany: V Nder SupervisionDokumen13 halamanBy3 Safaa Mohammed Abd-Elghany: V Nder SupervisiontrrdsBelum ada peringkat
- What Is Hypertensive NephropathyDokumen11 halamanWhat Is Hypertensive NephropathyIvy Pamanian DeldaBelum ada peringkat
- IntroductionDokumen4 halamanIntroductionCristina L. JaysonBelum ada peringkat
- Wa0011.Dokumen180 halamanWa0011.Mohamed AbdelmoniemBelum ada peringkat
- Healing Siddha Medicine-LibreDokumen4 halamanHealing Siddha Medicine-LibreVijay ShanmugasundaramBelum ada peringkat
- Chronic Kidney Disease in HIV Positive PatientsDokumen22 halamanChronic Kidney Disease in HIV Positive PatientsAdityaBelum ada peringkat
- Kidney18 407Dokumen4 halamanKidney18 407api-240473826Belum ada peringkat
- CKD SheetDokumen4 halamanCKD SheetMomy SutanBelum ada peringkat
- Assignment: Applied MedicineDokumen8 halamanAssignment: Applied MedicineKhadija BakhtawarBelum ada peringkat
- Assignment: Applied MedicineDokumen8 halamanAssignment: Applied MedicineKhadija BakhtawarBelum ada peringkat
- Diabetic Kidney Disease (Diabetic Nephropathy) : Yousaf Khan Renal Dialysis Lecturer Ipms-KmuDokumen18 halamanDiabetic Kidney Disease (Diabetic Nephropathy) : Yousaf Khan Renal Dialysis Lecturer Ipms-KmuDani DanyBelum ada peringkat
- I. Multiple Choice: (Type I, Type II)Dokumen2 halamanI. Multiple Choice: (Type I, Type II)greggy_rebel17Belum ada peringkat
- Remediation Essays - NursingDokumen12 halamanRemediation Essays - NursingLouis MatzBelum ada peringkat
- Chronic Kidney DiseaseDokumen6 halamanChronic Kidney Diseaseashi leginBelum ada peringkat
- Nephrotic SyndromeDokumen8 halamanNephrotic SyndromeKanmani AniBelum ada peringkat
- Normal Kidneys and Their Function: Chronic Kidney Disease OverviewDokumen12 halamanNormal Kidneys and Their Function: Chronic Kidney Disease OverviewLouie Collado100% (1)
- Chronic Kidney DiseaseDokumen27 halamanChronic Kidney DiseaseGloria KikiBelum ada peringkat
- Diabetic Nephropathy Case StudyDokumen61 halamanDiabetic Nephropathy Case Studyoxidalaj98% (63)
- Acute Vs Chronic Renal Failure.Dokumen3 halamanAcute Vs Chronic Renal Failure.Stephanie HurewitzBelum ada peringkat
- CKD UnsoedDokumen42 halamanCKD UnsoedSutan Malik IbrahimBelum ada peringkat
- Case StudyDokumen37 halamanCase StudyAnonymous t78m8ku100% (1)
- Diabetes HypertensionDokumen29 halamanDiabetes HypertensionAnimesh PaulBelum ada peringkat
- The Complete Guide to Type 1 Diabetes: Symptoms, Risks, Treatments and Support for DiabeticsDari EverandThe Complete Guide to Type 1 Diabetes: Symptoms, Risks, Treatments and Support for DiabeticsBelum ada peringkat
- Myocardial InfarctionDokumen2 halamanMyocardial InfarctionRanela Kwinkee Pastor SalazarBelum ada peringkat
- SLE PathophysiologyDokumen3 halamanSLE PathophysiologyRanela Kwinkee Pastor Salazar100% (7)
- Case Study Acute Pyelonephritis FINALDokumen20 halamanCase Study Acute Pyelonephritis FINALRanela Kwinkee Pastor SalazarBelum ada peringkat
- Best Photo ContestDokumen2 halamanBest Photo ContestRanela Kwinkee Pastor SalazarBelum ada peringkat
- Nicu CasesDokumen1 halamanNicu CasesRanela Kwinkee Pastor SalazarBelum ada peringkat
- 19-CPS Student Induction Presentation-RAZAK FinalDokumen18 halaman19-CPS Student Induction Presentation-RAZAK FinalbaderBelum ada peringkat
- Chest Drains Al-WPS OfficeDokumen16 halamanChest Drains Al-WPS OfficeisnainiviaBelum ada peringkat
- Px. Abdomen RadiologiDokumen46 halamanPx. Abdomen RadiologiwagigtnBelum ada peringkat
- Campus Advocates: United Nations Association of The USADokumen10 halamanCampus Advocates: United Nations Association of The USAunausaBelum ada peringkat
- GNM Orderno2269 ListDokumen32 halamanGNM Orderno2269 ListPriya singhBelum ada peringkat
- Surgeons in Rangpur, Bangladesh - SOSBDokumen11 halamanSurgeons in Rangpur, Bangladesh - SOSBWadood Md ZahedBelum ada peringkat
- Adolescent GangsDokumen289 halamanAdolescent GangsReuben EscarlanBelum ada peringkat
- The Tactical Combat Casualty Care Casualty Card - TCCC Guidelines - Proposed Change 1301Dokumen9 halamanThe Tactical Combat Casualty Care Casualty Card - TCCC Guidelines - Proposed Change 1301Paschalis DevranisBelum ada peringkat
- Trauma Tapping Technique (TTT) : Purpose: Defuses and Desensitizes Traumatic Memories and ReDokumen3 halamanTrauma Tapping Technique (TTT) : Purpose: Defuses and Desensitizes Traumatic Memories and ReMays Bags100% (1)
- PI e UREA 15Dokumen2 halamanPI e UREA 15dewi asnaniBelum ada peringkat
- Endocrine SystemDokumen41 halamanEndocrine SystemAtteya Mogote AbdullahBelum ada peringkat
- Tonometry and Care of Tonometers PDFDokumen7 halamanTonometry and Care of Tonometers PDFAnni MuharomahBelum ada peringkat
- Alwyn Naicker - Research Proposal Assignment 2 PDFDokumen15 halamanAlwyn Naicker - Research Proposal Assignment 2 PDFAlwyn NaickerBelum ada peringkat
- Fundamentals of Nursing - Nurse As ProfessionDokumen7 halamanFundamentals of Nursing - Nurse As ProfessionDarryl C. LocañasBelum ada peringkat
- VtM2nded1 Page PDFDokumen1 halamanVtM2nded1 Page PDFJoseph CortesBelum ada peringkat
- DNAlysis MygeneRx Sample ReportDokumen13 halamanDNAlysis MygeneRx Sample ReportTrustBelum ada peringkat
- Non Hodgkin Lymphoma Complete PDFDokumen104 halamanNon Hodgkin Lymphoma Complete PDFelika dwiBelum ada peringkat
- Prevention of Foodborne Disease: The Five Keys To Safer FoodDokumen2 halamanPrevention of Foodborne Disease: The Five Keys To Safer FoodThong Boon LeongBelum ada peringkat
- The Filipino Activity GuideDokumen9 halamanThe Filipino Activity GuideMark Joseph Maquiling100% (1)
- Alloimmunization in Pregnancy: Brooke Grizzell, M.DDokumen40 halamanAlloimmunization in Pregnancy: Brooke Grizzell, M.DhectorBelum ada peringkat
- Fatimah Kti FixDokumen71 halamanFatimah Kti FixOktaviani MuhaddistBelum ada peringkat
- Pseudo Bezoar in An Elderly ManDokumen4 halamanPseudo Bezoar in An Elderly Manchidiebere peter echiehBelum ada peringkat
- Star Health ProjectDokumen52 halamanStar Health ProjectParvat Patil83% (6)
- Psychological Well Being - 18 ItemsDokumen5 halamanPsychological Well Being - 18 ItemsIqra LatifBelum ada peringkat
- Hypnotherapy To Reduce Post-Stroke Muscle Tension in IndonesiaDokumen4 halamanHypnotherapy To Reduce Post-Stroke Muscle Tension in IndonesiaEndangAhmatBelum ada peringkat
- Concepts Underlying The Design of Health ProgramsDokumen31 halamanConcepts Underlying The Design of Health ProgramsMUTHUKUMARAN100% (1)
- Summary of Chapters NFPA 1852: Standard On Selection, Care, and Maintenance of Open-Circuit Self-Contained Breathing Apparatus (SCBA)Dokumen2 halamanSummary of Chapters NFPA 1852: Standard On Selection, Care, and Maintenance of Open-Circuit Self-Contained Breathing Apparatus (SCBA)Rolando CerezoBelum ada peringkat
- 10Dokumen19 halaman10Bruno100% (3)
- Ankylosing Spondylitis Disease Activity ScoreDokumen2 halamanAnkylosing Spondylitis Disease Activity ScoreDebarshi DasBelum ada peringkat
- Tetanus Treatment: Vitria DwiayuDokumen12 halamanTetanus Treatment: Vitria DwiayuvitriaBelum ada peringkat