Formal Report Distillation of ALcoholic Beverages
Diunggah oleh
patricia_moran_4Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Formal Report Distillation of ALcoholic Beverages
Diunggah oleh
patricia_moran_4Hak Cipta:
Format Tersedia
Organic Chemistry Laboratory Formal Report
Determination of percent ethanol and percent loss in Sombrero Tequila using simple distillation
CHEMISTR Y
200 L
Maria Patricia R. Moran*, Serra Nicole Marcelino, Miguel Gabriel Ocampo, John Edward Paz
Department of Biological Sciences, College of Science
*Corresponding author; e-mail: trixiemoran@yahoo.com
Abstract
In this experiment, the percent ethanol and percent loss was calculated through simple distillation of a 20mL sample of Sombrero Tequila and a flammability test. After the distillation process, 12.5mL of distillate was collected at 98C. Through the conduction of the flammability test, it was determined that 6.0mL of ethanol was collected. After the necessary computations, the percent ethanol was determined to be 30% and the computed percent loss was 36.25%.
Keywords: simple distillation, flammability test, percent ethanol, percent loss
Introduction Alcohol is a very important compound with varied functions ranging from commercial, medical and even industrial uses. This compound is a family of organic compounds with the characteristic functional group of hydroxyl (OH) (Figure 1) attached to a sp 3hybridized carbon (Klein, 2012).
Organic Chemistry Laboratory Formal Report
Figure 1. Structure of ethanol
One of the earliest applications of distillation would be concentrating the alcohol content of alcoholic beverages. Tequila is an example of an alcoholic beverage called spirits that is of Mexican origin and are made from the distillation of fermented juice of agave plants. The alcohol content of spirits range from 40-50% that is very much higher compared to wine (8-14%). Ethanol, a type of alcohol, is considered to be the active ingredient on alcoholic beverages (Figure 1). Production of ethanol usually involves fermentation, a process of metabolism of carbohydrates by yeast in an anaerobic environment. Unfortunately, excessive intake of these alcoholic beverages may produce biological effects that may harm the body. Liver damage may one of the leading effects of alcoholism that include symptoms of fever, abdominal pain and jaundice, or the yellowing of the skin. Drinking alcohol in moderation is found to have a beneficial effect on the heart, yet abuse would entail an increase in the blood pressure and an increased risk for heart attack and stroke. Mild effects would include heightened mood, slurred speech and delayed reaction times that could lead to violence or inappropriate behavior (NHMRC, 2009).
Organic Chemistry Laboratory Formal Report
Distillation is a method of separating liquids that has been practiced for thousands of years. Specifically, distillation is the process of separating compounds through the utilization of the differences in volatility, or the tendency to vaporize, of the liquids (Winkle, 1967). The basic principle of distillation concerns that the vapor produced through heating has a different composition compared to the liquid. Since the more volatile liquid is more likely to be vaporized first, the vapor produced is richer in terms of the more volatile component compared to the liquid (Palleros, 2000). Conversely, the less volatile component is richer in composition in the liquid. Ethanol boils at 79C and water boils at 100C. Therefore in this experiment, ethanol would be considered as the more volatile component and water as the less volatile component. Basically, there are two types of distillation: simple and fractional distillation. A simple distillation consists of a round-bottom flask, where the liquid is placed, distillation head, which connects the flask to the condenser, with a thermometer and an adapter that connects condenser to the receiving flask (Palleros, 2000). The set-up for simple distillation is shown in Figure 2.
Organic Chemistry Laboratory Formal Report
Figure 2. Simple distillation set-up
Figure 3. Fractional distillation set-up
As for the fractional distillation, the same set-up as with simple distillation is also required but there is an addition of a fractional column that is to be inserted between the
Organic Chemistry Laboratory Formal Report
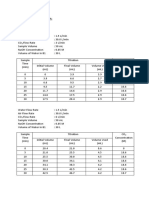
distillation flask and distillation head. This fractional column functions to provide a larger surface area for the mixture to be constantly vaporized and condensed (Palleros, 2000). One major difference between the two is that the simple distillation works efficiently with volatile liquids whose boiling point difference is very large usually of about 25C. Furthermore, it is advised that fractional distillation should be used when the boiling point difference is lower than this value. Distillation is a very cheap and convenient method of separating liquids in mixtures, yet one major disadvantage could be seen when handling liquids with very small difference in volatility or boiling point. The purpose of this experiment is to separate and calculate in percentage the alcohol content of a commercial alcoholic beverage, Tequila Sombrero, by distillation process and to compare the efficiency of simple and fractional distillation techniques. Results and discussion The table below shows the results of the conducted experiment, both the distillation process and the flammability test. Table 1. Volume, temperature and flammability test results of the distillate Volume of Distillate (mL) 0 0.5 1.0 1.5
Test Tube No. 1 1 2 3
Temperature 60C 73C 78C 80C
Flammability Test + + + +
5
Organic Chemistry Laboratory Formal Report
4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 9.5 10.0 10.5 11.0 11.5 12.0 12.5 81C 81C 81C 81C 82C 83C 83C 84C 85C 87C 88C 90C 92C 94C 95C 97C 97C 97C 97C 98C 98C 98C + + + + + + + + + -
Table 1 shows the direct relationship between the volume of the distillate and temperature. Therefore, as the volume increases, the temperature also increases. As observed in the results of the flammability test, there is a clear distinction between the
6
Organic Chemistry Laboratory Formal Report
12th and 13th test tubes that is indicated in red ink. A + sign was recorded up until the 12th test tube since the distillate produced a blue flame. This entails that up until the 12th test tube, ethanol is the more dominant compound of the distillate; knowing that ethanol is flammable. As the distillate reaches the 13th test tube, a - sign was recorded to signify that there was no flame produced. This could be explained by the fact that water became the more dominant thereby prohibiting the distillate from being flammable. Therefore, as the distillation process continues, the amount of ethanol produced in the distillate decreases (Palleros, 2000).
Figure 4. Relationship between temperature (oC) and volume (mL) of the distillate
Figure 4 shows the graph of the temperature in relation with the volume of the distillate. It can also be observed that towards the end of the graph where the temperature is nearing 100C, the temperature stopped increasing and remained constant. This constancy is called azeotrope, which is a mixture of two liquid that has a constant
7
Organic Chemistry Laboratory Formal Report
boiling point and composition throughout the distillation process. The azeotrope has a lower boiling point compared to the either of the individual components, so it should be completely distilled before the distillation of the excess component occurs (Pastos, 1998). Under distillation, two principles are present, namely: Daltons Law and Raoults Law. Daltons law of partial pressure states that for a mixture to boil, the sum of the partial pressures must be equal to the atmospheric pressure (Chang, 1986). On the other hand, Raoults law states that the change in vapor pressure of a substance can be determined by the product of the mole fraction of the substance multiplied by the vapor pressure of the pure substance. The formulas used for computing the percent ethanol and the percent loss were:
Using the formulas above, the percent alcohol obtained was,
Organic Chemistry Laboratory Formal Report
and the percent loss was,
Therefore, it was computed that the percent ethanol was 30% and that the percent ethanol lost was 36.25%. This percent loss may be attributed to different sources of error such as the mishandling of the distillate, excessive heating of the flask or evaporation. In conclusion, using fractional distillation is a much better option compared to simple distillation especially if handling liquids with close boiling point difference. The difference in boiling point of the two compounds (ethanol and water) would be 22C, therefore it should be requiring a fractional distillation set-up. Fractional distillation gives a much better separation of liquids since there is repetition of the vaporization and condensation process.
Experimental methodology Before the conduction of the experiment, the necessary materials were gathered such as the quick-fit distillation set-up, Sombrero Tequila sample, Bunsen burner, two boiling chips, thermometer and test tubes. All test tubes were calibrated 0.5mL using a marking
Organic Chemistry Laboratory Formal Report
pen. The set-up for simple distillation (Figure 2) was assembled, where glycerol was used to grease the joints to avoid vapor loss on the duration of the experiment. In the pear-shaped distilling flask, 20mL of the Sombrero brand tequila together with two boiling chips was placed. Using a Bunsen burner, the distilling flask was heated with the flame constantly being rotated around the flask. As the first drop of distillate was
collected in the first test tube, the initial reading for the temperature was recorded. Then, 0.5mL of the distillate was gathered and the corresponding temperature was recorded. In the same way, 0.5mL of the distillate was collected in the succeeding test tubes and the corresponding temperature was simultaneously recorded. The flame was removed after the temperature reading reached 98C to avoid the vaporization of water. The set up was cooled and the remaining liquid, also known as the residue, was poured into a graduated cylinder. The volume was recorded. As for the distillate contained in the test tubes, two drops of each were placed on a watch glass and underwent the flammability test, where a match a was used to see if the distillate will ignite or not. A + sign was used to signify flammability and - sign is used otherwise. The results were tabulated and a graph was constructed to determine the relationship between temperature and the volume of the distillate.
The percent ethanol was computed using the formula :
In addition, the percent loss was computed using the formula:
10
Organic Chemistry Laboratory Formal Report
References Chang, R. General Chemistry; Random House: New York, 1986. Hengstebeck, R.J. Distillation: Principles and Design Procedures; Chapman and Hall: London, New York, 1964.
11
Organic Chemistry Laboratory Formal Report
Kister, H.Z. Distillation Design; McGraw-Hill: New York, 1992. Klein, D.R. Organic Chemistry; Wiley: Hoboken, N.J., 2012. Lands, W.E. The Americal Journal of Clinical Nutrition. 1995, 62, 1101S-1106S. National Health and Medical Research Council (NHMRC). Australian Guidelines to Reduce Health Risks from Drinking Alcohol; Australia, 2009. Palleros, D.R. Experimental Organic Chemistry; Wiley: New York, 2000. Pasto, D.J. Experiments and Techniques in Organic Chemistry; Prentice-Hall: Englewood Cliffs, N.J., 1998. Prado-Ramirez, R. International Journal of Food Science and Technology. 2005, 40, 701-708. Winkle, M. Distillation; McGraw-Hill: New York, 1967. http://chemistry.about.com/cs/5/f/bldistillation.htm (retrieved on Dec 16, 2012)
http://www.chem.umass.edu/~samal/269/distill.pdf (retrieved on Dec 16, 2012)
12
Anda mungkin juga menyukai
- Standardization of Volumetric GlasswareDokumen8 halamanStandardization of Volumetric GlasswareJIEHASMARTBelum ada peringkat
- CSTRDokumen20 halamanCSTRSharing Caring100% (1)
- Determination of The Solubility Product Constant For Calcium HydroxideDokumen4 halamanDetermination of The Solubility Product Constant For Calcium HydroxideJasmeetSingh0% (2)
- MSOCHA3 Tutorial 1 Multicomponent AbsorptionDokumen5 halamanMSOCHA3 Tutorial 1 Multicomponent AbsorptionTshwarelo MahlakoaneBelum ada peringkat
- Soap Lab Report by ADDokumen12 halamanSoap Lab Report by ADAD50% (4)
- Q2015 Physical Chemistry Measurements Laboratory Chemistry Department, Campus Monterrey Practice # 8 Distribution CoefficientDokumen4 halamanQ2015 Physical Chemistry Measurements Laboratory Chemistry Department, Campus Monterrey Practice # 8 Distribution Coefficientandres_guadiana_7362100% (1)
- Plug Flow Reactor ExperimentDokumen16 halamanPlug Flow Reactor ExperimentN Afiqah RazakBelum ada peringkat
- Diffusivity of Liquid Into LiquidDokumen8 halamanDiffusivity of Liquid Into LiquidZahraa GhanemBelum ada peringkat
- Title: Aim: Date: Name: Matriculation Number: Serial Number: Group: Experiment Number: Level: Course Code: Instructor'S NameDokumen14 halamanTitle: Aim: Date: Name: Matriculation Number: Serial Number: Group: Experiment Number: Level: Course Code: Instructor'S NameJim0% (1)
- PH Determination and Buffer Preparation FRDokumen6 halamanPH Determination and Buffer Preparation FRFelipe VillanuevaBelum ada peringkat
- Gas Absorption: Determining Drag and Flooding FlowsDokumen5 halamanGas Absorption: Determining Drag and Flooding FlowsDean Joyce AlborotoBelum ada peringkat
- Chapter 4Dokumen43 halamanChapter 4aliBelum ada peringkat
- Fractional DistillationDokumen5 halamanFractional Distillationihack_101Belum ada peringkat
- J. Lab ReportDokumen3 halamanJ. Lab ReportJylla AngwayBelum ada peringkat
- Review Distillation PDFDokumen35 halamanReview Distillation PDFsupercubos1Belum ada peringkat
- Saponification LabDokumen5 halamanSaponification LabDeshan WolfeyBelum ada peringkat
- Extractive Distillation ReportDokumen17 halamanExtractive Distillation ReportAmanda Brown100% (1)
- Electroplating Experiment Determines Faraday's ConstantDokumen3 halamanElectroplating Experiment Determines Faraday's ConstantMaiSakurajima100% (1)
- Packed Distillation Column ExperimentDokumen20 halamanPacked Distillation Column ExperimentChan Chun ChenBelum ada peringkat
- Workshop IDokumen6 halamanWorkshop IValentina GonzálezBelum ada peringkat
- Spectrophotometric Analysis of AspirinDokumen4 halamanSpectrophotometric Analysis of Aspirinhamid_hinnaBelum ada peringkat
- Sample Lab Report Distillation of Ethyl AlcoholDokumen4 halamanSample Lab Report Distillation of Ethyl AlcoholLysandwr75% (4)
- Solubility of Organic CompoundsDokumen4 halamanSolubility of Organic CompoundsJeremy Noceda100% (1)
- CSTR 40LDokumen16 halamanCSTR 40LhishamBelum ada peringkat
- Optimal Operation of A Semi-Batch Reactive Distillation Column (2000)Dokumen7 halamanOptimal Operation of A Semi-Batch Reactive Distillation Column (2000)GodofredoBelum ada peringkat
- Plate Column Distillation EfficiencyDokumen7 halamanPlate Column Distillation EfficiencyVijay PrasadBelum ada peringkat
- Lab #2 - Hydrolysis of Ethyl Acetate & Reaction Rates - FinalDokumen13 halamanLab #2 - Hydrolysis of Ethyl Acetate & Reaction Rates - FinalMargaritavillejack83% (6)
- Intro CSTRDokumen6 halamanIntro CSTREmmanuel PlazaBelum ada peringkat
- Acid ValueDokumen2 halamanAcid ValueSyed Mohamed Yusuf100% (1)
- Adsorption IsothermDokumen4 halamanAdsorption Isothermahkiujtsw0% (1)
- Fractional Distillation Lab ReportDokumen7 halamanFractional Distillation Lab ReportOmar AlasBelum ada peringkat
- Synthesis of Methyl SalicylateDokumen2 halamanSynthesis of Methyl SalicylateMich Tolentino0% (1)
- Batch Distillation ExperimentDokumen8 halamanBatch Distillation ExperimentJonelou CusipagBelum ada peringkat
- Partition Coefficient DeterminationDokumen4 halamanPartition Coefficient DeterminationMostafa HamawandyBelum ada peringkat
- P4E2: Kinetics of Homogeneous Reaction in Batch and Continuous Stirred-Tank Reactor at Two Different TemperatureDokumen7 halamanP4E2: Kinetics of Homogeneous Reaction in Batch and Continuous Stirred-Tank Reactor at Two Different TemperaturejayaprinaBelum ada peringkat
- Process Flowsheeting BasicsDokumen93 halamanProcess Flowsheeting BasicsYunaida YusoffBelum ada peringkat
- Experiment: Packed Distillation ColumnDokumen4 halamanExperiment: Packed Distillation Columnnhalieza1067Belum ada peringkat
- Azeotropic and Extractive DistillationDokumen9 halamanAzeotropic and Extractive DistillationShahzaib IqbalBelum ada peringkat
- Diagram/ Image:: Experiment Number: 02Dokumen10 halamanDiagram/ Image:: Experiment Number: 02Roshan Dhikale100% (1)
- Distillation Report - Rotation 1Dokumen23 halamanDistillation Report - Rotation 1Рада МарјановићBelum ada peringkat
- Determination of Sugar As GlucoseDokumen4 halamanDetermination of Sugar As GlucoseIpsita ChakravartyBelum ada peringkat
- Regulation of climacteric and non-climacteric fruit ripening pathways in melonDokumen7 halamanRegulation of climacteric and non-climacteric fruit ripening pathways in melonsandraosouzaBelum ada peringkat
- Heat Solution LabDokumen6 halamanHeat Solution LabNicole Graham50% (2)
- IYOHA COLLINS 16CF020531 Batch Reactor ReportDokumen19 halamanIYOHA COLLINS 16CF020531 Batch Reactor ReportDavid OvieBelum ada peringkat
- VLE of Methanol-Water MixtureDokumen14 halamanVLE of Methanol-Water MixtureHafiniHambaliBelum ada peringkat
- Determination of Fluoride Concentration Using Ion Selective ElectrodeDokumen7 halamanDetermination of Fluoride Concentration Using Ion Selective ElectrodeAmanda WangBelum ada peringkat
- Phase Diagram of a Three-Component Liquid SystemDokumen7 halamanPhase Diagram of a Three-Component Liquid SystemEllaine TejadaBelum ada peringkat
- Partial Molar Volume of Ethanol-Water SolutionsDokumen24 halamanPartial Molar Volume of Ethanol-Water SolutionsIlyas Hassan100% (1)
- Acetylation Ferrocene 2012Dokumen3 halamanAcetylation Ferrocene 2012VargasArn50% (2)
- Measuring Viscosity with an Ostwald ViscometerDokumen8 halamanMeasuring Viscosity with an Ostwald ViscometerJohn Andrae MangloBelum ada peringkat
- Gas Absorption Pressure DropDokumen26 halamanGas Absorption Pressure DropNemo Cyzar LiisBelum ada peringkat
- Drying: Merry Jessah S. TorresDokumen6 halamanDrying: Merry Jessah S. TorresFrancis Val FranciscoBelum ada peringkat
- Analysis of A Mixture of Carbonate and BicarbonateDokumen2 halamanAnalysis of A Mixture of Carbonate and BicarbonateCharles Trono RacadioBelum ada peringkat
- Vapor Liquid EquilibriumDokumen7 halamanVapor Liquid Equilibriummahbub1332100% (1)
- Enzyme ConcentrationDokumen16 halamanEnzyme ConcentrationSya Subi100% (1)
- Gravimetric Determination of SulfateDokumen3 halamanGravimetric Determination of SulfateArmiee InfiniteBelum ada peringkat
- C4 Lab ReportDokumen11 halamanC4 Lab ReportchaitanyaBelum ada peringkat
- Gas Absorption Lab ReportDokumen3 halamanGas Absorption Lab ReportNur Shaffikha Azmi100% (1)
- EsterificationDokumen5 halamanEsterificationJoonsuk LeeBelum ada peringkat
- Deep Palmar ArchDokumen2 halamanDeep Palmar Archpatricia_moran_4Belum ada peringkat
- Catrina, Piano Bar and The Grand Ballroom) Are Designed To Meet All Your Needs Whether ForDokumen6 halamanCatrina, Piano Bar and The Grand Ballroom) Are Designed To Meet All Your Needs Whether Forpatricia_moran_4Belum ada peringkat
- Behavioral Medicine: Case Discussion 1Dokumen2 halamanBehavioral Medicine: Case Discussion 1patricia_moran_4Belum ada peringkat
- SEA Camp Application FormDokumen3 halamanSEA Camp Application Formpatricia_moran_4Belum ada peringkat
- Insight Nature Diversity e StabilityDokumen6 halamanInsight Nature Diversity e Stabilitypatricia_moran_4Belum ada peringkat
- Report On Philippine Compliance To MEAsDokumen48 halamanReport On Philippine Compliance To MEAspatricia_moran_4Belum ada peringkat
- Parkinson Booth and Lee 2012Dokumen7 halamanParkinson Booth and Lee 2012patricia_moran_4Belum ada peringkat
- Diversity StabilityDokumen15 halamanDiversity Stabilitypatricia_moran_4Belum ada peringkat
- Managing Laguna de Bay through the Laguna Lake Development AuthorityDokumen34 halamanManaging Laguna de Bay through the Laguna Lake Development AuthorityKatrina OblefiasBelum ada peringkat
- 246554Dokumen9 halaman246554patricia_moran_4Belum ada peringkat
- Lake Taal 1Dokumen7 halamanLake Taal 1patricia_moran_4Belum ada peringkat
- 08 JGVR Ayta Legend PaperDokumen7 halaman08 JGVR Ayta Legend Paperpatricia_moran_4Belum ada peringkat
- Daily Growth Increments Ôbserved M Otoliths From Juvenile East Baltic CodDokumen9 halamanDaily Growth Increments Ôbserved M Otoliths From Juvenile East Baltic Codpatricia_moran_4Belum ada peringkat
- Morales Nin 2000Dokumen15 halamanMorales Nin 2000patricia_moran_4Belum ada peringkat
- Diokno BudgetDokumen49 halamanDiokno BudgetDianne BausaBelum ada peringkat
- 04schiemer 063-080Dokumen18 halaman04schiemer 063-080patricia_moran_4Belum ada peringkat
- Room2room MondayDokumen1 halamanRoom2room Mondaypatricia_moran_4Belum ada peringkat
- Map DocxkjnDokumen2 halamanMap Docxkjnpatricia_moran_4Belum ada peringkat
- Comparison of Different Otolith Shape Descriptors and Morphometrics00Dokumen13 halamanComparison of Different Otolith Shape Descriptors and Morphometrics00patricia_moran_4Belum ada peringkat
- PAhuSCIyan Project ProposalDokumen4 halamanPAhuSCIyan Project Proposalpatricia_moran_4Belum ada peringkat
- Effects of Habitat Destruction, Domestic Dog Attacks, Bushfires and Road AccidentsDokumen2 halamanEffects of Habitat Destruction, Domestic Dog Attacks, Bushfires and Road Accidentspatricia_moran_4Belum ada peringkat
- Homework For AP Physics BDokumen41 halamanHomework For AP Physics BMuhammad Waseem SoomroBelum ada peringkat
- Map DocxkjnDokumen2 halamanMap Docxkjnpatricia_moran_4Belum ada peringkat
- Anatomy and PhysiologyDokumen2 halamanAnatomy and Physiologypatricia_moran_4Belum ada peringkat
- Item PricesDokumen1 halamanItem Pricespatricia_moran_4Belum ada peringkat
- Sardines OtolitheregDokumen7 halamanSardines Otolitheregpatricia_moran_4Belum ada peringkat
- Scarlet Constitution 2014Dokumen13 halamanScarlet Constitution 2014patricia_moran_4Belum ada peringkat
- Computer Final ReqnknjojoDokumen5 halamanComputer Final Reqnknjojopatricia_moran_4Belum ada peringkat
- App Application SheetDokumen4 halamanApp Application Sheetpatricia_moran_4Belum ada peringkat
- Weekly class scheduleDokumen1 halamanWeekly class schedulepatricia_moran_4Belum ada peringkat
- Asma Zaghbani Publications ScientifiquesDokumen2 halamanAsma Zaghbani Publications Scientifiquesapi-227010260Belum ada peringkat
- Hydrology - Groundwater Net Flow RateDokumen16 halamanHydrology - Groundwater Net Flow RatedbircsBelum ada peringkat
- Virtual Anchor Length - KBRDokumen22 halamanVirtual Anchor Length - KBRSammar Adhikari100% (2)
- Baker Safe-T-DataDokumen1 halamanBaker Safe-T-DataJonathan Saviñon de los SantosBelum ada peringkat
- Cell Membrane Transport: A Guide to Passive and Active ProcessesDokumen2 halamanCell Membrane Transport: A Guide to Passive and Active ProcessesZei-chen LocabenBelum ada peringkat
- Choline Chloride in Clay and Shale Stabilizer-619Dokumen4 halamanCholine Chloride in Clay and Shale Stabilizer-619Lê Công100% (1)
- HSChemistry StudyGuideDokumen49 halamanHSChemistry StudyGuidetoshiroelric07Belum ada peringkat
- MCAT 2015 - MiniTest - Ebook PDFDokumen36 halamanMCAT 2015 - MiniTest - Ebook PDFamelinaBelum ada peringkat
- Trimelitic AcidDokumen151 halamanTrimelitic AcidTrimerMixBelum ada peringkat
- Calculations and units in analytical chemistryDokumen17 halamanCalculations and units in analytical chemistryangelaBelum ada peringkat
- Fluid Mechanics IIDokumen26 halamanFluid Mechanics IIarunajsBelum ada peringkat
- Chapter 4 - Plasticity of SoilDokumen15 halamanChapter 4 - Plasticity of SoilHussein EssaBelum ada peringkat
- Formation of The Heavy ElementsDokumen17 halamanFormation of The Heavy ElementsSheiloiBelum ada peringkat
- 2023-JEE Advanced-2-PAPER-2Dokumen16 halaman2023-JEE Advanced-2-PAPER-2Devansh GoelBelum ada peringkat
- Madharasathul Ahmadhiyya: First Term ExaminationDokumen9 halamanMadharasathul Ahmadhiyya: First Term Examinationafoo1234Belum ada peringkat
- Conservation of Energy Lab AlternativeDokumen3 halamanConservation of Energy Lab Alternativeapi-292550476Belum ada peringkat
- Remediation of Contaminated Soil and GroundwaterDokumen3 halamanRemediation of Contaminated Soil and GroundwaterTamires NassarBelum ada peringkat
- Culinary Biophysics On The Nature of The 6X°C Egg 2011Dokumen8 halamanCulinary Biophysics On The Nature of The 6X°C Egg 2011Kenesei GyörgyBelum ada peringkat
- Newlands Law of Octaves Periodic TableDokumen5 halamanNewlands Law of Octaves Periodic TablePaarth Saxena X-B RNBelum ada peringkat
- Heat Transfer in Fire Tube Boiler Software SumulationDokumen7 halamanHeat Transfer in Fire Tube Boiler Software SumulationLiam Moylan50% (2)
- (Physics) 2010 TSSM Unit 4 ExamDokumen37 halaman(Physics) 2010 TSSM Unit 4 ExamakashBelum ada peringkat
- IL-6 Testing ProtocolDokumen7 halamanIL-6 Testing ProtocolUMMID WashimBelum ada peringkat
- Pengaruh Rentang Suhu Distilasi Fraksinasi Terhadap Kadar Patchouli Alcohol (PA) Pada Minyak NilamDokumen7 halamanPengaruh Rentang Suhu Distilasi Fraksinasi Terhadap Kadar Patchouli Alcohol (PA) Pada Minyak NilamBagusSatriyaBelum ada peringkat
- Physics of Aqueous Phase Evolution in Plutonic EnvironmentsDokumen11 halamanPhysics of Aqueous Phase Evolution in Plutonic Environmentsjunior.geologia100% (1)
- Book 1 System 2016Dokumen155 halamanBook 1 System 2016annis99Belum ada peringkat
- Pelican Cases Catalog 2021Dokumen29 halamanPelican Cases Catalog 2021thanakornBelum ada peringkat
- 7solutions Datasheet Gaztox enDokumen2 halaman7solutions Datasheet Gaztox enForum PompieriiBelum ada peringkat
- Answer Tutorial - Pharmaceutical CalculationsDokumen11 halamanAnswer Tutorial - Pharmaceutical CalculationsNur Nabilah50% (2)
- Plastic ExtrusionDokumen35 halamanPlastic ExtrusionOff Campus100% (2)
- Factors Affecting Solubility and Rate of Solution NotesDokumen3 halamanFactors Affecting Solubility and Rate of Solution Notesscribduser0811972Belum ada peringkat