Alvarado Final Paper
Diunggah oleh
Hồ Thắng0 penilaian0% menganggap dokumen ini bermanfaat (0 suara)
24 tayangan18 halamanHak Cipta
© © All Rights Reserved
Format Tersedia
PDF, TXT atau baca online dari Scribd
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PDF, TXT atau baca online dari Scribd
0 penilaian0% menganggap dokumen ini bermanfaat (0 suara)
24 tayangan18 halamanAlvarado Final Paper
Diunggah oleh
Hồ ThắngHak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PDF, TXT atau baca online dari Scribd
Anda di halaman 1dari 18
BIO-MEDIATED SOIL IMPROVEMENT: CEMENTATION OF
UNSATURATED SAND SAMPLES
Submitted to: NEES Inc.
Submitted by: Daniel Alvarado
Home Institution: Arizona State University
Host Institution: University of California, Davis
PhD Advisor: Jason DeJong
2
Table of Contents
1 Introduction ............................................................................................................. 3
2 Methodology............................................................................................................ 5
3 Results.................................................................................................................... 11
4 Discussion.............................................................................................................. 16
5 Acknowledgements................................................................................................ 17
Figures
Figure 1: Loose Sand to Sandstone............................................................................. 3
Figure 2: Bio-Mediated Calcite Precipitation (DeJong et al) ..................................... 5
Figure 3: Shear Wave Signal ...................................................................................... 6
Figure 4: Scanning Electron Microscope.................................................................... 7
Figure 5: Distribution of Calcite on Sand Grain Particles .......................................... 7
Figure 6: Initial Load Frame Setup (not to scale) ....................................................... 9
Figure 7: Percolation Test 2 Setup............................................................................ 10
Figure 8: Test Cell with Bender Element ................................................................. 10
Figure 9: Test Cells with Bender Elements and Split Spoon Aquarium Rock......... 10
Figure 10: pH vs Time .............................................................................................. 11
Figure 11: Shear Wave Velocity............................................................................... 12
Figure 12: Cameco Unconfined Compression 1....................................................... 13
Figure 13: Cameco Unconfined Compression 1....................................................... 13
Figure 14: SEM Ottawa 50-70 before Bio-Soil Treatment....................................... 14
Figure 15: SEM Ottawa 50-70 after Bio-Soil Treatment.......................................... 14
Figure 16: SEM Ottawa 50-70 after Bio-Soil Treatment X 10.00k zoom on calcite
structures at particle to particle contact .................................................................... 15
Figure 17: SEM Ottawa 50-70 after Bio-Soil Treatment X 2.00k zoom on calcite
structures at particle to particle contact .................................................................... 15
Equations
EQ. 1: Net Urea Hydrolysis Reaction
EQ. 2: Net pH increase: [OH-] generated from NH4+ production >> [Ca2+]
EQ. 3: Shear Wave Velocity V (m/s)
Abstract
Bio Mediated Soil Improvement (Bio-Soil) is new and innovative research within
geotechnical engineering which can be used in the fields of earthquake engineering and
liquefaction prevention. During events of cyclic loading from earthquakes and other
events, liquefaction in loose sands can occur, causing foundation deformation and/or
failure. The Bio-Soil method is an interdisciplinary field consisting of collaboration with
the studies of microbiology, geochemistry, and civil engineering to find natural
treatments for ground improvement. In this process, technically termed as Microbially
Induced Calcite Precipitation, calcium carbonate is precipitated within the sand particles
to form bonds; therefore the process transforms loose sand susceptible to liquefaction into
sandstone. Laboratory findings, observations, and test results are presented along with
future plans of optimization, up scaling, and transferring Bio-Soils into practical
application.
3
1 Introduction
1.1 Current Soil Improvement Practice
With over 40,000 yearly projects and approximately $6 billion/ year worldwide in the
industry, new environmentally friendly techniques for soil improvement have become
necessary (DeJong et al). Current grouting and ground improvement techniques in
practice include grouting via cement, chemical, compaction, fracture and jet, micro piles,
jacked piers, driven piers, ground anchors, shoring, soil nailing vibro compaction,
concrete columns and piers (Hayward Baker). Focusing on just grouting with the
exception of sodium silicate, almost all of these manmade synthetic chemical grouting
techniques are hazardous and/or toxic (DeJong et al).
1.2 Background and Motivation for Research
The purpose of the Bio-Mediated Soil Improvement research is to find a way to use
bacteria produced calcium carbonate to strengthen cohesive soils in the attempt to
eliminate the risk of liquefaction and generally increase the stability of soil during events
such as earthquakes, landslides, etc. Liquefaction is a geotechnical phenomenon which
occurs mostly in unconsolidated saturated cohesive soils such as loose sands and silts. In
the event of liquefaction, a soils consistency may go from a solid state to having the
properties of a heavy liquid. This occurs from the rise of pore water pressures during
cyclic undrained loading or softening (e.g. an earthquake) (Ishihara 353). The soils
effective stress decreases as each grain of sand or silt is suspended and surrounded by a
thin layer of water. Water has no shear strength which in effect causes structures to sink
until the displaced soil matches its weight (Youd).
With current practice of synthetic man made grouting techniques being harmful to the
environment and people, the Bio-Soil method is being studied as a natural solution to
synthetic grouting. Bacteria is harnessed to help prevent liquefaction and possibly used
for other applications by forming calcite structures within the sand particles to increase
the stiffness of the soil.
Figure 1: Loose Sand to Sandstone
Loose Sand Sandstone
4
1.3 Overview of Use of Calcite Precipitation to Increase Soil Strength
Bio soils are an integrated observation and experimentation between geotechnical
engineering, microbiology, and chemistry. Bio mediated soil improvement is the process
in which a bacteria precipitates calcium carbonate within a soil sample in attempt to
increase its shear strength and overall resistance to liquefaction. Sporosarcina Pasteurii
, used to precipitate the calcite, is an aerobic bacterium which is found to naturally occur
in soil deposits (Fritzges). Since the bacteria are innate to the earth, it may not pose
environmental risk in ideas of future in field use (Fritzges).
When the bacteria are microbially induced, meaning it is controlled biologically; it can
precipitate calcite through the chemical process and alter the engineering properties of
loose sand. Outlined by EQ 1-2 and displayed in Fig. 1 is the chemical process involved
in the precipitation of calcite throughout a typical sand sample during the biological
treatment process. The main catalyst for the precipitation of calcite and food for the
bacteria is the Urea Broth Solution (ubroth) consisting of variable concentrations of
NaHCO
3
, NH
4
Cl, CaCl
2
, Urea, and Bacto (trademarked various blend of nutrients).
Microbially Induced Calcite Precipitation (MICP) is a chemical process in which the
bacteria consumes and breaks down urea to form ammonia, bicarbonate and carbonate
ions. The calcium ions within the ubroth solution fed to the bacteria are then free to bond
with the carbonate to form a level of cementation on each sand grain. This makes a
more cohesive bond within the soil sample particle matrix as it is one of the most reactive
and common minerals found in the earths surface (Morse). During this process the
ammonia plays an important role as it helps increase the pH making an ideal environment
for the bacteria to feed on the urea and precipitate calcite (Fritzges).
Net Urea Hydrolysis Reaction: NH
2
CONH
2
+3H
2
O 2NH
4
+
+ HCO
3
-
+ OH
-
EQ. 1
Net pH increase: [OH-] generated from NH4+ production >> [Ca2+] EQ. 2
5
Figure 2: Bio-Mediated Calcite Precipitation (DeJong et al)
2 Methodologies
2.1 Process Monitoring and Collecting Data
Characteristics of liquefaction in soils have been determined by a combination of
laboratory tests of undisturbed samples and in situ tests. The in situ tests used include the
standard penetration test (SPT), the cone penetration tests, and the dilatometer test.
Considering these all are in situ penetration tests that are somewhat unreliable at different
depths and unfeasible for lab testing, shear wave velocity measurement has also been
used (Tokimasu 33).
Shear Wave Velocity
The shear wave velocity is a property of soil that can help identify density and more
directly stiffness (Lee). It is used directly in liquefaction analysis and to identify the
general characteristics of a soil in both the lab and in situ testing (Tokimasu 33). A
standard loose sand may have a shear velocity between 100-200 m/s. A liquefiable soil is
any soil falling under a shear velocity of 500 m/s. The goal of the MICP is to raise that
shear wave velocity above 500 m/s and to stay in the range of 500-1000 m/s with the
properties more associated with that of sandstone. This shear wave velocity is the time
measured using bender elements in a sample to propagate a wave and measure the return
of that wave through the sample using an oscilloscope (Fritzges). Fig. 3 shows a typical
shear wave signal achieved during a Bio-Soil treatment. The bottom of the first arrival
time is recorded as seen in Fig. 3 as T and inserted into a conversion equation EQ 1. A
Bio-Soil process that is precipitating calcite will have a gradual increase in shear wave
NH2CONH2 + H2O 2NH3 + CO2
2NH3 + 2H2O 2NH4
+
+ 2OH
-
CO2 + OH
-
HCO3
-
Net Urea Hydrolysis Reaction: NH
2
CONH
2
+3H
2
O 2NH
4
+
+ HCO
3
-
+ OH
-
- - - - - - - - - - - - - - - - -
-
-
-
-
-
-
-
-
Ca
2+
Ca
2+
+ HCO3
-
+ OH
-
CaCO3 + H2O
Net pH increase: [OH
-
] generated from NH
4
+
production >> [Ca
2+
]
Bacteria Cell: Sporosarcina pasteurii
Particle
NH3
CO
2
Ca
2+
attracted
to Cell
-
H
2
O
H
2
O
H
2
O
H
2
O
CaC0
3
CaC0
3
CaC0
3
6
velocity in relation to the short time (2-4 hours, 1 treatment) after the pH reaches an
average of ideal pH 9.
V (m/s) = [Tip to Tip Distance of Bender Elements (mm)]/[(T (sec)-5)/1000] EQ. 3
Figure 3: Shear Wave Signal
pH
PH is monitored as a technique to check if biological activity within the sand sample is
occuring. The effluent of each sample is measured using a pH strip after it has been
sitting in the sample for the alloted time (approx 1.5 -2.5 hours) as a reasurrance of
calcite precipitating in the sand sample. The ideal range of pH falls between 8.5 and 9.3,
specifically at 9 for bacteria to precipitate calcite.
SEM
In addition to the pH and bender element readings Scanning Electron Microscope (SEM)
observations have been conducted on extruded samples to compare with data from the
pumped samples. The SEM (see Fig. 4) is a tool used in most material sciences when the
magnification is needed to have more contrast. How the SEM works is that it images the
sample surface by scanning it with a high energy beam of electrons in a certain pattern
called the raster scan pattern much like in a television. The scan gives us clear images of
the topography of the item observed or as in this case the sand particles and calcite
structures. The ideal distribution of calcite would be having calcite precipitated only at
the particle contact points only as seen in Fig. 2. What actually happens though is most
of the calcite precipitating with a layer of calcite structures forming on the sand particles.
Fig. 11-14 in the Results section displays the SEM of what the percolation methods
treatment accomplished as somewhat more preferential distribution. The SEM also gives
us a clear image of where the bacteria was precipitating calcite and an idea of how dense
and dispersed the bacteria was throughout the sample.
7
Figure 4: Scanning Electron Microscope
Figure 5: Distribution of Calcite on Sand Grain Particles
The data collected from both percolation device tests will give a better comparison of a
non saturated sample to a saturated sample which is treated with calcite precipitation
along with the effects of increasing the concentration of the ubroth.
Unconfined Compression and Flow Rates
Uniform Preferential
Actual
Distribution Alternatives
8
Future additional data collected for the unconfined compression and flow rates of the
percolation method in comparison to the pumping method will give valuable data on
comparing to the two methods for optimization. Further tests can also be conducted after
extruding the sample and performing triaxial and/or direct shear testing (DeJong et al).
2.2 Set-up for the Percolation Device
Several methods and variations of those methods have been used for the microbial
induced cementation process at the UC Davis Soil Interactions Laboratory. The testing is
still at the small scale laboratory stage with optimization as the primary objective. The
current method of pumping with complete saturation has been used with several
variations of continuous pumping with effluent, reverse directional pumping, nutrient
circulation, and using pH and bender element readings to observe the shear wave velocity
and calcite precipitation as process monitoring techniques. An additional test of
observing percolation and the bacterias calcite precipitation through a soil sample will
display valuable data to compare when a sample is not completely saturated. An
important factor to note within the percolation method is that the ubroth concentration
has also been increased which may have effected the results of calcite precipitated.
2.3 Test 1: 4 day Percolation Test
The percolation device is set up much like the standard 6 inch cells that have been used
for previous tests. Four 12 cells with a 2 diameter were assembled and observed for
the first percolation test. To begin, 3 large porous stones were to be used for each cell.
One stone was placed on the bottom cap with the cell then connecting to the bottom cap.
The soil sample was then pluviated into the cell at 4.25 inches tall. The following layer
consisted of two more porous stones with a drilled top cap resting above (see Figure 6).
The test cell was then placed in the load frame with 45 lbs of confining stress. A tube for
effluent was attached and the test began (see Figure 6).
9
Figure 6: Initial Load Frame Setup (not to scale)
The percolation device test 1 spanned over a 4 day testing period. For the day 1 test a
batch of bacteria with the new ubroth concentration was poured through at approximately
.75 pore volumes. A three hour set time was observed just as the ubroth 1 without
bacteria was allowed to percolate through. At 1.5 hours after the new ubroth 1 was
introduced, the effluent was then poured through the sample and allowed to percolate
through for another 1.5 hours. During this time, pH readings were made to make sure
that precipitation was occurring along with observation of percolation using different
colored dyes. After this 3 hour time for the ubroth 1 a new batch of ubroth 2 was made
and allowed to percolate through at the same method. This process continued until 12
AM where day 1 was complete. Day 2 began the next day at 6 AM and observed the
same process excluding the initial bacteria ubroth. This process continued for days 3 and
4 with the same procedure. At the end of day 4 the sample was then allowed to be
extruded, cleaned, and observed for calcite precipitation. The final step was to clean up
the equipment and gather the effluent pH data and observations for analysis.
2.4 Percolation Test 2: Bender Elements
The percolation device test 2 is identical to the original 4 day span test with the addition
of bender elements on the cells and test time decrease to 48 hours (see Figures 7-9).
Ottawa 20-30, 50-70 and Aquarium Rock are more poorly graded sands or samples used
in Test 2 with bender elements. Cameco being a more well graded sand is also sampled
with bender elements for Test 2. As pH readings are being taken the bender elements are
used to collect shear wave velocities throughout the test. In addition to the 4 bender
4.25
3
1.5
12
8.25
45lbs
10
element cells, several other cells were set up to observe flow rates, pH, and allow for
more extruded samples to be measured with unconfined compression. Data is to be
collected and analyzed to compare the change in shear wave velocity as calcite is
precipitated over time.
Figure 7: Percolation Test 2 Setup
Figure 8: Test Cell with Bender Element
Figure 9: Test Cells with Bender Elements and Split Spoon Aquarium Rock
11
3 Results
Using the process monitoring techniques of pH levels, shear wave velocity, unconfined
compression, flow rates, and scanning electron microscopy (SEM), valuable data has
been collected to help optimize the biological process of calcite precipitation. The
following results and data observations were made from Percolation Test 2 conducted
over 48 hours.
Observing Fig. 6 the pH reached the optimum range pH between 8.5 and 9.3 after
approximately 20 hours of treatment. This steady measurement averaging out to a pH of
9 is the ideal environment for the bacteria to precipitate calcite.
pH
7.0
7.5
8.0
8.5
9.0
9.5
0.00 4.00 8.00 12.00 16.00 20.00 24.00 28.00 32.00 36.00 40.00 44.00 48.00
Time (hr)
p
H
Aquarium Rock
Ottawa 20-30
Cameco
Ottawa 50-70
Figure 10: pH vs. Time
12
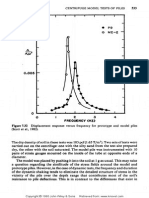
Observing Figure 11 the shear wave velocity of 500 m/s, beyond the limit of a liquefiable
soil, correlates with the ideal pH level of 9 as most of the test samples reach over that
level at about 25 hrs into the test. The highest shear wave velocity of 1600 m/s occurs in
the Ottawa 20-30 sample which is a poorly graded sand sample.
Shear Wave Velocities
0.0
200.0
400.0
600.0
800.0
1000.0
1200.0
1400.0
1600.0
1800.0
0.00 4.00 8.00 12.00 16.00 20.00 24.00 28.00 32.00 36.00 40.00 44.00 48.00
Time (hr)
S
h
e
a
r
W
a
v
e
V
e
l
o
c
i
t
y
(
m
/
s
)
Aquarium Rock
Ottawa 20-30
Cameco
Ottawa 50-70
Figure 11: Shear Wave Velocity
Figures 12 (psi) and 13 (kPa) display the unconfined compression results obtained from
the Cameco sand sample. The maximum stress of approximately 300 psi and strain of
1.39% was achieved before any cracking or deformation occurred.
13
Cameco Unconfined Compression
0.00
50.00
100.00
150.00
200.00
250.00
300.00
350.00
0.00% 0.20% 0.40% 0.60% 0.80% 1.00% 1.20% 1.40% 1.60%
Strain (%)
S
t
r
e
s
s
(
p
s
i
)
Figure 12: Cameco Unconfined Compression 1
Cameco Unconfined Compression
0.00
500.00
1000.00
1500.00
2000.00
2500.00
0.00% 1.00% 2.00% 3.00% 4.00% 5.00% 6.00% 7.00%
Strain (%)
S
t
r
e
s
s
(
k
P
a
)
Figure 13: Cameco Unconfined Compression 1
14
The SEM images (Figures 14-17) display the before and after affect of Bio-Soil treatment
for the Ottawa 50-70 sample for the Percolation Test 2. Figure 14 displays the gaps and
voids in which water can fill during an earthquake leading to liquefaction. Figure 15 is
an overview of the treated sand sample with calcite precipitated throughout the soil
sample. Closely observing the Ottawa 50-70 from the zoom on the calcite structures (see
Figures 16-17) the preferential precipitation of calcite at the particle contacts can be seen.
Also, observing Fig. 17 the bacteria indentations can be seen as being dispersed
throughout the entire calcite structure. It can be roughly assumed that with these SEM
observations that calcite was evenly distributed throughout the Ottawa 50-70 sample.
Figure 14: SEM Ottawa 50-70 before Bio-Soil Treatment
Figure 15: SEM Ottawa 50-70 after Bio-Soil Treatment
15
Figure 16: SEM Ottawa 50-70 after Bio-Soil Treatment X 10.00k zoom on calcite
structures at particle to particle contact
Figure 17: SEM Ottawa 50-70 after Bio-Soil Treatment X 2.00k zoom on calcite
structures at particle to particle contact
16
4 Discussion
4.1 Optimization
The main goal of research to be achieved in the 2008 summer REU program was
optimization of the laboratory Bio-Soil treatment methods. The first month involved
pumping treatments through fully saturated samples. The next 6 weeks involved the
percolation of treatments through unsaturated samples with increased ubroth
concentrations. It has been observed that the percolation method has displayed results of
stiffer extruded sand samples. It is still unknown if these results occur from the actual
method of percolation or the fact that the ubroth concentration increase may give the
bacteria more to feed on and precipitate calcite or possibly a combination of both.
Calculations of the number of particle to particle contacts and volume of liquid retained
at those contacts are currently being conducted as a way to better optimize and
understand the effect of unsaturated media and varying the concentration of ubroth.
4.2 Up scaling
Recent funding for the up scaling to centrifuge modeling has been approved for future
research studies of Bio-Soil treatments. The UC Davis centrifuge will be used in
conjunction with the findings from laboratory tests from the SIL to move closer to
practical field applications.
4.3 Future Research and Goals
Some purpose to the further conduction of these tests may also come from the correlation
of shear wave velocity to liquefaction resistance having fairly new findings with limited
field testing (Tokimasu 34). There have been recent advances in the research of bio
mediated soil improvement to allow for more accurate measurements of bacteria calcite
precipitation and up-scaling to centrifuge experiments (DeJong et al). The accuracy may
increase from the measurement of the ratio between urea injected and urea found in the
effluent measured by a spectrometer as the centrifuge tests will give more in field
results (DeJong et al). The Nesslerization Method of using spectrometer readings from
ammonia in ubroth effluent will assist in the optimization of both laboratory tests and
centrifuge modeling for the Bio-Soil process. The end result may be achieved in the
possible future replacement of chemical and synthetic man made grouting techniques of
today and with the natural, more environmentally friendly Bio-Mediated Soil
Improvement techniques.
17
5 Acknowledgements
The research conducted at the University of California, Davis is supported by the George
E. Brown, Jr. Network for Earthquake Engineering Simulation (NEES) with funding
provided by the National Science Foundation (NSF). A special thanks is extended out to
NEES CEO Steve McCabe and all NEES staff for offering opportunities to
undergraduates to obtain research experience and including but not limited to the Soil
Interactions Laboratory PI Jason DeJong, Centrifuge PIs Bruce Kutter and Dan Wilson,
graduate student mentors Brina Mortensen, Brian Martinez, Matt Weil, Robbie Jaeger
and Nick Yafrate, and undergraduate student researcher Jack Waller.
18
References
DeJong, Jason et al [2008]. Bio-Mediated Soil Improvement, 1
st
International
Conference on Bio-Geo-Civil Engineering. Delft, The Netherlands.
Fritzges, Michael B. [2005]. Biologically Induced Improvement of the Response of
Sands to Monotonic Loading, M.S. thesis, Department of Civil & Environmental
Engineering., University of Massachusetts, Amherst, Massachusetts.
Ishihara, K. [1993]. Liquefaction and flow failure during earthquakes,
Geotechnique 43, No. 3, 351-415
Hayward Baker. Services. 2003. 9 Sept 2008
<http://haywardbaker.com/services/index.htm>
Lee, J.S., and Santamarina, J.C. [2007]. Seismic monitoring short-duration events:
liquefaction in 1g models, Canada Geotech. J. 44: 659-672
Morse, J.W. [1983]. The Kinetic of Calcium Carbonate Dissolution Precipitation,
Carbonates: Geology and Chemistry, 227-264
Tokimatsu, K., and Uchida, A. [1990]. Correlation Between Liquefaction Resistance
And Shear Wave Velocity, Soils and Foundations, Vol. 30, No.2, 33-42
Youd, T.L., and Idriss, I.M. [2001]. "Liquefaction Resistance of Soils: Summary report
from the 1996 NCEER and 1998 NCEER/NSF Workshops on Evaluation of
Liquefaction Resistance of Soils", Journal of Geotechnical and
Geoenvironmental Engineering, ASCE, 127(4), 297-313
Anda mungkin juga menyukai
- Specifications, Supervision, and InspectionDokumen23 halamanSpecifications, Supervision, and InspectionHồ ThắngBelum ada peringkat
- Dke78 Ch19Dokumen30 halamanDke78 Ch19Hồ ThắngBelum ada peringkat
- Special Applications of Chemical GroutsDokumen14 halamanSpecial Applications of Chemical GroutsHồ ThắngBelum ada peringkat
- Grouting To Shut Off SeepageDokumen25 halamanGrouting To Shut Off SeepageHồ ThắngBelum ada peringkat
- Dke78 Ch18Dokumen26 halamanDke78 Ch18Hồ ThắngBelum ada peringkat
- Field Procedures and TestsDokumen18 halamanField Procedures and TestsHồ ThắngBelum ada peringkat
- Grout Curtains: Chap. 11Dokumen30 halamanGrout Curtains: Chap. 11Hồ ThắngBelum ada peringkat
- Dke78 Ch10Dokumen30 halamanDke78 Ch10Hồ ThắngBelum ada peringkat
- Dke78 Ch11Dokumen78 halamanDke78 Ch11Hồ ThắngBelum ada peringkat
- Dke78 Ch13Dokumen39 halamanDke78 Ch13Hồ ThắngBelum ada peringkat
- Dke78 Ch12Dokumen8 halamanDke78 Ch12Hồ ThắngBelum ada peringkat
- Dke78 Ch9Dokumen21 halamanDke78 Ch9Hồ ThắngBelum ada peringkat
- Determining The Intrinsic Compressibility of Fine-Grained SoilsDokumen17 halamanDetermining The Intrinsic Compressibility of Fine-Grained SoilsHồ ThắngBelum ada peringkat
- 07bDokumen56 halaman07bHồ ThắngBelum ada peringkat
- Piling, Nailing, and MixingDokumen10 halamanPiling, Nailing, and MixingHồ ThắngBelum ada peringkat
- Water Removal and WellpointingDokumen12 halamanWater Removal and WellpointingHồ ThắngBelum ada peringkat
- Dke78 Ch2Dokumen27 halamanDke78 Ch2Hồ ThắngBelum ada peringkat
- Answer The Question - Writing Task 2Dokumen4 halamanAnswer The Question - Writing Task 2Hồ ThắngBelum ada peringkat
- Numerical and Analytical Analysis of Geogrid Reinforced Soil Wall Subjected To Dynamic LoadingDokumen1 halamanNumerical and Analytical Analysis of Geogrid Reinforced Soil Wall Subjected To Dynamic LoadingHồ ThắngBelum ada peringkat
- Viswanadh Pen PortDokumen1 halamanViswanadh Pen PortHồ ThắngBelum ada peringkat
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDari EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RacePenilaian: 4 dari 5 bintang4/5 (895)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDari EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifePenilaian: 4 dari 5 bintang4/5 (5794)
- Shoe Dog: A Memoir by the Creator of NikeDari EverandShoe Dog: A Memoir by the Creator of NikePenilaian: 4.5 dari 5 bintang4.5/5 (537)
- Grit: The Power of Passion and PerseveranceDari EverandGrit: The Power of Passion and PerseverancePenilaian: 4 dari 5 bintang4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)Dari EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Penilaian: 4 dari 5 bintang4/5 (98)
- The Little Book of Hygge: Danish Secrets to Happy LivingDari EverandThe Little Book of Hygge: Danish Secrets to Happy LivingPenilaian: 3.5 dari 5 bintang3.5/5 (400)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDari EverandNever Split the Difference: Negotiating As If Your Life Depended On ItPenilaian: 4.5 dari 5 bintang4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDari EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FuturePenilaian: 4.5 dari 5 bintang4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDari EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryPenilaian: 3.5 dari 5 bintang3.5/5 (231)
- Rise of ISIS: A Threat We Can't IgnoreDari EverandRise of ISIS: A Threat We Can't IgnorePenilaian: 3.5 dari 5 bintang3.5/5 (137)
- The Emperor of All Maladies: A Biography of CancerDari EverandThe Emperor of All Maladies: A Biography of CancerPenilaian: 4.5 dari 5 bintang4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDari EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaPenilaian: 4.5 dari 5 bintang4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDari EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersPenilaian: 4.5 dari 5 bintang4.5/5 (345)
- On Fire: The (Burning) Case for a Green New DealDari EverandOn Fire: The (Burning) Case for a Green New DealPenilaian: 4 dari 5 bintang4/5 (74)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDari EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyPenilaian: 3.5 dari 5 bintang3.5/5 (2259)
- Team of Rivals: The Political Genius of Abraham LincolnDari EverandTeam of Rivals: The Political Genius of Abraham LincolnPenilaian: 4.5 dari 5 bintang4.5/5 (234)
- The Unwinding: An Inner History of the New AmericaDari EverandThe Unwinding: An Inner History of the New AmericaPenilaian: 4 dari 5 bintang4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDari EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You ArePenilaian: 4 dari 5 bintang4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Dari EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Penilaian: 4.5 dari 5 bintang4.5/5 (121)
- Her Body and Other Parties: StoriesDari EverandHer Body and Other Parties: StoriesPenilaian: 4 dari 5 bintang4/5 (821)
- VT 300dDokumen3 halamanVT 300dAndrei IulianBelum ada peringkat
- Budi PekertiDokumen13 halamanBudi PekertiAPRI100% (1)
- Crüe, Chatting Done CorrectlyDokumen7 halamanCrüe, Chatting Done Correctlyctjw.financeBelum ada peringkat
- #4 Nov.7-11, 2016 DLLDokumen3 halaman#4 Nov.7-11, 2016 DLLRoselyn MyerBelum ada peringkat
- Catalogo GreenfieldDokumen24 halamanCatalogo GreenfieldTeodoroBelum ada peringkat
- Them As Foreigners': Theme 2: OtherizationDokumen3 halamanThem As Foreigners': Theme 2: OtherizationNastjaBelum ada peringkat
- Siwes Report CHISOM SAM ORJIDokumen41 halamanSiwes Report CHISOM SAM ORJISOMSKI1100% (2)
- Parts of The Microscope Quiz PDFDokumen2 halamanParts of The Microscope Quiz PDFEnriele De GuzmanBelum ada peringkat
- Trainer Competency ModelDokumen4 halamanTrainer Competency ModelpattapuBelum ada peringkat
- TFG Lost in Intercultural Translation (Beta)Dokumen25 halamanTFG Lost in Intercultural Translation (Beta)Oxo AfedoxoBelum ada peringkat
- Reaction Paper Failon Ngayon: Kalamidad Sa Pabahay Failon Ngayon: Barangay Health StationsDokumen4 halamanReaction Paper Failon Ngayon: Kalamidad Sa Pabahay Failon Ngayon: Barangay Health StationsRomeo Chavez BardajeBelum ada peringkat
- Assignment 1 FRS651 Mac-July 2020Dokumen2 halamanAssignment 1 FRS651 Mac-July 2020aizatBelum ada peringkat
- Statement of PurposeDokumen3 halamanStatement of PurposeSamarth NegiBelum ada peringkat
- Risk Matrix Going On African Photo SafariDokumen15 halamanRisk Matrix Going On African Photo SafariScribdTranslationsBelum ada peringkat
- Significance Testing (T-Tests)Dokumen3 halamanSignificance Testing (T-Tests)Madison HartfieldBelum ada peringkat
- Traditions of The North American Indians, Vol. 1 (Of 3) by Jones, James AthearnDokumen125 halamanTraditions of The North American Indians, Vol. 1 (Of 3) by Jones, James AthearnGutenberg.org100% (2)
- FCOE Directory 2015-16Dokumen196 halamanFCOE Directory 2015-16Cheryl WestBelum ada peringkat
- Types of EconomyDokumen2 halamanTypes of EconomyAyush GargBelum ada peringkat
- Learning Plan FinalDokumen3 halamanLearning Plan FinalMary AnalynBelum ada peringkat
- Amazon Web ServicesDokumen85 halamanAmazon Web ServicesSrinivasa Reddy KarriBelum ada peringkat
- Voting PDFDokumen227 halamanVoting PDFrapsjadeBelum ada peringkat
- Quality Procedures and Forms 2016Dokumen30 halamanQuality Procedures and Forms 2016Mohammed Musa100% (9)
- For All Sessions, Refer To The Downloaded Course: Customized PlanDokumen7 halamanFor All Sessions, Refer To The Downloaded Course: Customized PlanVishal MalhotraBelum ada peringkat
- Pierderea Auzului Cauzata de Afectarea Transmiterii Sunetelor (Surditatea de Transmisie) : SunteleDokumen3 halamanPierderea Auzului Cauzata de Afectarea Transmiterii Sunetelor (Surditatea de Transmisie) : SunteleBianca AndreeaBelum ada peringkat
- I PU Geography English Medium PDFDokumen211 halamanI PU Geography English Medium PDFMADEGOWDA BSBelum ada peringkat
- PLC & HMI Interfacing For AC Servo Drive: Naveen Kumar E T.V.Snehaprabha Senthil KumarDokumen5 halamanPLC & HMI Interfacing For AC Servo Drive: Naveen Kumar E T.V.Snehaprabha Senthil KumarNay Ba LaBelum ada peringkat
- Introduction To Media ART: Course: History of Media Art Instructor: Zeib JahangirDokumen17 halamanIntroduction To Media ART: Course: History of Media Art Instructor: Zeib JahangirLiaqat AleeBelum ada peringkat
- Stochastic ProgrammingDokumen315 halamanStochastic Programmingfeiying1980100% (1)
- Codex Standard For Boiled Dried Salted Anchovies CODEX STAN 236-2003Dokumen6 halamanCodex Standard For Boiled Dried Salted Anchovies CODEX STAN 236-2003henryBelum ada peringkat
- Computer ProfessionalDokumen185 halamanComputer ProfessionalSHAHSYADBelum ada peringkat