Physics I Problems PDF
Diunggah oleh
bosschellen0%(1)0% menganggap dokumen ini bermanfaat (1 suara)
2K tayangan1 halamanCalculate the heat flow Q for each of the four processes ab, bc, ad, and dc. In each process, does the system absorb or liberate heat? how much?.
Deskripsi Asli:
Judul Asli
Physics I Problems (203).pdf
Hak Cipta
© © All Rights Reserved
Format Tersedia
PDF, TXT atau baca online dari Scribd
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniCalculate the heat flow Q for each of the four processes ab, bc, ad, and dc. In each process, does the system absorb or liberate heat? how much?.
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PDF, TXT atau baca online dari Scribd
0%(1)0% menganggap dokumen ini bermanfaat (1 suara)
2K tayangan1 halamanPhysics I Problems PDF
Diunggah oleh
bosschellenCalculate the heat flow Q for each of the four processes ab, bc, ad, and dc. In each process, does the system absorb or liberate heat? how much?.
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PDF, TXT atau baca online dari Scribd
Anda di halaman 1dari 1
648
CHAPTER 19 The First Law of Thermodynamics
19.41 .. When a system is Figure P19.41
taken from state a to state b in
p
Fig. P19.41 along the path acb,
c
b
90.0 J of heat ows into the system and 60.0 J of work is done
by the system. (a) How much
heat ows into the system along
path adb if the work done by the
a
d
system is 15.0 J? (b) When the
V
system is returned from b to a O
along the curved path, the
absolute value of the work done by the system is 35.0 J. Does the
system absorb or liberate heat? How much heat? (c) If Ua = 0 and
Ud = 8.0 J, nd the heat absorbed in the processes ad and db.
19.42 . A thermodynamic system is taken from state a to state Figure P19.42
c in Fig. P19.42 along either
p
path abc or path adc. Along path
c
b
abc, the work W done by the
system is 450 J. Along path adc,
W is 120 J. The internal energies
a
d
of each of the four states shown
in the gure are Ua = 150 J,
V
O
Ub = 240 J, Uc = 680 J, and
Ud = 330 J. Calculate the heat
ow Q for each of the four processes ab, bc, ad, and dc. In each
process, does the system absorb or liberate heat?
19.43 . A volume of air (assumed
to be an ideal gas) is rst cooled Figure P19.43
without changing its volume and
p (Pa)
then expanded without changing
a
5
its pressure, as shown by the path 3.0 3 10
abc in Fig. P19.43. (a) How does 2.0 3 105
the nal temperature of the gas
5
compare with its initial tempera- 1.0 3 10 b
c
ture? (b) How much heat does
V (m3)
O 0.02 0.04 0.06
the air exchange with its surroundings during the process
abc? Does the air absorb heat or release heat during this process?
Explain. (c) If the air instead expands from state a to state c by the
straight-line path shown, how much heat does it exchange with its
surroundings?
19.44 . Three moles of argon gas (assumed to be an ideal gas)
originally at a pressure of 1.50 * 10 4 Pa and a volume of
0.0280 m3 are rst heated and expanded at constant pressure to a
volume of 0.0435 m3, then heated at constant volume until the
pressure reaches 3.50 * 10 4 Pa, then cooled and compressed at
constant pressure until the volume is again 0.0280 m3, and nally
cooled at constant volume until the pressure drops to its original
value of 1.50 * 10 4 Pa. (a) Draw the pV-diagram for this cycle.
(b) Calculate the total work done by (or on) the gas during the
cycle. (c) Calculate the net heat exchanged with the surroundings.
Does the gas gain or lose heat overall?
19.45 .. Two moles of an ideal monatomic gas go through the
cycle abc. For the complete cycle, 800 J of heat ows out of the
gas. Process ab is at constant pressure, and process bc is at constant volume. States a and b have temperatures Ta = 200 K and
Tb = 300 K. (a) Sketch the pV-diagram for the cycle. (b) What is
the work W for the process ca?
19.46 .. Three moles of an ideal gas are taken around the cycle
acb shown in Fig. P19.46. For this gas, Cp = 29.1 J>mol # K.
Process ac is at constant pressure, process ba is at constant volume,
and process cb is adiabatic. The temperatures of the gas in states a,
c, and b are Ta = 300 K, Tc = 492 K, and Tb = 600 K. Calculate
the total work W for the cycle.
Figure P19.46
Figure P19.47
p 1atm2
p
b
2.0
c
0.50
V 1 L2
0.20
19.47 .. Figure P19.47 shows a pV-diagram for 0.0040 mole of
ideal H 2 gas. The temperature of the gas does not change during
segment bc. (a) What volume does this gas occupy at point c?
(b) Find the temperature of the gas at points a, b, and c. (c) How
much heat went into or out of the gas during segments ab, ca, and bc?
Indicate whether the heat has gone into or out of the gas. (d) Find
the change in the internal energy of this hydrogen during segments
ab, bc, and ca. Indicate whether the internal energy increased or
decreased during each of these segments.
19.48 .. The graph in Fig. Figure P19.48
P19.48 shows a pV-diagram
p 1 Pa 3 105 2
for 3.25 moles of ideal helium
(He) gas. Part ca of this
a
b
process is isothermal. (a) Find
the pressure of the He at point
a. (b) Find the temperature of
the He at points a, b, and c. (c)
2.0
c
How much heat entered or left
V 1 m3 2
the He during segments ab, bc,
O 0.010
0.040
and ca? In each segment, did
the heat enter or leave? (d) By how much did the internal energy of
the He change from a to b, from b to c, and from c to a? Indicate
whether this energy increased or decreased.
19.49 .. (a) One-third of a mole of He gas is taken along the path
abc shown as the solid line in Fig. P19.49. Assume that the gas
may be treated as ideal. How much heat is transferred into or out of
the gas? (b) If the gas instead went from state a to state c along the
horizontal dashed line in Fig. P19.49, how much heat would be
transferred into or out of the gas? (c) How does Q in part (b) compare with Q in part (a)? Explain.
Figure P19.49
p (Pa)
4 3 105
3 3 10
2 3 105
1 3 105
O
0.002 0.004 0.006 0.008 0.010
V (m3)
19.50 .. Two moles of helium are initially at a temperature of
27.0C and occupy a volume of 0.0300 m3. The helium rst
Anda mungkin juga menyukai
- Solution of Tut 2 PDFDokumen11 halamanSolution of Tut 2 PDFWesley0% (1)
- Bending StressDokumen19 halamanBending StressMohsin Islam RifatBelum ada peringkat
- Some Theorems On Right TrianglesDokumen4 halamanSome Theorems On Right TrianglesGlecy AdrianoBelum ada peringkat
- Assignment-3 - Engg Mech PDFDokumen6 halamanAssignment-3 - Engg Mech PDFNiteshBelum ada peringkat
- Newton's Law of CoolingDokumen2 halamanNewton's Law of CoolingChristian Elim SoliganBelum ada peringkat
- Final SolutionDokumen6 halamanFinal SolutionIR Ika EtyEtyka DoraBelum ada peringkat
- Moments MechanicsDokumen173 halamanMoments MechanicsChicha WatersBelum ada peringkat
- Problem Statement 12.16 - : Normal and Tangential Components of AccelerationDokumen4 halamanProblem Statement 12.16 - : Normal and Tangential Components of AccelerationErick MangalinoBelum ada peringkat
- Tutorial Ch1 ProblemDokumen5 halamanTutorial Ch1 ProblemKiat HauBelum ada peringkat
- Differential Calculus: The Respondent's Email Address (1503438@ub - Edu.ph) Was Recorded On Submission of This FormDokumen12 halamanDifferential Calculus: The Respondent's Email Address (1503438@ub - Edu.ph) Was Recorded On Submission of This FormJoseph LantoBelum ada peringkat
- Solns of 5 PointsDokumen2 halamanSolns of 5 PointsLorenz BerroyaBelum ada peringkat
- DynamicsDokumen3 halamanDynamicsjj012586Belum ada peringkat
- Solution 08Dokumen44 halamanSolution 08楊顗融Belum ada peringkat
- Normal and Shear Stress PDFDokumen25 halamanNormal and Shear Stress PDFmohanadymtBelum ada peringkat
- Set 1Dokumen2 halamanSet 1King LemuelBelum ada peringkat
- The Poisson DistributionDokumen13 halamanThe Poisson DistributionShehzad khanBelum ada peringkat
- EASC1081 Tutorial Week 13 SolutionDokumen5 halamanEASC1081 Tutorial Week 13 SolutionNita NabanitaBelum ada peringkat
- Vibration LectureDokumen49 halamanVibration LectureMark Oliver BernardoBelum ada peringkat
- Normal and Tangential ComponentsDokumen19 halamanNormal and Tangential ComponentsGarrey BrionesBelum ada peringkat
- Elasticity & Oscillations: Ut Tension, Sic Vis As Extension, So Force. Extension Is Directly Proportional To ForceDokumen11 halamanElasticity & Oscillations: Ut Tension, Sic Vis As Extension, So Force. Extension Is Directly Proportional To ForceJustin Paul VallinanBelum ada peringkat
- AnalyticDokumen8 halamanAnalyticMark Neil Delgaco GastiloBelum ada peringkat
- Statics AnDokumen13 halamanStatics AnIsaac Davy100% (1)
- CH 12Dokumen24 halamanCH 12Roberta Moraes MarcondesBelum ada peringkat
- Module 3 FUNDASURV 215 1Dokumen12 halamanModule 3 FUNDASURV 215 1rai100% (1)
- M4.1 WORK & ENERGY - Problems and SolutionDokumen6 halamanM4.1 WORK & ENERGY - Problems and SolutionsubyBelum ada peringkat
- Phet Lab Sim Forces & Motion BasicsDokumen5 halamanPhet Lab Sim Forces & Motion BasicsPeepBelum ada peringkat
- CH 04Dokumen43 halamanCH 04pwmvsiBelum ada peringkat
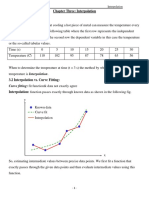
- Chapter Three: Interpolation: Curve Fitting: Fit Function& Data Not Exactly AgreeDokumen21 halamanChapter Three: Interpolation: Curve Fitting: Fit Function& Data Not Exactly AgreeMohsan HasanBelum ada peringkat
- CE 371 HW2 - SolutionDokumen6 halamanCE 371 HW2 - SolutionJoddediah MacasambatBelum ada peringkat
- Addition of Vectors - Practice ProblemDokumen2 halamanAddition of Vectors - Practice Problemstill jungkookBelum ada peringkat
- CIEM5390 Problem Set 2 - SolutionDokumen10 halamanCIEM5390 Problem Set 2 - SolutionMatthew LiBelum ada peringkat
- Chapter 16 Planar Kinematics of Rigid BodyDokumen47 halamanChapter 16 Planar Kinematics of Rigid Bodyrana abdul waasay0% (1)
- Mathalino: Ar AutomationDokumen4 halamanMathalino: Ar AutomationDeniell Joyce MarquezBelum ada peringkat
- ANS: (2.59807m/s2 Horizontal) (1.5m/s2 Vertical) (12.93725 Degree Angle That The Water Surface Makes With The Horizontal)Dokumen5 halamanANS: (2.59807m/s2 Horizontal) (1.5m/s2 Vertical) (12.93725 Degree Angle That The Water Surface Makes With The Horizontal)Lolly UmaliBelum ada peringkat
- Of The Bar by Equating The Strain: Problem 2.7-9Dokumen16 halamanOf The Bar by Equating The Strain: Problem 2.7-9EDiane LlanosBelum ada peringkat
- Hidraulik-Hydrostatic Pressure ApparatusDokumen16 halamanHidraulik-Hydrostatic Pressure ApparatusMohd AmranBelum ada peringkat
- 2020 Sept14 ME004ADokumen11 halaman2020 Sept14 ME004ADaniel ManivoughBelum ada peringkat
- Lesson 1-3Dokumen66 halamanLesson 1-3Andrich Rey CortesBelum ada peringkat
- Physics II ProblemsDokumen1 halamanPhysics II ProblemsBOSS BOSSBelum ada peringkat
- TOPIC 4 - Curve Fiiting and InterpolationDokumen23 halamanTOPIC 4 - Curve Fiiting and InterpolationBryan YuBelum ada peringkat
- Strema Act 2Dokumen42 halamanStrema Act 2James Ryu ZagalaBelum ada peringkat
- MorroofdDokumen2 halamanMorroofdAnonymous ym7PJCj0% (1)
- Mathematical&Usaefuldiscrete ExamplesDokumen2 halamanMathematical&Usaefuldiscrete ExamplesJenalyn QuemadaBelum ada peringkat
- Chapter 4 Equilibrium of A Rigid BodyDokumen12 halamanChapter 4 Equilibrium of A Rigid BodyWilliam JugalbotBelum ada peringkat
- Algebra2 - Exam - AnswerDokumen1 halamanAlgebra2 - Exam - AnswerRennel MallariBelum ada peringkat
- FIZ101E TotDokumen88 halamanFIZ101E TotdeniBelum ada peringkat
- MECH 333fluid Chapter3Dokumen22 halamanMECH 333fluid Chapter3Zion HelloBelum ada peringkat
- Engineering Mechanics Competency Exam Set B Students 20181Dokumen1 halamanEngineering Mechanics Competency Exam Set B Students 20181Justine Marowe AustriaBelum ada peringkat
- Bernoulli's Equation ProblemsDokumen9 halamanBernoulli's Equation ProblemsAboo Sdam AL MkahalBelum ada peringkat
- CE RefDokumen3 halamanCE Refcat buenafeBelum ada peringkat
- Surveying Lecture 100620Dokumen2 halamanSurveying Lecture 100620John Dalton ValenciaBelum ada peringkat
- Bugtong, Trixie Jael P.Dokumen10 halamanBugtong, Trixie Jael P.yeng botzBelum ada peringkat
- Chapter Three: Kinetics of ParticlesDokumen71 halamanChapter Three: Kinetics of Particlesyohannes lemiBelum ada peringkat
- Analytic GeometryDokumen5 halamanAnalytic Geometrylakr lamaBelum ada peringkat
- Exercice Thermodynamics PART IDokumen8 halamanExercice Thermodynamics PART Ichitl.23bi14075Belum ada peringkat
- ExercisesDokumen19 halamanExercisesNhật MinhBelum ada peringkat
- Physics Chapter 19 Class ProblemsDokumen3 halamanPhysics Chapter 19 Class ProblemsRuba AlBelum ada peringkat
- P Thermo DynamicsDokumen3 halamanP Thermo DynamicsJose CruzBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFBOSS BOSSBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I Problems PDFDokumen1 halamanPhysics I Problems PDFbosschellenBelum ada peringkat
- Physics I ProblemsDokumen1 halamanPhysics I ProblemsbosschellenBelum ada peringkat
- Chapter 2 - Environmental SamplingDokumen71 halamanChapter 2 - Environmental SamplingMuhammad Amirul Mat DzakiBelum ada peringkat
- Steam Pressure Curve of Saturated Steam (Marcet Boiler)Dokumen11 halamanSteam Pressure Curve of Saturated Steam (Marcet Boiler)muhammad aqmal100% (1)
- Rotomolded Tanks - Importance of Rotomolded TanksDokumen3 halamanRotomolded Tanks - Importance of Rotomolded Tanksrotomould03Belum ada peringkat
- A. Velocity B. Length C. Density D. Temperature E. AreaDokumen3 halamanA. Velocity B. Length C. Density D. Temperature E. AreaFitria Istikomah DewiBelum ada peringkat
- Exp 1Dokumen9 halamanExp 1Amirul Ramlan100% (1)
- Astronomy Chapter 1 NotesDokumen4 halamanAstronomy Chapter 1 NoteskrmooshBelum ada peringkat
- Conductance of SolutionDokumen6 halamanConductance of SolutionHatmylifeBelum ada peringkat
- ASEJ D 23 01681 ReviewerDokumen12 halamanASEJ D 23 01681 ReviewerKashaf ChBelum ada peringkat
- Kunal JRFDokumen3 halamanKunal JRFAakash KumarBelum ada peringkat
- Forrest E. Ames - An Introduction To Compressible FlowDokumen296 halamanForrest E. Ames - An Introduction To Compressible FlowyuryumaoBelum ada peringkat
- Assignment. III Year Mp-IIDokumen4 halamanAssignment. III Year Mp-IIshanthakumargcBelum ada peringkat
- F2 IS Exam1 (04-05)Dokumen9 halamanF2 IS Exam1 (04-05)羅天佑Belum ada peringkat
- FP-Lect10 - Water PropertiesDokumen34 halamanFP-Lect10 - Water PropertiesMohamed SadekBelum ada peringkat
- FiltrationDokumen70 halamanFiltrationEny Nurhayati100% (1)
- 2-Electrochemistry: Week 3 - Topic 2Dokumen65 halaman2-Electrochemistry: Week 3 - Topic 2ClarkBelum ada peringkat
- Household Services: Perform Mensuration and Calculation (MC)Dokumen41 halamanHousehold Services: Perform Mensuration and Calculation (MC)Sally Consumo Kong100% (4)
- Quick Reference Guide: Agilent 500-MS LC/MS SuppliesDokumen2 halamanQuick Reference Guide: Agilent 500-MS LC/MS SuppliesAGF DHJBelum ada peringkat
- Harmonically Excitation VibrationDokumen17 halamanHarmonically Excitation VibrationCherry ObiasBelum ada peringkat
- HeatecDokumen10 halamanHeatecMogtaba Osman100% (1)
- Effects of Ultrasound and Microwave Pretreatments of Carrot Slices Before Drying On The Color Indexes and Drying RateDokumen9 halamanEffects of Ultrasound and Microwave Pretreatments of Carrot Slices Before Drying On The Color Indexes and Drying RateAbhishek ChandraBelum ada peringkat
- Photocure TXR 10 TDSDokumen2 halamanPhotocure TXR 10 TDSjm sanchezBelum ada peringkat
- Py201 Assignment IIDokumen2 halamanPy201 Assignment IISiddharth RajamohananBelum ada peringkat
- exercise固態物理Dokumen31 halamanexercise固態物理Keiko AyanoBelum ada peringkat
- Chem 240 Lab Manual With Problems - 2013Dokumen177 halamanChem 240 Lab Manual With Problems - 2013Mark Cliffton BadlonBelum ada peringkat
- Solar Heat Worldwide 2021Dokumen86 halamanSolar Heat Worldwide 2021KorkmazAlpSakızlıoğluBelum ada peringkat
- Natural Sciences: Grade 8 Term 3 TrackerDokumen17 halamanNatural Sciences: Grade 8 Term 3 Trackerndodana SibandaBelum ada peringkat
- Context Clues: "Using Context Clues in The Sentences or Paragraph"Dokumen4 halamanContext Clues: "Using Context Clues in The Sentences or Paragraph"Rizki fitriana dewiBelum ada peringkat
- Thermodynamic 2019-2: Nedher Sánchez RamírezDokumen47 halamanThermodynamic 2019-2: Nedher Sánchez RamírezGenesis FloresBelum ada peringkat
- Release Coats Presentation PSTCDokumen26 halamanRelease Coats Presentation PSTCShawn ZhengBelum ada peringkat