Modern Management of Pyogenic Hepatic Abscess: A Case Series and Review of The Literature
Diunggah oleh
Astari Pratiwi NuhrintamaJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Modern Management of Pyogenic Hepatic Abscess: A Case Series and Review of The Literature
Diunggah oleh
Astari Pratiwi NuhrintamaHak Cipta:
Format Tersedia
Heneghan et al.
BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
RESEARCH ARTICLE
Open Access
Modern management of pyogenic hepatic
abscess: a case series and review of the literature
Helen M Heneghan1,2*, Nuala A Healy1,2, Sean T Martin1, Ronan S Ryan3, Niamh Nolan4, Oscar Traynor5 and
Ronan Waldron1
Abstract
Background: Pyogenic hepatic abscesses are relatively rare, though untreated are uniformly fatal. A recent
paradigm shift in the management of liver abscesses, facilitated by advances in diagnostic and interventional
radiology, has decreased mortality rates. The aim of this study was to review our experience in managing pyogenic
liver abscess, review the literature in this field, and propose guidelines to aid in the current management of this
complex disease.
Methods: Demographic and clinical details of all patients admitted to a single institution with liver abscess over a
5 year period were reviewed. Clinical presentation, aetiology, diagnostic work-up, treatment, morbidity and
mortality data were collated.
Results: Over a 5 year period 11 patients presented to a single institution with pyogenic hepatic abscess (55%
males, mean age 60.3 years). Common clinical features at presentation were non-specific constitutional symptoms
and signs. Aetiology was predominantly gallstones (45%) or diverticular disease (27%). In addition to empiric
antimicrobial therapy, all patients underwent radiologically guided percutaneous drainage of the liver abscess at
diagnosis and only 2 patients required surgical intervention, including one 16-year old female who underwent
hemi-hepatectomy for a complex and rare Actinomycotic abscess. There were no mortalities after minimum followup of one year.
Conclusions: Pyogenic liver abscesses are uncommon, and mortality has decreased over the last two decades.
Antimicrobial therapy and radiological intervention form the mainstay of modern treatment. Surgical intervention
should be considered for patients with large, complex, septated or multiple abscesses, underlying disease or in
whom percutaneous drainage has failed.
Background
Pyogenic abscesses account for almost 80% of all liver
abscesses in the developed world and are most often
polymicrobial [1]. Amoebic and fungal abscesses are less
common, and predominantly occur in Southeast Asia
and Africa. Though relatively rare, with a reported incidence of 0.5-0.8% in the Western world and a frequency
of 20 per 100,000 admissions in hospitalized patients,
pyogenic hepatic abscesses are potentially lethal [2].
Hence there is a need to recognize and treat this condition urgently. The aetiology of these abscesses has
changed over the last few decades. Historically, the
* Correspondence: helenheneghan@hotmail.com
1
Department of Surgery, Mayo General Hospital, Castlebar, Mayo, Ireland
Full list of author information is available at the end of the article
commonest cause had been acute appendicitis but, with
evolution and advancement of surgical practice and
microbiology over time, its frequency as the primary
source of abscess has decreased [3]. In contrast, the
increasing frequency of cholelithiasis and biliary tract
pathology, with their potential to incite ascending portal
sepsis, have replaced appendicitis as the leading cause of
hepatic abscess [2,4,5].
Liver abscess was previously regarded a high morbidity
disease requiring open surgical drainage, with mortality
rates between 9% and 80%. If untreated, it was uniformly fatal [4,6,7]. In the last quarter of a century we
have witnessed a major paradigm shift in the management of pyogenic hepatic abscesses, with a concomitant
decrease in mortality to 5-30% [8]. Advances in diagnostic and interventional radiology over the last three
2011 Heneghan et al; licensee BioMed Central Ltd. This is an open access article distributed under the terms of the Creative
Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
decades have facilitated a minimally invasive approach
to management of this condition. In combination with
targeted antimicrobial therapy, percutaneous drainage
techniques now form the mainstay of treatment. However, a small proportion of patients do not respond
appropriately to minimally invasive management strategies; it is critical to promptly recognize these patients,
for whom traditional open surgical intervention is the
definitive treatment. We have reviewed our experience
in managing pyogenic liver abscess over the last 5 years,
to illustrate the current etiology, management and outcomes of this disease. We also reviewed the literature in
this field, and present a summary of current practice
patterns which may serve as a useful guide for the modern management of pyogenic hepatic abscess.
Methods
Our institutions ethical committee granted ethical
approval for this study, which was then carried out in
compliance with the Helsinki Declaration. Demographic
and clinical details of all patients admitted to our unit
with liver abscess(es), between June 2004 and June 2009,
were prospectively entered onto a database. Data were
retrieved from a combination of chart review, theatre
records, Hospital In-Patient Enquiry (HIPE) and histopathology databases. A review of these data was performed to document the clinical presentation, aetiology,
diagnostic work-up, treatment, morbidity and mortality.
Radiologically-guided aspiration and drainage of
abscesses was performed under local anaesthesia, with
an 18-gauge needle and a range of different sized drainage catheters (8-12Fr) placed by the Seldinger technique. Pus aspirated at the time of initial abscess drainage
was routinely sent for microbial culture and sensitivity
analysis. Following completion of their treatment regimens, all patients were followed-up clinically and radiologically for at least 1 year to verify abscess resolution.
Results
Clinical presentation
Over a 5-year period, 11 patients presented to our unit
with pyogenic liver abscess. Mean age was 60.27 years
(range 16-85 years); there were 6 males and 5 females.
Underlying comorbidities were present in7 of 11
patients (63.6%) including diabetes (n = 2), hypertension
(n = 3), atrial fibrillation (n = 1), asthma requiring regular inhaled steroids (n = 1), and 2 patients had a history
of diverticular disease (two patient had more than one
comorbidity at presentation). No patient was taking
immunosuppressant medications. The commonest presenting features were vague non-specific symptoms
including fever (n = 7), right upper quadrant (RUQ)
pain (n = 5), jaundice (n = 3), anorexia and malaise (n =
3). RUQ tenderness and pyrexia were present in the
Page 2 of 8
majority of patients (n = 8). Biochemical and haematological findings are documented in Table 1. The most
consistent blood abnormality was an elevated C Reactive
Protein (CRP), which was observed in all cases.
Diagnostic work-up
All patients were initially evaluated with abdominal
ultrasonography (US). Following confirmation of an
abscess in the liver, computed tomography (CT) was
performed to further define its anatomic features, and to
try to determine its aetiology (Figure 1). The sensitivities
of US and CT were 100% in this series. Abscess location
was predominantly in the right lobe of the liver (72% of
patients). Ten patients had a solitary abscess; only one
patient in this series presented with multiple abscess
cavities. All patients proceeded to undergo US-guided
abscess drainage, either at the time of diagnosis or
within 24 hours. Characteristics of these hepatic
abscesses, with corresponding clinical data, are presented in Table 2.
Aetiology
Gallstones were present on abdominal US in 5 patients
(45.4%) although evidence of acute cholecystitis was
only one present in one of these cases. One patient who
presented with obstructive jaundice had evidence of
intra-hepatic biliary dilatation on US, secondary to compression of the hepatic ductal confluence in the porta
hepatitis by a left lobe abscess. CT scan identified diverticular disease in 3 additional cases (27.2%), without
overt diverticulitis. In the remaining 2 cases there was
no radiologically identifiable cause for liver abscess
(18.2%).
Microbiology
Microbial culture of pus drained from the hepatic
abscess was positive in 7 cases (64%) whilst blood cultures were positive in only 2 cases. Gram positive cocci
(Group B, Group C and Milleri Group Streptococci)
were the most commonly isolated organisms. Coliforms
were isolated in 3 cases (27%). Actinomyces was isolated
from the abscess of a 16 year old Irish girl who had no
history of immunocompromise, foreign travel, recent
abdominal trauma or surgery. PAS and Grocotts stains
Table 1 Biochemical and haematological derangements
in patients with liver abscess
Biochemical/haematological abnormality
% of cases (n)
C-Reactive Protein
100% (11)
White cell count
91% (10)
Liver function tests
82% (9)
Normochromic normocytic anaemia
64% (7)
Serum Creatinine
45% (5)
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
Page 3 of 8
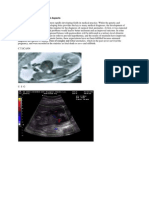
Figure 1 (a-d) CT images of four patients with hepatic abscesses.
demonstrated growth of the actinomyces organisms in
clusters of tangled filaments surrounded by neutrophils
(Figure 2).
Treatment
All patients were empirically commenced on broad
spectrum intravenous antibiotics at presentation. The
most commonly used empiric antimicrobial regimen
was a 3rd generation cephalosporin and metronidazole.
This was changed according to the sensitivities of organisms cultured from each patients abscess aspirate or
blood. Ciprofloxacin was used as an alternative to a
cephalosporin in one patient who had a history of penicillin anaphylaxis. All patients underwent radiologically
guided percutaneous drainage of the liver abscess at the
time of diagnosis or within 24 hours of presentation. In
10 cases (91%) a drain was placed in the abscess cavity
and left in-situ following initial aspiration. Duration of
drain placement ranged from 4 to 7 days. The timing of
catheter removal was guided by repeat imaging (US)
demonstrating a significant decrease in abscess size, or
diminution of drain output along with improvement in
clinical condition. One patient with a complex septated
abscess required placement of a second drain after 36
hours, as his clinical condition did not improve and
drain output was minimal. Four patients required transfer to the National Liver Unit as they failed to improve
despite maximal non-operative primary therapy. Two of
these patients (18% of all cases) underwent surgical
intervention. A 61 yr old male, with a 7 cm complex
septated abscess in the right lobe of his liver, underwent
right hemi-hepatectomy due to progressive deterioration
in his clinical condition despite percutaneous catheter
drainage, and a suspicion of malignancy on preoperative
imaging. The second operative case was that of a 16year-old Irish female who presented with a two-week
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
Page 4 of 8
Table 2 Characteristics of the liver abscesses
Clinical presentation
Characteristics of hepatic abscess
Management
details
Case Gender Age Predominant symptoms
Duration Site
(Lobe)
Number Size
(cm)
Likely etiology
Drain Surgery
Male
61
Fever, epigastric pain, anorexia/malaise
5 days
Right
Single
Portal vein
sepsis*
Yes
Yes
Male
72
Fever, rigors
11 days
Left
Single
Portal vein
sepsis*
Yes
No
Male
75
Fever, myalgia, pleuritic lower right chest
pain
10 days
Right
Single
Portal vein
sepsis*
Yes
No
Female
78
Fever, anorexia/malaise
2 weeks
Right
Single
10
Other^
Yes
No
5
6
Male
Female
85
56
Anorexia/malaise
RUQ pain, nausea
4 weeks
6 weeks
Right
Right
Single
Single
4
6
Other^
Biliary tract
sepsis
No
Yes
No
No
7
8
Female
Female
16
53
Fever, RUQ pain, jaundice
RUQ pain
5 days
5 days
Right
Left
Single
Single
12
7
Yes
Yes
Yes
No
Male
33
Fever, jaundice, dark urine
3 days
Right
Multiple
11
Yes
No
10
Male
64
RUQ pain, jaundice
7 days
Right
Single
Yes
No
11
Female
70
Fever, RUQ pain
5 days
Right
Single
10
Actinomycosis
Biliary tract
sepsis
Biliary tract
sepsis
Biliary tract
sepsis
Biliary tract
sepsis
Yes
No
*Portal vein sepsis; secondary to gastrointestinal sources of infection, including diverticulitis and appendicitis.
^Other causes include: hematogenous seeding from a distant site of infection, or unknown etiology.
Figure 2 Pathological images from two patients with hepatic abscess: (a-b) Demonstrates pathology of a 61yr old male with a 7 cm
abscess, of biliary aetiology, in the right lobe of his liver. (c-f) Gross and microscopic pathological mages from a 16-year-old Irish female who
presented with a 12 cm Actinomycotic abscess in right lobe of liver. (2a) H/E section (x 200 mag) of a pyogenic liver abscess showing
inflammatory cells. (2b) H/E section in higher magnification (x 400 mag) showing acute inflammatory cells forming an abscess. (2c) Section of
liver illustrating a well circumscribed abscess cavity (2d) H/E section (x 200 mag) of a pyogenic liver abscess showing a microabscess with a
central filamentous organism (Actinomycosis) (2e) PAS stain (x 400 mag) highlighting the actinomyces organisms (2f) Grocott stain (x 400 mag)
highlighting the actinomyces organisms (black).
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
history of RUQ pain, fever, and anorexia. At presentation she had profound systemic sepsis. Abdominal US
and CT demonstrated a large (12 cm) complex abscess
in right lobe of liver and despite broad spectrum intravenous antibiotics, percutaneous aspiration and drainage, her clinical condition deteriorated and she
underwent right hemi-hepatectomy and cholecystectomy
for a complex Actinomycotic abscess.
Outcome
There were no mortalities in this series. All patients
made a full recovery, and demonstrated evidence of
complete abscess resolution after a minimum of 1-year
follow up. The overall morbidity rate was 45% (n = 5).
Four patients developed severe sepsis with organ dysfunction over the course of their admission. These
patients all required intensive care and transfer to a specialized hepatobiliary unit for further management. The
young female with a primary Actinomycotic liver
abscess required a 6-month course of oral penicillin
post-operatively. One other patient developed a lower
limb deep venous thrombosis (DVT) despite prophylactic treatment with low molecular weight heparin. Overall the mean length of hospital stay for patients
managed non-operatively (n = 9) was 21 days. It was
considerably longer for those patients who underwent
surgical intervention (35 days, on average). All patients
were followed up for minimum of 12 months following
hospital discharge; repeat liver biochemistry and abdominal imaging (US or CT) confirmed abscess resolution in
all cases.
Discussion
Hepatic abscess was first described in the time of Hippocrates around 400B.C. In 1938, Ochsners, seminal
review of 47 cases of pyogenic liver abscesses heralded
open surgical drainage as the definitive therapy. This
series highlighted the uniformly fatal outcome of
untreated abscesses. Despite the recommended aggressive approach to treatment, mortality rates throughout
the mid twentieth century remained high at 60-80%.
Advances in diagnostic and therapeutic radiology,
coupled with improvements in microbiological identification and therapy, have recently decreased mortality
rates to <5-30% [2]. In our series of 11 patients, there
were no deaths and morbidity rate was 45%.
With the development of more sophisticated diagnostic techniques and longer lifespan, the demographic for
peak incidence of liver abscess is shifting towards the
sixth and seventh decades of life [9]. Liver abscesses are
rare in children and adolescents, and are usually associated with an underlying immune deficiency state or
trauma. Clinical presentation of pyogenic hepatic
abscess is non-specific and similar to that of other
Page 5 of 8
hepatobiliary inflammatory or infectious process. A
combination of nonspecific systemic symptoms is the
most common presentation; including fever and rigors,
nausea and vomiting, right upper quadrant pain, anorexia, weight loss, weakness and malaise. Less frequently
reported symptoms include cough or hiccups due to
diaphragmatic irritation, with referred pain to the right
shoulder. Patients with small solitary lesions usually
have a more insidious course with associated weight
loss, fatigue and anaemia of chronic disease. With such
symptoms, malignancy is often the initial concern. Multiple abscesses usually result in more acute presentations, as reflected by the one case in this series with
multiple abscesses who presented after a short 3 day illness, with symptoms and signs of systemic toxicity [3,9].
Regarding laboratory abnormalities associated with
hepatic abscesses, elevated inflammatory markers are
sensitive but non-specific. In our series, we observed
raised CRP in all cases. With regard to liver biochemistry, an elevated ALP is the single most common finding,
observed in up to 90% of cases. Approximately 50% of
patients demonstrate a general derangement of liver biochemistry, including elevated serum bilirubin, and hepatic enzymes AST and ALT. Other haematological
abnormalities include leucocytosis and normochromic
normocytic anaemia. Blood cultures are positive in less
than 50% of cases [9]; we observed positive blood cultures in only 18% of our case series. Definitive diagnosis
requires radiological evidence of an abscess in the liver
parenchyma. Ultrasonography and computed tomography of the abdomen are the gold standard diagnostic
modalities. In addition, they have therapeutic utility as
they are employed to guide percutaneous aspiration and
drainage of an abscess. These radiological investigations
may also reveal the underlying aetiology, for example a
contiguous infection within the biliary tree or peritoneal
cavity.
Although the incidence of pyogenic liver abscess has
remained relatively unchanged over time, the underlying
aetiology has changed in unison with developments in
surgical and microbiology practices. Biliary tract disease
has replaced appendicitis as the commonest cause of
bacterial liver abscess [4]. Chole(docho)lithiasis,
obstructing tumours, strictures, and congenital anomalies of the biliary tree, are common causative conditions.
An alternative mechanism for liver abscess formation,
apart from direct spread from contiguous infection, is
embolisation of a septic coagulum from an intra-abdominal infection. Diverticulitis, inflammatory bowel disease
and perforated hollow viscera are possible sources of
septic emboli in addition to appendicitis. Rarely, abscess
formation results from haematogenous dissemination of
organisms in association with a systemic bacteraemia, e.
g. from endocarditis or pyelonephritis. Such cases have
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
been reported in immunocompromised individuals with
chronic granulomatous disease and leukemia [5]. Our
series reflected this changing trend in aetiology; with a
biliary source in 45%, diverticular aetiology in 27.5%,
and likely haematogenous dissemination in the remaining 27.5% of patients. The organisms isolated from a
liver abscess are varied, usually polymicrobial, and
reflective of the infectious source (Table 3). Whilst this
study and review focused primarily on bacterial causes
of liver abscess, it should be noted that parasitic organisms are also common etiological factors, particularly in
tropical and subtropical climates. Entamoeba histolytica
and the nematode Ascaris lumbricoides are two such
parasites associated with liver abscess formation, and
should be considered in patients from endemic areas
[1,10,11].
Untreated, pyogenic hepatic abscess is uniformly fatal
due to ensuing complications such as sepsis, or peritonitis secondary to rupture of the abscess cavity into
the pleural or peritoneal cavities. Systemic antimicrobial therapy remains the mainstay primary treatment.
The choice of empiric antibiotics is based on the most
likely source of infection, but should be broad spectrum and administered parenterally. The regimen must
be reviewed and altered, if appropriate, to target specific organisms isolated from the abscess aspirate and/or
blood cultures. The recommended duration of parenteral antibiotic therapy is 2-3 weeks, or until there is a
favourable clinical response. Complementary oral antimicrobial therapy must then be continued for a further
2-4 weeks or until clinical, biochemical and radiological follow-up demonstrates complete resolution of the
abscess cavity. Evidence suggests that antibiotic therapy alone is usually not sufficient to entirely resolve a
liver abscess unless it is small (<3 cm) [12,13]. If antibiotics are selected as the sole therapeutic modality,
then a prolonged course of systemic and oral therapy
is warranted, and the choice of antimicrobial agent
must be guided by the sensitivities of causative
Table 3 Causative organisms of hepatic abscess
Source of infection
Common Organism
Biliary
Enteric gram negative organisms (enterococci)
Pelvic
Other intraperitoneal
source
Haematogenous
seeding
Bacteroides fragilis
Mixed aerobic/anaerobic organisms (e.g. B.
fragilis)
Single organism usually e.g. Staphylococcus,
Streptococcus (including Strep. milleri)
Immunocompromised Candida species
Other
Pyogenic: Klebsiella pneumoniae (Asia),
Actinomyces (rare).
Amebic: Entamoeba histolytica (Amebic
abscess),
Parasitic: Ascaris lumbricoides
Page 6 of 8
organisms [14,15]. There is debate however as to the
size of abscess above which antibiotic treatment alone
is unlikely to be successful. Malik et al recently
reported their experience of managing 169 pyogenic
liver abscesses, 16 of which were treated with intravenous antibiotics alone for 2 weeks [10]. This conservative approach was successful in only 6 of the 16
patients; the remaining 10 required open surgical drainage for definitive control of sepsis. The authors criteria for choosing antibiotics as the single therapeutic
modality were an uncomplicated abscess, <6 cm in
size. The authors low success rates with this approach
provide support for draining abscesses smaller than
6 cm, as recommended by Krige et al [1]. Indeed various reports suggest that any abscess larger than 3 cm
should be routinely drained [13,16,17]. In addition to
abscess size, other criteria for percutaneous drainage
include: continued pyrexia after 48-72 h of adequate
medical treatment, and clinical or ultrasonographic
features suggest impending perforation [10,18].
Surgical drainage is associated with high therapeutic
success rates, and was the standard of care until the
introduction of percutaneous drainage techniques in the
mid 1970s. With refinement of image-guided techniques
in recent years, percutaneous drainage and aspiration
have emerged as appropriate alternatives to open drainage, providing similarly high success rates but with the
advantages of a minimally invasive approach[19-21]. The
decision to leave a drainage catheter in the abscess cavity following aspiration, rather than performing repeated
needle aspirations, is also contentious. Those who prefer
multiple aspirations believe it to be as effective and safe
as catheter drainage, but simpler and quicker to perform, with decreased risk of procedural complications
and post-procedural sepsis [17,22,23]. However, this
approach requires careful follow-up, and often multiple
repeat imaging procedures to monitor response to therapy. Insertion of a drainage catheter has been shown to
be more effective than needle aspiration, particularly for
abscesses larger than 5 cm. Failure rates are higher, and
the average time to achieve a 50% reduction in size of
an abscess cavity has been shown to be significantly
longer, with needle aspiration compared to abscesses in
which a drainage catheter has been placed [17,22].
Despite the reported success of percutaneous drainage
techniques, there remains a role for open surgical intervention in the management of pyogenic abscess. Indications for surgical intervention include:
No clinical response after 4-7 days of drainage via
a catheter placed in the abscess cavity.
Multiple, large, or loculated abscess
Thick walled abscess with viscous pus
Concurrent intra-abdominal surgical pathology.
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
Bertel et al [24] compared open surgery with percutaneous drainage for the management of pyogenic liver
abscesses greater than 5 cm. Whilst morbidity was comparable for both approaches, those who underwent open
surgical resection/drainage required fewer secondary
procedures, and achieved higher rates of abscess resolution. Very recently, it has been shown that using multidetector CT to analyze abscesses could identify factors
predictive of percutaneous drainage failure [25]. The
strongest predictor of failure included the presence of
gas in the abscess. Other notable factors were an abscess
size greater than 7.3 cm, and a short length to the liver
capsule of <0.25 cm. In the setting of failed percutaneous drainage of a liver abscess, another consideration
prior to proceeding with open surgery is laparoscopic
drainage. Aydin et al systematically reviewed the literature describing experience with this approach, and identified 53 cases of liver abscess treated by laparoscopy.
The overall success rate was 90.5%, and no reported
case required conversion to open [26]. From their
review of this data, the authors concluded that laparoscopic surgery has the advantages of high success rates
of open surgery, and the minimal invasiveness of percutaneous drainage. In light of this, it should be considered as an alternative before open drainage, when other
modalities have failed.
Actinomycotic liver abscess
Primary actinomycotic liver abscess is extremely rare. In
this series, we include the first report to our knowledge
of this type of liver abscess in Ireland and the United
Kingdom. Actinomycosis is a chronic suppurative granulomatous disease characterized by abscess formation, tissue fibrosis and discharging sinuses. The actinomyces
organism is a gram-positive, semi-facultative, pleomorphic non-spore-forming, non-acid-fast bacillus of
the genus Actinomyces. The most common presentation
of this disease is infection of the oral and cervicofacial
region. However, the thoracic region, abdomino-pelvic
region and CNS are also frequently involved. Although
actinomyces species are part of the normal oral and gut
flora, (where they have low pathogenicity) any disruption of the gastrointestinal mucosal barrier can lead to
disease. Once the organism breaches mucosal barriers
and is established locally, it is notoriously pathogenic
and spread of infection has no regard for anatomic
planes or boundaries. The few previously described
cases of actinomycotic GI infection have occurred where
there has been a loss of mucosal integrity, as occurs
with surgery, appendicitis, diverticulitis, trauma, or foreign bodies including intrauterine contraceptive devices
[27,28]. Possible causes of hepatic infection by actinomyces include disseminated infection from oral disease
via the hepatic artery, from intra-abdominal disease via
Page 7 of 8
the portal vein, or from direct extension of a subdiaphragmatic or subhepatic process. However, in the majority of cases no cause is identifiable, as in our case. The
16 year old female in this series had no history of foreign travel, infectious contacts, past medical or surgical
illness, abdominal trauma or foreign body in-situ. In
addition, her dental hygiene was good, and she had no
evidence of oral or cervical infection. Culture of the
organism is difficult; it requires anaerobic or microaerophillic conditions, gram stain of the organism, haematoxylin-eosin stain of the sulfur granules, or more
specific stains such as Grocott-Gomori methenaminesilver nitrate stain and P-aminosalicylic acid (Figure 2e
&2f). Management of an actinomycotic liver abscess
should follow the general principles described above,
primarily with antimicrobial agents and abscess drainage. Actinomyces is sensitive to penicillin; the recommended course for hepatic actinomycosis is parenteral
penicillin for 2-6 weeks, followed by oral therapy with
penicillin or amoxicillin for 6-12 months [29]. Drainage
of these liver abscesses is guided by their size, complexity and the clinical condition of the patient. Our patient
required transfer to a specialized liver unit for surgical
management because of clinical deterioration despite
maximal non-operative therapy. Following hemi-hepatectomy and the recommended course of penicillin
based systemic therapy she made an excellent recovery
and remains well after 36 months.
Conclusion
Although our series of hepatic abscess cases is small,
there are few reports in recent years of outcomes with
modern management techniques and thus our experience may provide valuable information to clinicians who
encounter this uncommon condition. In particular, the
rare case of an actinomycotic liver abscess in a healthy
Irish female is interesting and noteworthy. Based on our
review of existing literature and our experience to date,
we propose the following summary to guide the management of pyogenic hepatic abscess:
1. Prompt administration of empiric broad spectrum
parenteral antibiotics
2. Ultrasound and/or CT scan to confirm diagnosis,
with simultaneous radiologically guided aspiration of
all abscesses >3 cm, +/- drain placement.
3. Microbiological analysis of abscess aspirates and
blood cultures: antibiotic regimen should be adjusted
according to culture results and sensitivities.
4. Early recognition of septicaemia or organ failure
and appropriate transfer to critical care unit. Consider repeat imaging to confirm correct drain placement and to determine response to treatment and
final resolution of the abscess.
Heneghan et al. BMC Research Notes 2011, 4:80
http://www.biomedcentral.com/1756-0500/4/80
5. Surgical intervention should be considered for
patients with large, complex, septated or multiple
abscesses, underlying disease or in whom percutaneous drainage has failed.
Consent
Written consent for publication was obtained from all
patients whose clinical images are used in this article.
Author details
1
Department of Surgery, Mayo General Hospital, Castlebar, Mayo, Ireland.
2
Department of Surgery, National University of Ireland, Galway, Ireland.
3
Department of Radiology, Mayo General Hospital, Castlebar, Mayo, Ireland.
4
Department of Histopathology, St.Vincents University Hospital, Dublin,
Ireland. 5Liver Unit, St. Vincents University Hospital, Dublin, Ireland.
Authors contributions
HMH was involved in conception and design of the study, literature review,
acquisition of data, analysis and interpretation of the data, and drafted the
manuscript. STM was involved in data acquisition, drafting and editing the
manuscript. NH contributed to data acquisition, literature review and
manuscript preparation. RR was the radiologist involved in diagnostic
imaging and radiologically guided management of all cases. NN was the
pathologist involved in the surgical cases, made the rare diagnosis of
Actinomycosis, acquired images for Figure 2, and edited the manuscript. OT
and RW directed management of the patients clinically, performed the
surgeries, supplied relevant clinical information about the patients and
revised the manuscript for important intellectual content. All authors read
and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Received: 17 November 2010 Accepted: 24 March 2011
Published: 24 March 2011
References
1. Krige JE, Beckingham IJ: ABC of diseases of liver, pancreas, and biliary
system. BMJ 2001, 322(7285):537-540.
2. Huang CJ, Pitt HA, Lipsett PA, Osterman FA Jr, Lillemoe KD, Cameron JL,
Zuidema GD: Pyogenic hepatic abscess. Changing trends over 42 years.
Ann Surg 1996, 223(5):600-607, discussion 607-609.
3. Ochsner A, DeBakey M, Murray S: Pyogenic Abscess of the Liver II. An
Analysis of Forty-Seven Cases with Review of the Literature. Am J Surg
1938, XL:292-319.
4. Branum GD, Tyson GS, Branum MA, Meyers WC: Hepatic abscess. Changes
in etiology, diagnosis, and management. Ann Surg 1990, 212(6):655-662.
5. Rahimian J, Wilson T, Oram V, Holzman RS: Pyogenic liver abscess: recent
trends in etiology and mortality. Clin Infect Dis 2004, 39(11):1654-1659.
6. Farges O, Leese T, Bismuth H: Pyogenic liver abscess: an improvement in
prognosis. Br J Surg 1988, 75(9):862-865.
7. Pitt HA, Zuidema GD: Factors influencing mortality in the treatment of
pyogenic hepatic abscess. Surg Gynecol Obstet 1975, 140(2):228-234.
8. Chen SC, Huang CC, Tsai SJ, Yen CH, Lin DB, Wang PH, Chen CC, Lee MC:
Severity of disease as main predictor for mortality in patients with
pyogenic liver abscess. Am J Surg 2009, 198(2):164-172.
9. Chou FF, Sheen-Chen SM, Chen YS, Chen MC: Single and multiple
pyogenic liver abscesses: clinical course, etiology, and results of
treatment. World J Surg 1997, 21(4):384-388, discussion 388-389.
10. Malik AA, Bari SU, Rouf KA, Wani KA: Pyogenic liver abscess: Changing
patterns in approach. World J Gastrointest Surg 2010, 2(12):395-401.
11. Bari S, Sheikh KA, Ashraf M, Hussain Z, Hamid A, Mufti GN: Ascaris liver
abscess in children. J Gastroenterol 2007, 42(3):236-240.
12. Chung YF, Tan YM, Lui HF, Tay KH, Lo RH, Kurup A, Tan BH: Management
of pyogenic liver abscesses - percutaneous or open drainage? Singapore
Med J 2007, 48(12):1158-1165, quiz 1165.
Page 8 of 8
13. Hope WW, Vrochides DV, Newcomb WL, Mayo-Smith WW, Iannitti DA:
Optimal treatment of hepatic abscess. Am Surg 2008, 74(2):178-182.
14. Rintoul R, ORiordain MG, Laurenson IF, Crosbie JL, Allan PL, Garden OJ:
Changing management of pyogenic liver abscess. Br J Surg 1996,
83(9):1215-1218.
15. Nazir NT, Penfield JD, Hajjar V: Pyogenic liver abscess. Cleve Clin J Med
2010, 77(7):426-427.
16. Hui JY, Yang MK, Cho DH, Li A, Loke TK, Chan JC, Woo PC: Pyogenic liver
abscesses caused by Klebsiella pneumoniae: US appearance and
aspiration findings. Radiology 2007, 242(3):769-776.
17. Rajak CL, Gupta S, Jain S, Chawla Y, Gulati M, Suri S: Percutaneous
treatment of liver abscesses: needle aspiration versus catheter drainage.
AJR Am J Roentgenol 1998, 170(4):1035-1039.
18. Porras-Ramirez G, Hernandez-Herrera MH, Porras-Hernandez JD: Amebic
hepatic abscess in children. J Pediatr Surg 1995, 30(5):662-664.
19. Berger LA, Osborne DR: Treatment of pyogenic liver abscesses by
percutaneous needle aspiration. Lancet 1982, 1(8264):132-134.
20. Pearce N, Knight R, Irving H, Menon K, Prasad K, Pollard S, Lodge J,
Toogood G: Non-operative management of pyogenic liver abscess. HPB
(Oxford) 2003, 5(2):91-95.
21. OFarrell N, Collins CG, McEntee GP: Pyogenic liver abscesses: Diminished
role for operative treatment. Surgeon 2010, 8(4):192-196.
22. Zerem E, Hadzic A: Sonographically guided percutaneous catheter
drainage versus needle aspiration in the management of pyogenic liver
abscess. AJR Am J Roentgenol 2007, 189(3):W138-142.
23. Giorgio A, Tarantino L, Mariniello N, Francica G, Scala E, Amoroso P,
Nuzzo A, Rizzatto G: Pyogenic liver abscesses: 13 years of experience in
percutaneous needle aspiration with US guidance. Radiology 1995,
195(1):122-124.
24. Bertel CK, van Heerden JA, Sheedy PF: Treatment of pyogenic hepatic
abscesses. Surgical vs percutaneous drainage. Arch Surg 1986,
121(5):554-558.
25. Liao WI, Tsai SH, Yu CY, Huang GS, Lin YY, Hsu CW, Hsu HH, Chang WC:
Pyogenic liver abscess treated by percutaneous catheter drainage:
MDCT measurement for treatment outcome. Eur J Radiol 2011.
26. Aydin C, Piskin T, Sumer F, Barut B, Kayaalp C: Laparoscopic drainage of
pyogenic liver abscess. JSLS 2010, 14(3):418-420.
27. Cintron JR, Del Pino A, Duarte B, Wood D: Abdominal actinomycosis. Dis
Colon Rectum 1996, 39(1):105-108.
28. Perlow JH, Wigton T, Yordan EL, Graham J, Wool N, Wilbanks GD:
Disseminated pelvic actinomycosis presenting as metastatic carcinoma:
association with the progestasert intrauterine device. Rev Infect Dis 1991,
13(6):1115-1119.
29. Russo T, Mandell G, Bennett J, Dolin R: Agents of actinomycosis. Mandell,
Douglas and Benetts Principles and Practice of Infectious Diseases New York:
Churchill Livingstone , 4 1995, 2280-2287.
doi:10.1186/1756-0500-4-80
Cite this article as: Heneghan et al.: Modern management of pyogenic
hepatic abscess: a case series and review of the literature. BMC Research
Notes 2011 4:80.
Submit your next manuscript to BioMed Central
and take full advantage of:
Convenient online submission
Thorough peer review
No space constraints or color figure charges
Immediate publication on acceptance
Inclusion in PubMed, CAS, Scopus and Google Scholar
Research which is freely available for redistribution
Submit your manuscript at
www.biomedcentral.com/submit
Anda mungkin juga menyukai
- Microbes Predict Outcomes Complicated Lung InfectionsDokumen9 halamanMicrobes Predict Outcomes Complicated Lung InfectionsabhikanjeBelum ada peringkat
- Anesthesiaforemergency Abdominalsurgery: Carol Peden,, Michael J. ScottDokumen13 halamanAnesthesiaforemergency Abdominalsurgery: Carol Peden,, Michael J. ScottJEFFERSON MUÑOZBelum ada peringkat
- CasAre There Risk Factors For Splenic Rupture During Colonoscopy CaseDokumen6 halamanCasAre There Risk Factors For Splenic Rupture During Colonoscopy Caseysh_girlBelum ada peringkat
- Etiological Pattern and Outcome of Non-Variceal Upper G.I. BleedingDokumen5 halamanEtiological Pattern and Outcome of Non-Variceal Upper G.I. BleedingJawad ZafarBelum ada peringkat
- Management of Liver Abscess in Children: Our Experience: 10.5005/jp-Journals-10018-1206Dokumen4 halamanManagement of Liver Abscess in Children: Our Experience: 10.5005/jp-Journals-10018-1206Ivan VeriswanBelum ada peringkat
- Annsurg00166 0011Dokumen8 halamanAnnsurg00166 0011Muhammad FadillahBelum ada peringkat
- Acute Cholecystitis: From ER To Surgery: Riginal RticleDokumen5 halamanAcute Cholecystitis: From ER To Surgery: Riginal RticleKrystel Joy AuroBelum ada peringkat
- 260-Article Text-875-1-10-20220728Dokumen4 halaman260-Article Text-875-1-10-20220728hasan andrianBelum ada peringkat
- General Surgery: Ruptured Liver Abscess: A Novel Surgical TechniqueDokumen3 halamanGeneral Surgery: Ruptured Liver Abscess: A Novel Surgical TechniqueRahul SinghBelum ada peringkat
- Infections After Laparoscopic and Open Cholecystectomy: Ceftriaxone Versus Placebo A Double Blind Randomized Clinical TrialDokumen6 halamanInfections After Laparoscopic and Open Cholecystectomy: Ceftriaxone Versus Placebo A Double Blind Randomized Clinical TrialvivianmtBelum ada peringkat
- Tatalaksana Abses HeparDokumen6 halamanTatalaksana Abses HeparAdhi Rizky PutraBelum ada peringkat
- 1749-7922-6-37 - SalinDokumen5 halaman1749-7922-6-37 - SalinAnonymous InJS6aYZBelum ada peringkat
- Systematized Management of Postoperative Enterocutaneous Fistulas. A 14 Years ExperienceDokumen5 halamanSystematized Management of Postoperative Enterocutaneous Fistulas. A 14 Years ExperienceYacine Tarik AizelBelum ada peringkat
- HPB 09 S2 071Dokumen120 halamanHPB 09 S2 071Widya Dahlia JuwitaBelum ada peringkat
- Histopathologic Features and Clinical Outcomes in 71 Cases of Bladder DiverticulaDokumen6 halamanHistopathologic Features and Clinical Outcomes in 71 Cases of Bladder DiverticulaIrma Suriani DarwisBelum ada peringkat
- Gastro 1004Dokumen4 halamanGastro 1004Paul HartingBelum ada peringkat
- Mortality in Typhoid Intestinal Perforation-A Declining TrendDokumen3 halamanMortality in Typhoid Intestinal Perforation-A Declining Trendaura009Belum ada peringkat
- Epidemiology and Prognostic Factors of Liver Abscess Complications in Northeastern MexicoDokumen6 halamanEpidemiology and Prognostic Factors of Liver Abscess Complications in Northeastern MexicoLeonhardt RowikaBelum ada peringkat
- 2014 Article 639Dokumen8 halaman2014 Article 639Anida HasnaBelum ada peringkat
- Research Article: Massive Pleural Fluid Collection in Adult NigeriansDokumen5 halamanResearch Article: Massive Pleural Fluid Collection in Adult NigeriansHotiemah Aina ZhakiyahBelum ada peringkat
- Abbas Et Al, Angka Kejadian PLADokumen4 halamanAbbas Et Al, Angka Kejadian PLAhasan andrianBelum ada peringkat
- Acute Pyelonephritis in Adults: A Case Series of 223 PatientsDokumen6 halamanAcute Pyelonephritis in Adults: A Case Series of 223 PatientsshiaBelum ada peringkat
- Abcesele FicatuluiDokumen4 halamanAbcesele FicatuluiMihaela AlinaBelum ada peringkat
- Liver Abscess DissertationDokumen4 halamanLiver Abscess DissertationPayForAPaperAtlanta100% (1)
- Complications of Pancreatic SurgeryDokumen5 halamanComplications of Pancreatic Surgeryhenok seifeBelum ada peringkat
- Organ-Specific Infections: Types, Causes, TreatmentsDokumen4 halamanOrgan-Specific Infections: Types, Causes, TreatmentsMin dela FuenteBelum ada peringkat
- Cholangitis: Causes, Diagnosis, and ManagementDokumen10 halamanCholangitis: Causes, Diagnosis, and ManagementDianaBelum ada peringkat
- Bleeding peptic ulcer epidemiology and prognosisDokumen8 halamanBleeding peptic ulcer epidemiology and prognosisJain KasparBelum ada peringkat
- Upper Gastrointestinal Bleeding Ugb Causes and Treatment 2475 3181 1000153Dokumen3 halamanUpper Gastrointestinal Bleeding Ugb Causes and Treatment 2475 3181 1000153Envhy AmaliaBelum ada peringkat
- Variation in The Use of Laparoscopic Cholecystectomy For Acute CholecystitisDokumen5 halamanVariation in The Use of Laparoscopic Cholecystectomy For Acute CholecystitisAziz EzoBelum ada peringkat
- Abdominal Tuberculosis in Nepal Medical College Teaching Hospital, KathmanduDokumen4 halamanAbdominal Tuberculosis in Nepal Medical College Teaching Hospital, KathmanduSavitri Maharani BudimanBelum ada peringkat
- A Clinicohaematological Profile of Splenomegaly: Varsha S. Dabadghao, Arundhati G.Diwan, Amol M. RaskarDokumen9 halamanA Clinicohaematological Profile of Splenomegaly: Varsha S. Dabadghao, Arundhati G.Diwan, Amol M. RaskarLianSiahaanBelum ada peringkat
- Maat 12 I 3 P 271 DDDDDokumen5 halamanMaat 12 I 3 P 271 DDDDKacamata BagusBelum ada peringkat
- Management of Infected Pancreatic Necrosis 65Dokumen4 halamanManagement of Infected Pancreatic Necrosis 65zahir_jasBelum ada peringkat
- Hepatobiliary and pancreatic disorders overviewDokumen2 halamanHepatobiliary and pancreatic disorders overviewiweBelum ada peringkat
- File 1Dokumen4 halamanFile 1Marsya Yulinesia LoppiesBelum ada peringkat
- Debiri 1Dokumen12 halamanDebiri 1paquidermo85Belum ada peringkat
- Van Nella 2010Dokumen9 halamanVan Nella 2010Anonymous tSdibBveBelum ada peringkat
- Incidental Gallbladder Cancer. Residual Cancer Discovered at Oncologic Extended Resection Determines Outcome. A Report From High - and Low-IncidencDokumen10 halamanIncidental Gallbladder Cancer. Residual Cancer Discovered at Oncologic Extended Resection Determines Outcome. A Report From High - and Low-IncidencCarlos SotoBelum ada peringkat
- Worldwide Trends in Surgical Techniques in The Treatment of Esophageal and Gastroesophageal Junction CancerDokumen7 halamanWorldwide Trends in Surgical Techniques in The Treatment of Esophageal and Gastroesophageal Junction CancerLuca BellaioBelum ada peringkat
- Acute CholecystitisDokumen5 halamanAcute CholecystitisAdrian BăloiBelum ada peringkat
- Liver A BSC DrainDokumen8 halamanLiver A BSC DrainAngelica AmesquitaBelum ada peringkat
- Adler 2015Dokumen7 halamanAdler 2015nathaliepichardoBelum ada peringkat
- Literature Review of Liver AbscessDokumen4 halamanLiterature Review of Liver Abscessc5ng67v1100% (1)
- Medi 98 E14234Dokumen5 halamanMedi 98 E14234فرجني موغBelum ada peringkat
- Cholelithiasis: Causative Factors, Clinical Manifestations and ManagementDokumen6 halamanCholelithiasis: Causative Factors, Clinical Manifestations and ManagementmusdalifahBelum ada peringkat
- Palliative Management of Malignant Rectosigmoidal Obstruction. Colostomy vs. Endoscopic Stenting. A Randomized Prospective TrialDokumen4 halamanPalliative Management of Malignant Rectosigmoidal Obstruction. Colostomy vs. Endoscopic Stenting. A Randomized Prospective TrialMadokaDeigoBelum ada peringkat
- PrognosisDokumen10 halamanPrognosisAnida HasnaBelum ada peringkat
- Clinical Profile, Diagnosis and Therapeutic Management of Pyogenic Liver AbscessDokumen11 halamanClinical Profile, Diagnosis and Therapeutic Management of Pyogenic Liver AbscessIJAR JOURNALBelum ada peringkat
- Lamp IranDokumen5 halamanLamp IranTanwilyBelum ada peringkat
- Colitis Induced ChemotherapyDokumen11 halamanColitis Induced Chemotherapyhandris yanitraBelum ada peringkat
- Abdominal Sepsis Prognostic FactorDokumen6 halamanAbdominal Sepsis Prognostic FactorPrasojo JojoBelum ada peringkat
- Ca SekumDokumen7 halamanCa SekumNely M. RosyidiBelum ada peringkat
- Neutropenic Colitis in Children 2011Dokumen4 halamanNeutropenic Colitis in Children 2011IRINA SULEY TIRADO PEREZBelum ada peringkat
- Colorectal Cancer in Young Patients A Case ReportDokumen4 halamanColorectal Cancer in Young Patients A Case ReportPaula Carter CastelloBelum ada peringkat
- Acute Allograft Rejection in Liver Transplant Recipients: Incidence, Risk Factors, Treatment Success, and Impact On Graft FailureDokumen12 halamanAcute Allograft Rejection in Liver Transplant Recipients: Incidence, Risk Factors, Treatment Success, and Impact On Graft Failurenathalialusman98Belum ada peringkat
- Absceso Hepatico PiogenoDokumen6 halamanAbsceso Hepatico PiogenoPablo Eduardo AmadorBelum ada peringkat
- Critical Care for Potential Liver Transplant CandidatesDari EverandCritical Care for Potential Liver Transplant CandidatesDmitri BezinoverBelum ada peringkat
- The Evolving Landscape of Liver Cirrhosis ManagementDari EverandThe Evolving Landscape of Liver Cirrhosis ManagementHitoshi YoshijiBelum ada peringkat
- Q1 - Caillier (2014) - PSM, Goal Clarity, O Commit, ExtraRoleDokumen20 halamanQ1 - Caillier (2014) - PSM, Goal Clarity, O Commit, ExtraRoleAstari Pratiwi NuhrintamaBelum ada peringkat
- 1 - Big Five Personality, OCB PredictorDokumen11 halaman1 - Big Five Personality, OCB PredictorAstari Pratiwi NuhrintamaBelum ada peringkat
- Andrew Dan Cazarez (2015) - PSM, OCB, O PerformanceDokumen23 halamanAndrew Dan Cazarez (2015) - PSM, OCB, O PerformanceAstari Pratiwi NuhrintamaBelum ada peringkat
- NIST-Forensics Science ActivityDokumen24 halamanNIST-Forensics Science ActivityAstari Pratiwi NuhrintamaBelum ada peringkat
- Biografi Kaoru IshikawaDokumen16 halamanBiografi Kaoru IshikawaAstari Pratiwi Nuhrintama100% (1)
- 5357 11760 1 PBDokumen9 halaman5357 11760 1 PBYoshiee X-friend SurabayaBelum ada peringkat
- 18 Management Building BlocksDokumen10 halaman18 Management Building BlocksAstari Pratiwi NuhrintamaBelum ada peringkat
- Vermont Forensic Laboratory Physical Comparison UnitDokumen52 halamanVermont Forensic Laboratory Physical Comparison UnitAstari Pratiwi NuhrintamaBelum ada peringkat
- Diabetes Mellitus and Diabetic Eye Disease: DR Russell J WatkinsDokumen62 halamanDiabetes Mellitus and Diabetic Eye Disease: DR Russell J WatkinsAstari Pratiwi NuhrintamaBelum ada peringkat
- Automated Clarity and Quality Assessment For Latent FingerprintsDokumen6 halamanAutomated Clarity and Quality Assessment For Latent FingerprintsAstari Pratiwi NuhrintamaBelum ada peringkat
- An Evaluation of The Techniques Used To Collect Latent Prints From DocumentsDokumen68 halamanAn Evaluation of The Techniques Used To Collect Latent Prints From DocumentsAstari Pratiwi NuhrintamaBelum ada peringkat
- EmpyemaDokumen3 halamanEmpyemaAstari Pratiwi NuhrintamaBelum ada peringkat
- On Matching Latent FingerprintDokumen8 halamanOn Matching Latent FingerprintAstari Pratiwi NuhrintamaBelum ada peringkat
- Department of Forensic Medicine, University of Dundee Lecture NotesDokumen5 halamanDepartment of Forensic Medicine, University of Dundee Lecture NotesWisnu AdryantoBelum ada peringkat
- Latent Print Develipment Chap 7Dokumen68 halamanLatent Print Develipment Chap 7kuala1982Belum ada peringkat
- Antenatal Diagnosis - Surgical AspectsDokumen3 halamanAntenatal Diagnosis - Surgical AspectsAstari Pratiwi NuhrintamaBelum ada peringkat
- Liver AbscessDokumen1 halamanLiver AbscessAstari Pratiwi Nuhrintama0% (1)
- Diabetic Thy Studies by FSDokumen10 halamanDiabetic Thy Studies by FSAnitha BalakrishnanBelum ada peringkat
- English: The MembersDokumen5 halamanEnglish: The MembersAstari Pratiwi NuhrintamaBelum ada peringkat
- Puerperium Normal Abnormal LW Feb 2011Dokumen16 halamanPuerperium Normal Abnormal LW Feb 2011Astari Pratiwi NuhrintamaBelum ada peringkat
- Jadwal Jaga Koas Rskdia Sitti Fatimah Periode 28 Desember 2014 - 3 Januari 2015Dokumen5 halamanJadwal Jaga Koas Rskdia Sitti Fatimah Periode 28 Desember 2014 - 3 Januari 2015Astari Pratiwi NuhrintamaBelum ada peringkat
- CerpenDokumen13 halamanCerpenAstari Pratiwi NuhrintamaBelum ada peringkat
- TransfusiDokumen10 halamanTransfusiAstari Pratiwi NuhrintamaBelum ada peringkat
- eDokumen119 halamaneAstari Pratiwi NuhrintamaBelum ada peringkat
- Esophageal AhalasiaDokumen6 halamanEsophageal AhalasiaSitti RahmadaniBelum ada peringkat
- Diaphragmatic HerniaDokumen11 halamanDiaphragmatic HerniaAstari Pratiwi NuhrintamaBelum ada peringkat
- Daftar Peserta Olimpiade Sains Nasional (OSN) 2008 Bidang Studi KomputerDokumen3 halamanDaftar Peserta Olimpiade Sains Nasional (OSN) 2008 Bidang Studi KomputerAstari Pratiwi NuhrintamaBelum ada peringkat
- (Psychology) Personal Depression Therapy (By James Herndon, MD)Dokumen62 halaman(Psychology) Personal Depression Therapy (By James Herndon, MD)Wiwie Dwi FitriBelum ada peringkat
- The Secret of A Perfect MurderDokumen7 halamanThe Secret of A Perfect MurderAstari Pratiwi NuhrintamaBelum ada peringkat
- Smoking Cessation: Prepared by Dr. Ahmed El MasryDokumen77 halamanSmoking Cessation: Prepared by Dr. Ahmed El Masryاحمد المصرىBelum ada peringkat
- DR, DR Luh Made Mas Rusyati SPKK (K) Finsdv Dermatology and Venereology Department Medical Faculty Udayana University/ Sanglah HospitalDokumen41 halamanDR, DR Luh Made Mas Rusyati SPKK (K) Finsdv Dermatology and Venereology Department Medical Faculty Udayana University/ Sanglah HospitalJohan CahyadirgaBelum ada peringkat
- Heart Block - Types, Diagnosis, Treatment, Follow-UpDokumen14 halamanHeart Block - Types, Diagnosis, Treatment, Follow-UpShehab AhmedBelum ada peringkat
- Understanding Nutritional Needs for Ulcerative ColitisDokumen7 halamanUnderstanding Nutritional Needs for Ulcerative ColitisJessica GlitterBelum ada peringkat
- Endometrial Carcinoma: Amina Adel Al-QaysiDokumen42 halamanEndometrial Carcinoma: Amina Adel Al-Qaysitaufik perdanaBelum ada peringkat
- Frontotemporal Dementia Frontotemporal DementiaDokumen4 halamanFrontotemporal Dementia Frontotemporal DementiaJenny LeeBelum ada peringkat
- Lab Diagnosis of Diarrheal DiseasesDokumen37 halamanLab Diagnosis of Diarrheal DiseasesGIST (Gujarat Institute of Science & Technology)Belum ada peringkat
- FLUID AND ELECTROLYTE MANAGEMENT IN SURGICAL PATIENTSDokumen64 halamanFLUID AND ELECTROLYTE MANAGEMENT IN SURGICAL PATIENTSTonie AbabonBelum ada peringkat
- PaclitaxelDokumen3 halamanPaclitaxelGwyn Rosales100% (1)
- NASSISMISS 2019 Conference Welcome MessageDokumen20 halamanNASSISMISS 2019 Conference Welcome MessageFariza Nur AiniBelum ada peringkat
- Colon Cancer Case Study - 64yo Male Diagnosed After Rectal BleedingDokumen2 halamanColon Cancer Case Study - 64yo Male Diagnosed After Rectal BleedingDragan Djordjevic100% (4)
- 50 Studies on Vaccines and Neurodevelopmental DisordersDokumen5 halaman50 Studies on Vaccines and Neurodevelopmental DisordersshifanahmedBelum ada peringkat
- Recent Guidelines For Hypertension: ViewpointsDokumen3 halamanRecent Guidelines For Hypertension: ViewpointsCamila ZorrillaBelum ada peringkat
- Concept of Triage - Principles, Classification and AssessmentDokumen42 halamanConcept of Triage - Principles, Classification and AssessmentAnandha SilviBelum ada peringkat
- Ortho LMRP 2019Dokumen44 halamanOrtho LMRP 2019skBelum ada peringkat
- HNF 8 SRO Blanchard2018Dokumen11 halamanHNF 8 SRO Blanchard2018Abhinav IngleBelum ada peringkat
- CDC guidelines for treating syphilisDokumen27 halamanCDC guidelines for treating syphilisGabrielaGushikenBelum ada peringkat
- Jurnal BPH Fathan Ula SalinanDokumen15 halamanJurnal BPH Fathan Ula Salinanelsy oktaviaBelum ada peringkat
- Parathyroid AdenomaDokumen5 halamanParathyroid AdenomaSatish kumarBelum ada peringkat
- DSM V Clinical Cases - Chapter 12 Sleep Wake DisordersDokumen13 halamanDSM V Clinical Cases - Chapter 12 Sleep Wake DisordersIzzyinOzzieBelum ada peringkat
- End PointsDokumen22 halamanEnd Pointsnikhil1802Belum ada peringkat
- Sample Tweets - ZikaDokumen151 halamanSample Tweets - ZikaAlisonBelum ada peringkat
- Drug Card MotrinDokumen2 halamanDrug Card MotrinAdrianne BazoBelum ada peringkat
- Problem Prioritization 1Dokumen9 halamanProblem Prioritization 1Bernard A. EstoBelum ada peringkat
- Altitude PDFDokumen47 halamanAltitude PDFMohd AsadBelum ada peringkat
- Non-Steroidal Anti-Inflammatory Drugs (Nsaids) : Dr. Nishant B. BhansaliDokumen24 halamanNon-Steroidal Anti-Inflammatory Drugs (Nsaids) : Dr. Nishant B. Bhansaliankit ahirBelum ada peringkat
- Omssat2008 (1) (1) 1Dokumen253 halamanOmssat2008 (1) (1) 1Ganga Hospital100% (2)
- Respiratory Emergency First AidDokumen13 halamanRespiratory Emergency First AidFaithBelum ada peringkat
- Pediatric Hematologic Disorders - : Blood Blood CoagulationDokumen6 halamanPediatric Hematologic Disorders - : Blood Blood Coagulationcayla mae carlosBelum ada peringkat
- Urticaria and Angioedema - An Update On Classification and Pathogenesis PDFDokumen14 halamanUrticaria and Angioedema - An Update On Classification and Pathogenesis PDFancillaagraynBelum ada peringkat