Van Der Waals Lecture
Diunggah oleh
Edgar SanchezHak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Van Der Waals Lecture
Diunggah oleh
Edgar SanchezHak Cipta:
Format Tersedia
JO H A N N E S D .
VAN DER
AALS
The equation of state for gases and liquids
Nobel Lecture, December 12, 1910
Now that I am privileged to appear before this distinguished gathering to
speak of my theoretical studies on the nature of gases and liquids, I must
overcome my diffidence to talk about myself and my own work. Yet the
thought that you are entitled to expect that of me leads me to hope that you
will forgive me if I state my views in this field with utter conviction, even
in regard to aspects which are not yet universally known and which have
so far not achieved universal recognition. I intend to discuss in sequence:
(1) the broad outlines of my equation of state and how I arrived at it;
(2) what my attitude was and still is to that equation;
(3) how in the last four years I have sought to account for the discrepancies
which remained between the experimental results and this equation;
(4) how I have also sought to explain the behaviour of binary and ternary
mixtures by means of the equation of state.
(1) The first incentive to this my lifes work came to me when, after my
studies at university, I learned of a treatise by Clausius (1857) on the nature
of the motion which we call heat. In this treatise, which is now taught with
negligible modifications in every highschool in Holland, he showed how
Boyles law can very readily be derived on the assumption that a gas consists
of material points which move at high velocity, that this velocity is of the
order of that of sound and increases in proportion to the square root of the
absolute temperature. It was subsequently realized that this is only the root
mean square of the velocities and that, as Maxwell stated, although he erroneously considered to have proved it, there is a law for the distribution
of the velocities known as the Maxwell law. Boltzmann was the first to give
the correct proof for this velocity distribution law. Clausius treatise was a
revelation for me although it occurred to me at the same time that if a gas
in the extremely dilute state, where the volume is so large that the molecules
can be regarded as points, consists of small moving particles, this is obviously
still so when the volume is reduced; indeed, such must still be the case down
to the maximum compression and also in liquids, which can only be re-
EQUATION OF STATE FOR GASES AND LIQUIDS
255
garded as compressed gases at low temperature. Thus I conceived the idea
that there is no essential difference between the gaseous and the liquid state
of matter - that the factors which, apart from the motion of the molecules,
act to determine the pressure must be regarded as quantitatively different
when the density changes and perhaps also when the temperature changes,
but that they must be the very factors which exercise their influence throughout. And so the idea of continuity occurred to me. I then also asked myself
how the solid state behaved. Although I have not as yet seriously gone into
that question I do think that in the amorphous state the close proximity of
the molecules impedes their mutual displacement. The crystalline state definitely behaves in a slightly different way. Actually I should still be silent
on this question. Nevertheless it can now scarcely be doubted that continuity
exists between the other two states of aggregation.
As you are aware the two factors which I specified as reasons why a nondilute aggregate of moving particles fails to comply with Boyles law are
firstly the attraction between the particles, secondly their proper volume.
Turning first to the second factor I should like to point out the following:
originally I had anticipated that simply the total volume must be decreased
with the total volume of the molecules to find the volume remaining for
the motion. But closer examination showed me that matters were not so
simple. To my surprise I realized that the amount by which the volume
must be reduced is variable, that in the extremely dilute state this amount,
which I have notated b, is fourfold the molecular volume - but that this
amount decreases with decreasing external volume and gradually falls to
about half. But the law governing this decrease has still not been found. This
very point has proved to be the most difficult in the study of the equation of
state. Korteweg, Lorentz, Boltzmann, Jeans, and of my pupils, Van Laar,
my son, and Kohnstamm have subsequently worked on it. I had thought
that it was simply a matter of studying how the mean free path decreases
between the collisions of the molecules which are the consequence of their
extensiveness, i.e. of the fact that they must not be considered material points
but small particles with a real volume in common with all bodies known to
us, and so I arrived at this formula:
(A)
where bg is fourfold the molecular volume.
256
1910 J.D. VAN DER WAALS
Boltzmann has shown, however, that this is inadequate and later on Kohnstamm proved that the formula will be more complex and that for b/bg a
quotient will be obtained of two series in which powers of b g/v occur. It is
so difficult to determine the coefficients , etc. that Van Laar was compelled
to carry out fearfully long calculations when determining the second coefficient by my method ( gave rise to far less trouble). This prevented me
from proceeding further. And here I have come to the weak point in the
study of the equation of state. I still wonder whether there is a better way.
In fact this question continually obsesses me, I can never free myself from it,
it is with me even in my dreams.
As regards the other cause underlying the non-compliance of real gases
and liquids with Boyles law, i.e. the mutual attraction of the molecules, the
situation is somewhat better although here again the last word has not yet
been spoken. Using the procedure adopted by Laplace in his capillarity theory, in my continuous theory I have reduced this attraction, which acts in the
whole volume, to a surface force which acts towards the interior and thus,
together with the external pressure, holds together the moving molecules.
Laplace regards his liquid actually as a continuum; at the time he was still
unaware of molecules. And if we were dealing with stationary molecules it
would have been inadmissible to reduce the attracting forces in the interior
to just a surface force. Yet since the molecules are in motion each point in the
interior will certainly not be filled with matter at every moment. But space
may be regarded as continuously filled with matter of mean - normal - density. I shall return to this point however when I speak of my studies in recent
years. The above considerations brought me to the following formula:
which became universally known only as a result of Eilhard Wiedemanns
efforts.
(2) Having reached the second part of my lecture I must now mention my
own attitude to this equation. It will be abundantly plain from my earlier
comments that I never expected this equation, with a and b assigned a constant value, to give results numerically in agreement with experiment and
yet people almost always act as though that were my opinion. This astonishes
me as in my treatise of 1873 not only did I expressly emphasize the variability
EQUATION OF STATE FOR GASES AND LIQUIDS
257
of b but also quoted a series of b-values from Andrews' experiment in which
for small volumes the change in b with the volume is calculated. For carbon
dioxide I moved from the limiting value bg = 0.0023 down to b = 0.001565.
In this series of values the volumes of the liquid even go below the limiting
value of bg for infinite volume.
Perhaps the reason why my opinion that b is variable has rarely been regarded as seriously intended is to be found in the manner in which I calculated the critical parameters. In this calculation I had to assume b as invariable. But that was because I believed that Andrews values justified still
assigning the limiting value bg to b in the critical volume. But as I later
showed that was a mistake; the value of b in the critical volume has, of
course, slightly decreased. Yet to determine the critical volume when b
changes with the volume, the first and second differential quotients db/dv
and d2b/dv 2 also need to be known. To determine the critical volume, as I
later showed, we obtain the equation:
(C)
Only when we neglect db/dv and d 2b/dv 2 do we find vk = 3b. And it can
again be seen from this equation that what I have termed the weak point
of my theory is actually responsible for the theoretical impossibility of calculating accurately the critical volume. Using an approximation formula for
b I was able to determine v k as about 2.2 bg. This equation for determining
the critical volume I gave and used in my paper in honour of Boltzmann;
it was repeated in part in my 1910 treatises. When I had realized this large
deviation in the critical volume I also feared a large deviation in the other
critical data, i.e. non-compliance with the formulae:
To my great joy, however, and not without astonishment, I found that the
two equations used to calculate a and b from the critical data remain unchanged. That I was able to conclude from the results of Sydney Youngs
admirable experiments on the volumes of co-existing vapour and liquid
258
1910 J.D. VAN DER WAALS
phases and on the saturation pressure level at various temperatures. I showed
this recently in a treatise to which I shall refer again in the present lecture.
However I can explain it as follows: Sydney Young determines inter alia
the value of
(D)
at the critical point and finds 1/3.77. If p and T have the same values as in
my theory, providing b is imagined to be constant, the difference from 3/8, the
value which I found for pv/RT, can be ascribed wholly to the volume. And
this, we can say, is entirely correct. The critical volume is not 3 bg, but (3/2)
bg = 2.125 bg (approximately). And the product pv/RT is not 3/8 but 3/8 %/t/2.
For the value of
2
my equation gives a/pv . A value of about 6 is found for this value
from Sydney Youngs experiments. Therefore for p k we have the value:
p k = a/6vk2, and with vk = (3/2)bg the value p k = (1/27)a/b g 2 , as was
originally found, is again obtained for pk.
It will thus be seen that I have never been able to consider that the last
word had been said about the equation of state and I have continually returned to it during other studies. As early as 1873 I recognized the possibility
that a and b might vary with temperature, and it is well known that Clausius
even assumed the value of a to be inversely proportional to the absolute
temperature. Thus he thought he could with probability account for the
equation
only the half being found when a and b are held constant. The foregoing
remarks signify that this value does not arise from a change in a with temperature, but merely from the change in b with the volume.
For a long time I searched for a definite characteristic to find whether just
making b variable is sufficient to bring about complete agreement between
EQUATION OF STATE FOR GASES AND LIQUIDS
259
my formula and experiment, and in the case of remaining discrepancies,
whether perhaps it is necessary to assume variability of a and b with temperature; and now I come to my latest studies on the equation of state.
(3) In 1906 before the Royal Academy of Sciences in Amsterdam I gave a
lecture entitled: pseudo association. At the time, however, I contented myself with an oral communication. But one of my pupils, Dr. Hallo, took it
down in shorthand and later Dr. Van Rij incorporated it in his inaugural
dissertation and developed the theme further. As the criterion whether the
variability of b with the volume would be sufficient to bring about agreement between my formula and Sydney Youngs experimentally determined
liquid and vapour volumes I used in my lecture the Clapeyron principle
which, with b varying only with v and not with T, and with a constant,
leads to the equation
or
To ensure that only parameters are involved which are directly determinable
by experiment, we may also write
All the parameters on the left of this equation were accurately determined
by Sydney Young for a series of substances. At the critical temperature the
value of the left-hand side is, of course, equal to unity; but what is the value
at temperatures that are only a fraction of T k? Here it appears that as the
temperature decreases the value of the left-hand side increases, very rapidly
at first, then at an imperceptible rate.
260
1910 J.D. VAN DER WAALS
Sydney Youngs experiments only go as far as a value of T/T k = 2/3. The
right-hand side has then increased to 1.4 and seems to be approaching
asymptotically the value 1.5. The value of the right-hand side may very
accurately be represented by the empirical formula
where T / T k = m.
At a temperature very close to the critical, e.g. m = 0.99, the increase in
this expression is as much as 0.1 whereas if the square root of 1 - m did not
occur in the formula and the increase were only to be represented by 1 - m,
it would be less by a factor of ten.
I then had to examine whether the assumption that a or b is temperaturedependent could account correctly for this increase of the right-hand side;
and were such not the case, whether another expression for the internal
pressure, which I have always written as a/v 2, could explain it. As a result
of this examination it was found that each temperature function for a and b
compatible with such a rapid initial increase of the right-hand side, must
contain VI - m. Yet above the critical temperature that would give imaginary values for a and b and is obviously at variance with the whole behaviour of gases. It also appeared that no other assumption for the internal
pressure can lead to such a rapid initial increase as the one under discussion.
It should not be forgotten that the critical temperature is actually not a
special temperature. At this temperature the co-existing densities are equal
in magnitude. That alone gives this temperature a meaning which under
all other circumstances it does not have. An abrupt jump, a rapid increase
either in a or b would make the temperature quite a special one at any level
of compression and its determination would then be possible at any density
level. In fact, bluntly speaking, the result would be: an equation of state
compatible with experimental data is totally impossible. No such equation
is possible, unless something is added, namely that the molecules associate
to form larger complexes; this year, therefore, I have given two treatises
at the Academy in Amsterdam on this possible association. I have termed it
"pseudo association" to differentiate it from the association which is of chemical origin. The possible formation of larger molecular complexes, particularly in the liquid state, has frequently been emphasized and the finding that
the assumption is necessary to achieve agreement between the state equation
EQUATION OF STATE FOR GASES AND LIQUIDS
261
and experiment will hence cause no surprise. Unfortunately my examination
is still incomplete. I have found it arduous. And I have had to make use of
every piece of evidence to derive something concrete. Nevertheless so much
has emerged that it will have to be assumed that a large number of single
molecules are required to form a new group which holds together and behaves as a new, larger unit in the molecular motion.
What is the origin of this complex formation, this pseudo association? I
was compelled to assume it because it seemed to me the only way to make
an equation of state - whichever it is - compatible with the results of measurements. However, as a result of a remark by Debye in last months Annalen der Physik, I remembered a phrase of Boltzmanns. When a few years
ago I was privileged to have him with me for some days, among the many
matters which we discussed he told me in passing that he was unable to
reduce the attraction of molecules to a surface force. At the time the significance of this remark of Boltzmanns did not dawn on me - only now
do I think I appreciate what he meant. As far back as in my treatise of 1873
I came to the conclusion that the attraction of the molecules decreases extremely quickly with distance, indeed that the attraction only has an appreciable value at distances close to the size of the molecules. At the time
this even prompted me to state that in the case of gases the collisions alone
are responsible for their exhibiting an attraction. And it must have been
Boltzmanns view that it is only admissible to adopt Laplaces procedure and
assume a surface force as a consequence of the attraction provided it does not
fall off so rapidly.* Debyes remark implied that Boltzmann had predicted
the formation of a complex. Thus, so I believe, the assumption of pseudo
association is justified from the theoretical standpoint. And now I think I
may state how I proceeded in my latest treatise which appeared in November
of this year. Pseudo association differs from true association in that the latter
is the result of new chemical forces which arise only when molecules are
combined to form e.g. double molecules, whereas pseudo association must
be ascribed wholly to the normal molecular forces. Now, since this force
diminishes so quickly, it has two consequences. Firstly it results in the formation of a complex, but that is not all. Secondly it leads to a surface pressure,
although a lower one. And this is precisely what I was compelled to assume
in my calculations were I to have some prospect of accounting for the cited
differences. I thus sought to make this clear in the following manner. Let the
number of molecules that have combined into a complex be so large that it
* Cf. L. Boltzmann, Gastheorie, I I, p. 176.
262
1910 J.D. VAN DER WAALS
is possible to speak of a molecule at the centre surrounded by a single layer
containing almost as many other molecules as is possible simultaneously.
Then, for the surrounding molecules the attraction directed towards the
interior acts only to maintain the complex; and this part of its attraction is
lost for the surface pressure. Only the forces acting outwards from these
molecules can contribute to the formation of the internal pressure. But of
course, for pseudo association as for true association the number of formed
complexes increases with decreasing temperature and volume. At the critical
point, so I was compelled to conclude, only a very small part of the weight
is present as complexes.
If pseudo association exists in a substance, there are at least two types of
molecules, namely simple and complex. I say at least two types because it
cannot be assumed that all complexes are of equal size. But as a first step I
have assumed only two types, i.e. simple molecules and n-fold molecules.
For a really scientific treatment, of course, it would be necessary to assume
all values of n as possible and to seek the law of distribution for these values.
For the time being, however, I have confined myself to assuming only a
single type of complex. We then have a binary mixture. It was very fortunate that for many years I had made a serious study of the laws of binary
mixtures - and so I come to the fourth point of my lecture. Not to demand
too much of your attention, however, I promise to be quite brief.
(4) When I first conceived the idea of utilizing my equation to study the
properties of binary mixtures I can no longer say. But even 20 years ago,
at the insistence of my friend Kamerlingh Onnes, I was able to publish a
complete theory for binary mixtures. My "Thorie molculaire dune substance compose de deux matires diffrentes" (Molecular theory of a substance made up of two different constituents) appeared in the Archives Nerlandaises for 1890. I had written it in Dutch but my esteemed friend Bosscha
undertook the difficult task of rendering it into French, a task which was all
the more exacting as I had written it in an extremely concise form and the
mathematical treatment led to particular points (plait points) and particular curves which at the time had rarely been examined in detail. Previously, however, my friend Korteweg, to whom I had communicated in broad
outlines the outcome of my examinations, had studied the mathematical
properties of these points and curves, a study which I have often found of
great use. The reasons why I hesitated so long over publication were many
and it would serve little purpose to enumerate them here. But one of the
EQUATION OF STATE FOR GASES AND LIQUIDS
263
reasons of scientific importance was the question which I kept asking myself: is it any use, until the study of the equation of state has been completed,
applying it to mixtures ? I appreciated in advance that as long as I was ignorant
of the law governing the variability of b and hence had to assume that b did
not vary with the volume, the results for many parameters would yield
values exhibiting numerically large differences as compared with the values
of these parameters as determined by experiment. Nevertheless, the consideration that even with b constant my theory had not been unimportant in
the case of a single substance gave me the hope that many phenomena would
be explained qualitatively provided that a suitable value for a and b were
introduced for mixtures. It was of great importance for me to be acquainted
with Gibbs treatises on the equilibrium of heterogeneous substances which
he had sent me immediately after their appearance. I made use inter alia of
his principle that for a given amount of substance equilibrium sets in if the
free energy is minimum for the given temperature and volume. In his
honour I named the equilibrium surface for a binary system the -surface.
For, the free energy whose significance for the equilibrium he was the first
to recognize, he always represents by the sign .
The phenomena are really not complex in the case of a simple substance
and it is an easy matter to obtain a general picture of them. It is therefore
very surprising that in a mere binary mixture they become so complex that
they have often been compared with a labyrinth. This is particularly so
where three-phase pressure can exist. And it has now become clear that they
are at least qualitatively in agreement with the "Thorie molculaire, etc."
from which they can be derived and indeed often predicted. Owing to the
many experimental studies to which it has given rise I have frequently found
the opportunity of discussing it closely in special communications. Thus in
the years 1907 to 1909 about 15 treatises of this nature have appeared in the
proceedings of the Academy at Amsterdam. It would be an impressive
number were I just to mention the names of the physicists and chemists who,
guided by this theory, have studied and still are studying binary mixtures.
It begins with Kuenen and ends with Dr. Jean Timmermans of Belgium
whose studies have still to be completed but which have already been published in part.
And if now I may be permitted to look back on the way I have come, I
must confess that it may fairly be called a detour. Immediately the necessity
of assuming association became clear to me I extended my equation of state
264
1910 J.D. VAN DER WAALS
analogously to the formula for a binary mixture and introduced a new parameter, the degree of association. This degree of association is determined by
means of Gibbs equation to which I have referred. The degree of association
thus determined must then be introduced into the equation of state. And I
confess that this is a detour. Perhaps there is a direct way. That this way is
seriously being sought I know from those in direct contact with me. In the
search for this way Gibbs Elementary Principles of Statistical Mechanics will be
a necessary guide. In this lecture, I have only given the history of the origin
and further elaboration of my theoretical studies and therefore had to speak
of the difficulties that had to be surmounted. Consequently I could not, or
only meagrely, discuss how they have assisted in correctly understanding the
phenomena. One of the main conclusions, which I have termed the "Law
of corresponding states", has, I may say, become universally known. Nor
have I discussed how this law was a potent contributory factor in Dewars
determination of the method of liquefying hydrogen, and particularly in
Kamerlingh Onnes determination of the method of liquefying helium. I
have also forgone detailed discussion of the temperature at which, to use
Regnaults nomenclature, a gas starts to behave like a "pluperfect gas" (gaz
plus que parfait), which temperature has been found to have a value of
(27/8) T k - besides
the temperature at which it may be stated that the JouleKelvin effect reverses: its value is found to be (27/4) T k.
Neither have I spoken of my perhaps somewhat overhasty efforts to
determine the equation of state of the molecule itself. I have even omitted
to say why I thought I had to go to such trouble to determine the relation
between p, v and T for a substance. The formulae of thermodynamics are
effective and can actually be applied in all problems, even to determining
thermal parameters, only when this relation is known, otherwise they can
be regarded as just one equation between two unknowns. I have explicitly
emphasised this in the book in commemoration of Kamerlingh Onnes published in 1904.
But it was not my intention to discuss all that and I would not have had
sufficient time. Yet it does not seem to me superfluous, perhaps it is even
necessary, to make a general observation. It will be perfectly clear that in
all my studies I was quite convinced of the real existence of molecules, that
I never regarded them as a figment of my imagination, nor even as mere
centres of force effects. I considered them to be the actual bodies, thus what we
term "body" in daily speech ought better to be called "pseudo body". It is
an aggregate of bodies and empty space. We do not know the nature of a
EQUATION OF STATE FOR GASES AND LIQUIDS
265
molecule consisting of a single chemical atom. It would be premature to
seek to answer this question but to admit this ignorance in no way impairs
the belief in its real existence. When I began my studies I had the feeling
that I was almost alone in holding that view. And when, as occurred already
in my 1873 treatise, I determined their number in one gram-mol, their size
and the nature of their action, I was strengthened in my opinion, yet still
there often arose within me the question whether in the final analysis a
molecule is a figment of the imagination and the entire molecular theory
too. And now I do not think it any exaggeration to state that the real
existence of molecules is universally assumed by physicists. Many of those
who opposed it most have ultimately been won over, and my theory may
have been a contributory factor. And precisely this, I feel, is a step forward.
Anyone acquainted with the writings of Boltzmann and Willard Gibbs will
admit that physicists carrying great authority believe that the complex
phenomena of the heat theory can only be interpreted in this way. It is a
great pleasure for me that an increasing number of younger physicists find
the inspiration for their work in studies and contemplations of the molecular
theory. The crowning of my studies by the esteemed Royal Swedish Academy affords me satisfaction and fills me with gratitude, a gratitude which I
cannot call eternal and in my old age I cannot even promise that it will be
of long duration, but perhaps for this very reason my gratitude is all the
more intense.
Anda mungkin juga menyukai
- The Equation of State For Gases and Liquids: Nobel Lecture, December 12, 1910Dokumen12 halamanThe Equation of State For Gases and Liquids: Nobel Lecture, December 12, 1910AMIT PRAJAPATIBelum ada peringkat
- The Equation of State For Gases and Liquids: Nobel Lecture, December 12, 1910Dokumen12 halamanThe Equation of State For Gases and Liquids: Nobel Lecture, December 12, 1910Nataly CGBelum ada peringkat
- De Broglie WavelengthDokumen3 halamanDe Broglie WavelengthMarion Alyssa100% (1)
- ArticleDokumen9 halamanArticleStella MaryBelum ada peringkat
- GG Stokes 1845 PDFDokumen55 halamanGG Stokes 1845 PDFAnkit soniBelum ada peringkat
- Van Der Waals EquationDokumen10 halamanVan Der Waals EquationBraulio CasanuevaBelum ada peringkat
- Inversion EquationDokumen4 halamanInversion EquationAdil ShaffeeBelum ada peringkat
- Real Gases ArticlesDokumen15 halamanReal Gases ArticlesHabibBelum ada peringkat
- Fundamentals of Chemistry Course Code: ENV-1105Dokumen101 halamanFundamentals of Chemistry Course Code: ENV-1105Mashrufa HussainBelum ada peringkat
- Investigations On The Theory of The Photographic Process - Sheppard and MeesDokumen366 halamanInvestigations On The Theory of The Photographic Process - Sheppard and MeesJorgeCamachoBelum ada peringkat
- A. Chamblin and R. Emparan - Bubbles in Kaluza-Klein Theories With Spacelike or Timelike Internal DimensionsDokumen12 halamanA. Chamblin and R. Emparan - Bubbles in Kaluza-Klein Theories With Spacelike or Timelike Internal DimensionsHuntsmithBelum ada peringkat
- Chemistry 9 NotesDokumen4 halamanChemistry 9 NotesMuhammad AsifBelum ada peringkat
- Chapter Review 3Dokumen4 halamanChapter Review 3Othello McGinnisBelum ada peringkat
- Satish Chandra: Unit - I, Real GasesDokumen6 halamanSatish Chandra: Unit - I, Real GasesSanchari BiswasBelum ada peringkat
- EOS Van - Der - Waals - Equation Nov 2 2011Dokumen7 halamanEOS Van - Der - Waals - Equation Nov 2 2011Gilvan PirôpoBelum ada peringkat
- Equation of State - WikipediaDokumen78 halamanEquation of State - WikipediaPrecious OluwadahunsiBelum ada peringkat
- Bubbling Kun NiDokumen7 halamanBubbling Kun NiBelen CRBelum ada peringkat
- Igcse 52 Solidsliquids&GasesDokumen39 halamanIgcse 52 Solidsliquids&GasesHany ElGezawy100% (1)
- Properties of Liquids and Intermolecular Forces: General Chemistry 2Dokumen53 halamanProperties of Liquids and Intermolecular Forces: General Chemistry 2Cielo GatdulaBelum ada peringkat
- Time and Background Independence: A ResponseDokumen7 halamanTime and Background Independence: A ResponsecalamartBelum ada peringkat
- General Chemistry 2 - q3 - Slm2Dokumen10 halamanGeneral Chemistry 2 - q3 - Slm2Jonnel RoqueBelum ada peringkat
- SS1 Chemistry 2nd Term Lesson Note PDFDokumen58 halamanSS1 Chemistry 2nd Term Lesson Note PDFKelly Isaac100% (3)
- Properties of MatterDokumen8 halamanProperties of MatterAbdur RahmanBelum ada peringkat
- Physical PharmacyDokumen190 halamanPhysical PharmacyKate EvangelistaBelum ada peringkat
- Thermal Physics NotesDokumen17 halamanThermal Physics NotesNo PainBelum ada peringkat
- Pit Zer 1981Dokumen8 halamanPit Zer 1981Niraj ThakreBelum ada peringkat
- LMS TEMPLATE - NOTES Chap 4Dokumen6 halamanLMS TEMPLATE - NOTES Chap 4AlexBelum ada peringkat
- Can Thought Experiments Be Resolved by Experiment? The Case of Aristotle's WheelDokumen20 halamanCan Thought Experiments Be Resolved by Experiment? The Case of Aristotle's WheelItsmeBelum ada peringkat
- A Statistical Theory of Monomolecular Reactions PDFDokumen9 halamanA Statistical Theory of Monomolecular Reactions PDFwenhuishenBelum ada peringkat
- Kinetic Theory (The Gas Laws) - Chemistry Unit IDokumen6 halamanKinetic Theory (The Gas Laws) - Chemistry Unit Imcleodtravis14Belum ada peringkat
- Physica: Viscosity of Carbon Dioxide in The Liquid PhaseDokumen32 halamanPhysica: Viscosity of Carbon Dioxide in The Liquid PhasejtorerocBelum ada peringkat
- G10 SCI Q4WK1 LAS1-checked-and-editedDokumen5 halamanG10 SCI Q4WK1 LAS1-checked-and-editedKirsten Tiffany TriasBelum ada peringkat
- 8 Interpretation Theory of Variables.: Suggested of The Quantum in Tel@asDokumen14 halaman8 Interpretation Theory of Variables.: Suggested of The Quantum in Tel@asSaad KhalidBelum ada peringkat
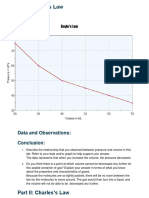
- Data and Observations: ConclusionDokumen2 halamanData and Observations: ConclusionStephany LeviBelum ada peringkat
- Materi 04 - Kimia Teknik - by Waluyo Nuswantoro 2021Dokumen11 halamanMateri 04 - Kimia Teknik - by Waluyo Nuswantoro 2021Ryan BageurBelum ada peringkat
- The History of EntropyDokumen32 halamanThe History of EntropyTiago MartinelliBelum ada peringkat
- 40 Chemistry1702951908Dokumen62 halaman40 Chemistry1702951908ilegbedionkBelum ada peringkat
- Science10 Q4 Mod1 BoyleslawDokumen17 halamanScience10 Q4 Mod1 BoyleslawPrincess PanulayaBelum ada peringkat
- Cadeliña - J - Output Week 1 B in General Chemistry 2Dokumen2 halamanCadeliña - J - Output Week 1 B in General Chemistry 2Jordano CadeliñaBelum ada peringkat
- 1923 Nature V111 p532-233Dokumen3 halaman1923 Nature V111 p532-233Ellena MaggyvinBelum ada peringkat
- Science 10 Las 4-1Dokumen5 halamanScience 10 Las 4-1Michael TuyayBelum ada peringkat
- POGIL: Kinetic Molecular Theory: Learning ObjectivesDokumen3 halamanPOGIL: Kinetic Molecular Theory: Learning ObjectivesSir JoshBelum ada peringkat
- Unsolved Problems in Fluid MechanicsDokumen11 halamanUnsolved Problems in Fluid MechanicsSameh AhmedBelum ada peringkat
- Equation of State: From Wikipedia, The Free EncyclopediaDokumen11 halamanEquation of State: From Wikipedia, The Free EncyclopediaPatricia de LeonBelum ada peringkat
- HW2 WordDokumen3 halamanHW2 WordRudiele SchankoskiBelum ada peringkat
- M2 For Printing PDFDokumen24 halamanM2 For Printing PDFcute chubbitBelum ada peringkat
- Q4 Module 1 CompressedDokumen2 halamanQ4 Module 1 CompressedFELIX ROBERT VALENZUELABelum ada peringkat
- Micelles, Vesicles and Micro EmulsionsDokumen29 halamanMicelles, Vesicles and Micro EmulsionsBárbara Damasceno BragaBelum ada peringkat
- Physical Science - Week 25Dokumen7 halamanPhysical Science - Week 25Mira VeranoBelum ada peringkat
- Entropy: Gibbs' Paradox and The Definition of EntropyDokumen4 halamanEntropy: Gibbs' Paradox and The Definition of Entropynoro70Belum ada peringkat
- Chem. LabDokumen5 halamanChem. LabCel AyunayunBelum ada peringkat
- CEGG2Dokumen26 halamanCEGG2miletinhoBelum ada peringkat
- Resource Guide: - Ideal Gases/Acid Rain & The Greenhouse EffectDokumen22 halamanResource Guide: - Ideal Gases/Acid Rain & The Greenhouse Effectkatherine_strangosBelum ada peringkat
- General Chemistry 1 Week 5 6Dokumen10 halamanGeneral Chemistry 1 Week 5 6Emmanuel ValenzuelaBelum ada peringkat
- Lesson Plan About Gay Lussacs Law 1Dokumen10 halamanLesson Plan About Gay Lussacs Law 1Lyca Mae De Villa100% (2)
- 1407 3162 PDFDokumen97 halaman1407 3162 PDFAnonymous 80p9OVBelum ada peringkat
- The Three States of MatterDokumen25 halamanThe Three States of MatterJelani GreerBelum ada peringkat
- 101.12 IMF Fa19Dokumen6 halaman101.12 IMF Fa19Jossmyr NarceBelum ada peringkat
- Gaussian Process TutorialDokumen59 halamanGaussian Process TutorialEdgar SanchezBelum ada peringkat
- BCI DYN PuRIPD V01 WS1617 Intro General Intro To ModelingDokumen54 halamanBCI DYN PuRIPD V01 WS1617 Intro General Intro To ModelingEdgar SanchezBelum ada peringkat
- Aspen User Guide 10Dokumen936 halamanAspen User Guide 10tryinghard18Belum ada peringkat
- Problem Statement: Schematic For Flow Along A ChannelDokumen9 halamanProblem Statement: Schematic For Flow Along A ChannelEdgar SanchezBelum ada peringkat
- Advanced Process DesignDokumen3 halamanAdvanced Process DesignEdgar SanchezBelum ada peringkat
- Basic EquationsDokumen19 halamanBasic EquationsEdgar SanchezBelum ada peringkat
- InfluenceOfViscous&BouyancyForcesOnMobilzOfResidualTCE PennellEtal EST30 (1996)Dokumen8 halamanInfluenceOfViscous&BouyancyForcesOnMobilzOfResidualTCE PennellEtal EST30 (1996)Edgar SanchezBelum ada peringkat
- Levenspiel 1999 - Chemical Reaction EngineeringDokumen4 halamanLevenspiel 1999 - Chemical Reaction EngineeringDrudervenBelum ada peringkat
- Indoor EcosystemsDokumen3 halamanIndoor EcosystemsEdgar SanchezBelum ada peringkat
- Rayner-Canham 5e Appendix 1 - Thermodynamic Properties of Some Selected Inorganic CompoundsDokumen12 halamanRayner-Canham 5e Appendix 1 - Thermodynamic Properties of Some Selected Inorganic CompoundsEdgar SanchezBelum ada peringkat
- Nucleosintesis 2Dokumen8 halamanNucleosintesis 2Edgar SanchezBelum ada peringkat
- Electric Vehicle Charging Station BplanDokumen19 halamanElectric Vehicle Charging Station BplanAjithBelum ada peringkat
- Zoomlion Gulf FZE Introduction: 1.1 ME Service Support 1.2 Construction CasesDokumen13 halamanZoomlion Gulf FZE Introduction: 1.1 ME Service Support 1.2 Construction CasesArk TradingBelum ada peringkat
- Antonov 225 - The Largest - Airliner in The WorldDokumen63 halamanAntonov 225 - The Largest - Airliner in The WorldFridayFunStuffBelum ada peringkat
- MY-SDK-10000-EE-005 - Method Statement For Concrete Pole Installation - GVB Rev1Dokumen7 halamanMY-SDK-10000-EE-005 - Method Statement For Concrete Pole Installation - GVB Rev1Seeths NairBelum ada peringkat
- Faithful Love: Guitar SoloDokumen3 halamanFaithful Love: Guitar SoloCarol Goldburg33% (3)
- PhysioEx Exercise 1 Activity 1Dokumen3 halamanPhysioEx Exercise 1 Activity 1edvin merida proBelum ada peringkat
- AXIOM75 50 25 1B - Rev.6 10.000MHzDokumen4 halamanAXIOM75 50 25 1B - Rev.6 10.000MHzTürkay PektürkBelum ada peringkat
- Chapter 1Dokumen20 halamanChapter 1Li YuBelum ada peringkat
- Steinecker Boreas: Wort Stripping of The New GenerationDokumen16 halamanSteinecker Boreas: Wort Stripping of The New GenerationAlejandro Javier Delgado AraujoBelum ada peringkat
- Strategi Meningkatkan Kapasitas Penangkar Benih Padi Sawah (Oriza Sativa L) Dengan Optimalisasi Peran Kelompok TaniDokumen24 halamanStrategi Meningkatkan Kapasitas Penangkar Benih Padi Sawah (Oriza Sativa L) Dengan Optimalisasi Peran Kelompok TaniHilmyTafantoBelum ada peringkat
- Omegas Prezentacija 01Dokumen20 halamanOmegas Prezentacija 01Predrag Djordjevic100% (1)
- Where Business Happens Where Happens: SupportDokumen19 halamanWhere Business Happens Where Happens: SupportRahul RamtekkarBelum ada peringkat
- DWDMDokumen41 halamanDWDMKarthik KompelliBelum ada peringkat
- 04 - Crystallogaphy III Miller Indices-Faces-Forms-EditedDokumen63 halaman04 - Crystallogaphy III Miller Indices-Faces-Forms-EditedMaisha MujibBelum ada peringkat
- OD - SAP Connector UtilityDokumen22 halamanOD - SAP Connector UtilityShivani SharmaBelum ada peringkat
- Comparativa Microplex F40 Printronix P8220 enDokumen1 halamanComparativa Microplex F40 Printronix P8220 enangel ricaBelum ada peringkat
- Merchant Accounts Are Bank Accounts That Allow Your Business To Accept Card Payments From CustomersDokumen43 halamanMerchant Accounts Are Bank Accounts That Allow Your Business To Accept Card Payments From CustomersRohit Kumar Baghel100% (1)
- Python PyDokumen19 halamanPython Pyakhilesh kr bhagatBelum ada peringkat
- A Study On Awareness of Mutual Funds and Perception of Investors 2Dokumen89 halamanA Study On Awareness of Mutual Funds and Perception of Investors 2Yashaswini BangeraBelum ada peringkat
- E&i QC Inspector Resum and DocumentsDokumen24 halamanE&i QC Inspector Resum and DocumentsIrfan 786pakBelum ada peringkat
- Trading Book - AGDokumen7 halamanTrading Book - AGAnilkumarGopinathanNairBelum ada peringkat
- Put Them Into A Big Bowl. Serve The Salad in Small Bowls. Squeeze Some Lemon Juice. Cut The Fruits Into Small Pieces. Wash The Fruits. Mix The FruitsDokumen2 halamanPut Them Into A Big Bowl. Serve The Salad in Small Bowls. Squeeze Some Lemon Juice. Cut The Fruits Into Small Pieces. Wash The Fruits. Mix The FruitsNithya SweetieBelum ada peringkat
- Tours and Travel MNGTDokumen16 halamanTours and Travel MNGTArpita Jaiswal100% (5)
- First Certificate Star SB PDFDokumen239 halamanFirst Certificate Star SB PDFPatricia Gallego GálvezBelum ada peringkat
- Nodal Mesh AnalysisDokumen20 halamanNodal Mesh Analysisjaspreet964Belum ada peringkat
- 1.2.2.5 Packet Tracer - Connecting Devices To Build IoTDokumen4 halaman1.2.2.5 Packet Tracer - Connecting Devices To Build IoTyayasan dharamabharataBelum ada peringkat
- France 10-Day ItineraryDokumen3 halamanFrance 10-Day ItineraryYou goabroadBelum ada peringkat
- Chiba International, IncDokumen15 halamanChiba International, IncMiklós SzerdahelyiBelum ada peringkat
- A Modified Linear Programming Method For Distribution System ReconfigurationDokumen6 halamanA Modified Linear Programming Method For Distribution System Reconfigurationapi-3697505Belum ada peringkat
- User Manual - Wellwash ACDokumen99 halamanUser Manual - Wellwash ACAlexandrBelum ada peringkat
- Higgs Discovery: The Power of Empty SpaceDari EverandHiggs Discovery: The Power of Empty SpacePenilaian: 3 dari 5 bintang3/5 (30)
- A Brief History of Time: From the Big Bang to Black HolesDari EverandA Brief History of Time: From the Big Bang to Black HolesPenilaian: 4 dari 5 bintang4/5 (2193)
- A Beginner's Guide to Constructing the Universe: The Mathematical Archetypes of Nature, Art, and ScienceDari EverandA Beginner's Guide to Constructing the Universe: The Mathematical Archetypes of Nature, Art, and SciencePenilaian: 4 dari 5 bintang4/5 (51)
- Dark Matter and the Dinosaurs: The Astounding Interconnectedness of the UniverseDari EverandDark Matter and the Dinosaurs: The Astounding Interconnectedness of the UniversePenilaian: 3.5 dari 5 bintang3.5/5 (69)
- The Simulated Multiverse: An MIT Computer Scientist Explores Parallel Universes, The Simulation Hypothesis, Quantum Computing and the Mandela EffectDari EverandThe Simulated Multiverse: An MIT Computer Scientist Explores Parallel Universes, The Simulation Hypothesis, Quantum Computing and the Mandela EffectPenilaian: 4.5 dari 5 bintang4.5/5 (20)
- Quantum Spirituality: Science, Gnostic Mysticism, and Connecting with Source ConsciousnessDari EverandQuantum Spirituality: Science, Gnostic Mysticism, and Connecting with Source ConsciousnessPenilaian: 4 dari 5 bintang4/5 (6)
- Summary and Interpretation of Reality TransurfingDari EverandSummary and Interpretation of Reality TransurfingPenilaian: 5 dari 5 bintang5/5 (5)
- The Holographic Universe: The Revolutionary Theory of RealityDari EverandThe Holographic Universe: The Revolutionary Theory of RealityPenilaian: 4.5 dari 5 bintang4.5/5 (76)
- Giza: The Tesla Connection: Acoustical Science and the Harvesting of Clean EnergyDari EverandGiza: The Tesla Connection: Acoustical Science and the Harvesting of Clean EnergyBelum ada peringkat
- Knocking on Heaven's Door: How Physics and Scientific Thinking Illuminate the Universe and the Modern WorldDari EverandKnocking on Heaven's Door: How Physics and Scientific Thinking Illuminate the Universe and the Modern WorldPenilaian: 3.5 dari 5 bintang3.5/5 (64)
- Packing for Mars: The Curious Science of Life in the VoidDari EverandPacking for Mars: The Curious Science of Life in the VoidPenilaian: 4 dari 5 bintang4/5 (1395)
- Midnight in Chernobyl: The Story of the World's Greatest Nuclear DisasterDari EverandMidnight in Chernobyl: The Story of the World's Greatest Nuclear DisasterPenilaian: 4.5 dari 5 bintang4.5/5 (410)
- Let There Be Light: Physics, Philosophy & the Dimensional Structure of ConsciousnessDari EverandLet There Be Light: Physics, Philosophy & the Dimensional Structure of ConsciousnessPenilaian: 4.5 dari 5 bintang4.5/5 (57)
- The Tao of Physics: An Exploration of the Parallels between Modern Physics and Eastern MysticismDari EverandThe Tao of Physics: An Exploration of the Parallels between Modern Physics and Eastern MysticismPenilaian: 4 dari 5 bintang4/5 (500)
- Chernobyl 01:23:40: The Incredible True Story of the World's Worst Nuclear DisasterDari EverandChernobyl 01:23:40: The Incredible True Story of the World's Worst Nuclear DisasterPenilaian: 4 dari 5 bintang4/5 (264)
- Chasing Heisenberg: The Race for the Atom BombDari EverandChasing Heisenberg: The Race for the Atom BombPenilaian: 4.5 dari 5 bintang4.5/5 (8)
- The Beginning of Infinity: Explanations That Transform the WorldDari EverandThe Beginning of Infinity: Explanations That Transform the WorldPenilaian: 5 dari 5 bintang5/5 (60)
- Strange Angel: The Otherworldly Life of Rocket Scientist John Whiteside ParsonsDari EverandStrange Angel: The Otherworldly Life of Rocket Scientist John Whiteside ParsonsPenilaian: 4 dari 5 bintang4/5 (94)
- Quantum Physics: What Everyone Needs to KnowDari EverandQuantum Physics: What Everyone Needs to KnowPenilaian: 4.5 dari 5 bintang4.5/5 (49)
- Too Big for a Single Mind: How the Greatest Generation of Physicists Uncovered the Quantum WorldDari EverandToo Big for a Single Mind: How the Greatest Generation of Physicists Uncovered the Quantum WorldPenilaian: 4.5 dari 5 bintang4.5/5 (8)
- Why Time Flies: A Mostly Scientific InvestigationDari EverandWhy Time Flies: A Mostly Scientific InvestigationPenilaian: 3.5 dari 5 bintang3.5/5 (17)
- Vibration and Frequency: How to Get What You Want in LifeDari EverandVibration and Frequency: How to Get What You Want in LifePenilaian: 4.5 dari 5 bintang4.5/5 (13)
- The End of Everything: (Astrophysically Speaking)Dari EverandThe End of Everything: (Astrophysically Speaking)Penilaian: 4.5 dari 5 bintang4.5/5 (157)