EXP5procedure PDF
Diunggah oleh
Geneva OrañoDeskripsi Asli:
Judul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
EXP5procedure PDF
Diunggah oleh
Geneva OrañoHak Cipta:
Format Tersedia
1
EXPERIMENT 5 - DETERMINATION OF PARTIAL MOLAR QUANTITIES
THEORY:
When one mixes 1 mole of H2O (which has a molar volume of 18 cm3) with a large
quantity of ethanol, the volume increase observed is not 18 cm 3. This simply
indicates that extensive properties of solutions (such as volume, free energy,
enthalpy, etc.) cannot be truly added when pure components are mixed. The
surrounding of a molecule is very important in determining how much volume it
will occupy, how much energy it will have, etc. So, a H2O molecule surrounded by
other molecules behaves quite differently from a H2O molecule surrounded by
ethanol molecules. The quantity of a substance in each environment is known as
the partial molar quantity of that substance in that given environment.
For a binary system with components A and B, the partial molar volume of the
component A is given by:
(1)
, ,
where V is the total volume, nA is the number of moles of A. Partial molar volume of
A is, therefore, the change in volume per mole of A added when an infinitesimal
amount of A is added to the solution at constant P and T.
The total volume of the binary solution is determined by adding the amounts of
each component.
V = n A V A+ n V B
(2)
where VA and VB are the partial molar volumes of A and B, respectively.
The total volume of a molar solution (when nT = 1) can be expressed as follows:
V= x
V A+ x V B
B
(3)
where xA and xB are the mole fractions of A and B.
It is possible to determine the partial molar volumes (or in general partial molar
quantities) of the components in a specific mixture and hence determine the total
volume of a molar solution (using equation 3) or any quantity solution (using
equation 2).
PROCEDURE:
1. Prepare approximately 25 ml samples of solutions of ethanol and water
containing 0, 20, 40, 60 80 and 100% (wt/wt) of ethanol. Prepare these
solutions by accurately weighing the two components into a stoppered bottle.
Determine the density of each solution at room temperature using the following
procedure:
a. Determine the weight of previously oven-dried and cooled pycnometer.
b. Fill up the empty pycnometer with distilled water, making sure that the
water level in the pycnometer reaches the top of the capillary and is free of
air bubbles.
c. Determine the weight of the pycnometer which is filled with water. When
doing this, make sure that the outside of the pycnometer is completely dry.
d. Given the density of water at 250C, = 0.997044 g/ml, and the density of
ethanol, = 0.791 g/ml, calculate the volume of pycnometer:
V=
W g W p
(4)
where Wg = weight of the pycnometer and water in it, Wp = weight of the
pycnometer only, and is the density of water at 250C.
e. Subsequently weigh the pycnometer which is filled with different mixtures
of ethanol and water, and calculate the densities of these solutions.
CALCULATIONS
1. The partial molar volume of ethanol (in units of ml/mol) is
M Wt
V=
density
(5)
The molecular weight of ethanol in equation 5 is 46.07 g/mol. Calculate the
specific volume (i.e. reciprocal density) of each solution.
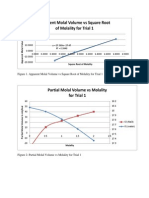
2. Plot the specific volume versus weight percent of ethanol for each solution.
3. Draw a smooth curve through all the points.
4. Draw tangent lines to this curve at different concentrations
(i.e. weight
percents).
5. Determine the y-intercepts of these tangent lines on x = 0% and x = 100%. The
former is the specific volume of water, and the latter the specific volume of
ethanol.
6. For each composition you have drawn a line and determined the specific
volumes, calculate the partial molar volume of each component using equation
5.
7. Now draw two partial molar volume versus mole fraction curves for water
and ethanol.
8. Determine the partial molar volumes for the components at xEtOH = 0.2, 0.4, 0.6
and 0.8 from the plots above.
9. Calculate the total volume of each solution using equation 2.
10. Determine the difference in the volume calculated and the volume you would
have expected to have had you not known partial molar volumes.
DATA SHEET:
Pycnometer
Wg (g)
w/ H2O
w/ 100%
EtOH
w/ 80%
EtOH
w/ 60%
EtOH
w/ 40%
EtOH
w/ 20%
EtOH
Anda mungkin juga menyukai
- Experiment 4 - Distribution (Formal Report)Dokumen5 halamanExperiment 4 - Distribution (Formal Report)joanne_blanco100% (6)
- Tutorials QuestionsDokumen3 halamanTutorials Questionskarri100% (1)
- Simple DistillationDokumen5 halamanSimple DistillationinstrutechBelum ada peringkat
- CE7 Partial Molar Volume Formal ReportDokumen24 halamanCE7 Partial Molar Volume Formal ReportIlyas Hassan100% (1)
- O Level Biology Practice Questions And Answers EnzymesDari EverandO Level Biology Practice Questions And Answers EnzymesPenilaian: 5 dari 5 bintang5/5 (1)
- Partial Molar VolumeDokumen4 halamanPartial Molar VolumeCorine CaracasBelum ada peringkat
- Problems in Mass TransferDokumen3 halamanProblems in Mass TransferAngelica Joyce BenitoBelum ada peringkat
- PVOH Chain LinkageDokumen23 halamanPVOH Chain Linkagevanessa olga100% (2)
- Concentration of SolutionsDokumen10 halamanConcentration of SolutionsBea Dacillo Bautista0% (1)
- Experiment 5 - Determination of Partial Molar QuantitiesDokumen2 halamanExperiment 5 - Determination of Partial Molar Quantitiesjayven minguillanBelum ada peringkat
- EXP5-Determination of Partial Molar QuantitiesDokumen2 halamanEXP5-Determination of Partial Molar QuantitiesJoseph Helir MuñozBelum ada peringkat
- EXP5-Determination of Partial Molar QuantitiesDokumen2 halamanEXP5-Determination of Partial Molar QuantitiesMimah MoetmainahBelum ada peringkat
- MDHKR 336 Kym 0Dokumen31 halamanMDHKR 336 Kym 0Fuse iTunesBelum ada peringkat
- Che121l Experiment 1 Partial Molar VolumesDokumen6 halamanChe121l Experiment 1 Partial Molar VolumesKim Lloyd A. Barrientos100% (1)
- Measurement of Partial Molar VolumeDokumen11 halamanMeasurement of Partial Molar Volumemogibsf100% (1)
- Experiment 2Dokumen8 halamanExperiment 2zidrick benjaminBelum ada peringkat
- Mete18 PMVDokumen5 halamanMete18 PMVOliver TabellBelum ada peringkat
- Chem 156.1 Experiment 5 - Determination of Partial Molar VolumesDokumen4 halamanChem 156.1 Experiment 5 - Determination of Partial Molar Volumesティン ヨロベBelum ada peringkat
- Equilibrim ConstantDokumen5 halamanEquilibrim ConstantArchibald MiguelBelum ada peringkat
- Experiment 3 Three Component SystemsDokumen8 halamanExperiment 3 Three Component Systemsmohammednoor_Belum ada peringkat
- Tugas Bab Campuran SederhanaDokumen2 halamanTugas Bab Campuran SederhanaMichael Lesa0% (2)
- Mol VolDokumen6 halamanMol Volティン ヨロベBelum ada peringkat
- Determine An Equilibrium ConstantDokumen13 halamanDetermine An Equilibrium ConstantMeMeMelol100% (2)
- Equilibrium Constant PupilDokumen13 halamanEquilibrium Constant PupilReinaldo RaymondBelum ada peringkat
- Exp 1 PartitionDokumen7 halamanExp 1 PartitionAnto Ondras JEllyBelum ada peringkat
- ReportDokumen4 halamanReportChadt Montague I'gautteBelum ada peringkat
- Case Study No 1. Ethanol - Water Solution Separation by Continuous DistillationDokumen10 halamanCase Study No 1. Ethanol - Water Solution Separation by Continuous DistillationLucas CorredorBelum ada peringkat
- The Partial Molar Volume 27 SeptDokumen6 halamanThe Partial Molar Volume 27 SeptmpumelaqqBelum ada peringkat
- Units of SolutionsDokumen17 halamanUnits of SolutionsamoBelum ada peringkat
- Distillation - Self Study QuestionsDokumen8 halamanDistillation - Self Study QuestionsEsther MaidenBelum ada peringkat
- Exp No.5 - LLE Phase DiagramDokumen4 halamanExp No.5 - LLE Phase DiagramPoonam ChauhanBelum ada peringkat
- Assignment Problems Batch I (R.No. 102117001 To 102117011)Dokumen7 halamanAssignment Problems Batch I (R.No. 102117001 To 102117011)Nishanth ChandranBelum ada peringkat
- DistillationDokumen6 halamanDistillationanita_shar29Belum ada peringkat
- Report1 Draft2 27-9Dokumen36 halamanReport1 Draft2 27-9Arvind RavichandranBelum ada peringkat
- Module 2 UpdatedDokumen27 halamanModule 2 UpdatedWinndell DupresBelum ada peringkat
- Chapter 1Dokumen36 halamanChapter 1maya 1DBelum ada peringkat
- Unit 5 Practical 3 - Calculating KCDokumen3 halamanUnit 5 Practical 3 - Calculating KCMuaaz IqbalBelum ada peringkat
- Methodology Conc AbstractDokumen3 halamanMethodology Conc Abstractmihau11235813Belum ada peringkat
- Density of Methanol + WaterDokumen5 halamanDensity of Methanol + WaterDanielRicardoDelgadoBelum ada peringkat
- Experiment 4 Journal ReportDokumen5 halamanExperiment 4 Journal Reportjoanne_blancoBelum ada peringkat
- ProblemsDokumen4 halamanProblemsmarslanjameelmalikBelum ada peringkat
- Exp A2Dokumen7 halamanExp A2nicolef_20Belum ada peringkat
- TDA 301T-7 - Tutorial 2 - 4 ChaptersDokumen58 halamanTDA 301T-7 - Tutorial 2 - 4 ChaptersPortia ShilengeBelum ada peringkat
- Lab Procedure Experiment 5 - PCDokumen6 halamanLab Procedure Experiment 5 - PCNur Farhana LukhmanBelum ada peringkat
- BenzoicDokumen4 halamanBenzoicdsde50% (2)
- CHP 294 NotesDokumen76 halamanCHP 294 Noteshr maBelum ada peringkat
- Quality Education For A Better Mindanao A. Bonifacio Avenue, Tibanga, 9200 Iligan City PhilippinesDokumen16 halamanQuality Education For A Better Mindanao A. Bonifacio Avenue, Tibanga, 9200 Iligan City PhilippinesClyde SuerteBelum ada peringkat
- MT Lab Final RecordDokumen85 halamanMT Lab Final RecordPRABATH MADHAVANBelum ada peringkat
- Exp. 8 PDFDokumen4 halamanExp. 8 PDFAna GonzálezBelum ada peringkat
- Solution (Compatibility Mode)Dokumen39 halamanSolution (Compatibility Mode)ApriyantiBelum ada peringkat
- Chemical EquilibriumDokumen8 halamanChemical EquilibriumjenniferBelum ada peringkat
- Chemical Equilibrium: CHE 195 Process ChemistryDokumen25 halamanChemical Equilibrium: CHE 195 Process ChemistryMohd Shahrul Nizam SallehBelum ada peringkat
- Experiment 4 Laboratory ReportDokumen13 halamanExperiment 4 Laboratory ReportXena Jenn Y. BacsarpaBelum ada peringkat
- Topic:-: SolutionsDokumen3 halamanTopic:-: SolutionsGnaneshwarBelum ada peringkat
- Unit-2 - Chemical MeasurementsDokumen15 halamanUnit-2 - Chemical Measurementsshimmy yayBelum ada peringkat
- Separation ProcessesDokumen3 halamanSeparation ProcessesJoshua SolomonBelum ada peringkat
- 4.5 Quantitative Chemical Analysis: TitrationDokumen9 halaman4.5 Quantitative Chemical Analysis: TitrationKaycee LomioBelum ada peringkat
- 9 HaftaDokumen16 halaman9 HaftaEmre KOÇBelum ada peringkat
- Record Book PCDokumen13 halamanRecord Book PCSmitBelum ada peringkat
- AnswersDokumen2 halamanAnswersGeneva OrañoBelum ada peringkat
- Fluorescence Intensity Vs Concentration of Mg2+Dokumen9 halamanFluorescence Intensity Vs Concentration of Mg2+Geneva OrañoBelum ada peringkat
- Fluorescence ExperimentDokumen2 halamanFluorescence ExperimentGeneva OrañoBelum ada peringkat
- Apparent Molal Volume Vs Square Root of Molality For Trial 1Dokumen3 halamanApparent Molal Volume Vs Square Root of Molality For Trial 1Geneva OrañoBelum ada peringkat
- Neraca MassaDokumen38 halamanNeraca MassaGunBelum ada peringkat
- Chemistry Practice Question 2Dokumen7 halamanChemistry Practice Question 2Taj AgraBelum ada peringkat
- Penyerahan Dan Penilaian TugasanDokumen4 halamanPenyerahan Dan Penilaian TugasanFAEZAL HAZLY B. MOHD NAJIB STUDENTBelum ada peringkat
- Percent CompositionDokumen23 halamanPercent CompositionJerome SarsonasBelum ada peringkat
- Stumpe 2007Dokumen9 halamanStumpe 2007VicBelum ada peringkat
- CH 14Dokumen37 halamanCH 14SingAnnBelum ada peringkat
- Concentration Expressions Serial DilutionDokumen34 halamanConcentration Expressions Serial DilutionBlessy MartinBelum ada peringkat
- KVPY 2013 SA SolutionDokumen23 halamanKVPY 2013 SA SolutionArshKhan50% (2)
- Analytical Chemistry For EngineersDokumen76 halamanAnalytical Chemistry For EngineersJmark Valentos ManalutiBelum ada peringkat
- Aspirin TitrationDokumen6 halamanAspirin TitrationAmita RanaBelum ada peringkat
- Reactor Exercises - 2013 PDFDokumen19 halamanReactor Exercises - 2013 PDFtehbear0% (1)
- Methane Production ProblemDokumen5 halamanMethane Production ProblemSheikh MuneebBelum ada peringkat
- Jadual Spesifikasi Ujian (Jsu) Tahun 2021Dokumen22 halamanJadual Spesifikasi Ujian (Jsu) Tahun 2021leong cheng liyBelum ada peringkat
- Chemistry - Exam Preparation Guide - Steve Owen - First Edition - Cambridge 2015Dokumen258 halamanChemistry - Exam Preparation Guide - Steve Owen - First Edition - Cambridge 2015purevdorj amarBelum ada peringkat
- BdeM Taller No. 1 Variables de Proceso 2017Dokumen2 halamanBdeM Taller No. 1 Variables de Proceso 2017Amilvia Vega OrozcoBelum ada peringkat
- Chapter 3 PowerpointDokumen56 halamanChapter 3 PowerpointLindseyBelum ada peringkat
- Exer 2 - Laboratory Measurements, Solutions and CalculationsDokumen10 halamanExer 2 - Laboratory Measurements, Solutions and CalculationsCheryl MountainclearBelum ada peringkat
- Concentration of SolutionsDokumen20 halamanConcentration of SolutionsRyanBelum ada peringkat
- Helpsheet 4 103Dokumen4 halamanHelpsheet 4 103Uday Prakash SahuBelum ada peringkat
- CH 4Dokumen53 halamanCH 4JayelleBelum ada peringkat
- Practice Exam With Answers PDFDokumen38 halamanPractice Exam With Answers PDFPaigeBelum ada peringkat
- 2019 Redefinition of SI Base UnitsDokumen101 halaman2019 Redefinition of SI Base UnitsGOPAL VERMABelum ada peringkat
- Limiting and Excess ReagentsDokumen22 halamanLimiting and Excess ReagentsGianna BaldoradoBelum ada peringkat
- Moles PDFDokumen9 halamanMoles PDFJinyuanLowBelum ada peringkat
- Chapter 3: Electrolytic Conductance 3.1. Specific Conductance Specific Conductance Is A Measure of The Electric Current in The Water SampledDokumen11 halamanChapter 3: Electrolytic Conductance 3.1. Specific Conductance Specific Conductance Is A Measure of The Electric Current in The Water SampledBùi Hữu ĐứcBelum ada peringkat
- Basic Chemical Concept IDokumen14 halamanBasic Chemical Concept Isujayan2005Belum ada peringkat
- Chapter3 Mole ConceptDokumen10 halamanChapter3 Mole Conceptmatyiman_123Belum ada peringkat
- Skema Pemarkahan Peperiksaan Pertengahan Tahun 2022Dokumen8 halamanSkema Pemarkahan Peperiksaan Pertengahan Tahun 2022Keeshnavi KumaranBelum ada peringkat
- Thermodynamics & Thermochemistry: Chapter Practice ProblemsDokumen2 halamanThermodynamics & Thermochemistry: Chapter Practice ProblemsAyush Chouhan100% (1)