Heme Synthesis and Porphyrias Notes
Diunggah oleh
Brett FieldsHak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Heme Synthesis and Porphyrias Notes
Diunggah oleh
Brett FieldsHak Cipta:
Format Tersedia
HEME SYNTHESIS AND PORPHYRIAS
HEME PROTEIN
o Heme is ferroprotoporphyrin IX, it is the prosthetic group of a number of
heme proteins
o Heme proteins:
Mb, Hb, mitochondrial chromosomes, microsomal cytochrome,
catalase and peroxidase, tryptophan pyrrolase
o Heme is usually associate noncovalently with its apoprotein through the
coordination of its iron with an aa side chain N
The heme in cytochrome C derivatives are an exception in being
covalently bound to the apoprotein through thioether linkages
STRUCTURE OF PORPHYRINS
o Heme is the iron chelate of protoporphyrin IX
o Porphyrins-metal free cyclic tetrapyrroles which are named according to
the nature and arrangement of their side chain groups
Positions 5,10,15,20 are C atoms that serve as bridges bt the 4
pyrrole rings (aka alpha, beta, gamma, delta)
These C bridges are unsaturated in porphyrins and are
called methene groups (-CH=)
o Because they are unsaturated, the entire structure is
conjugated, therefore, porphyrins are colored (usu a
shade or red or brown)
o Porphyrin Biosynthesis
First the molecule goes through a stage where the bridge carbons
are saturated=methylene bridge
Its presence in the molecule disrupts the conjugation of the
molecule and therefore, the tetrapyrrole is colorless
Porphyrinogen=when all the carbon bridges are methylene
groups
Poryphrins are named by the groups attached to them
Acetic Acid
Proprionic acid
Methyl
Vinyl
Occur as:
o AP, MP, MV
The porphyrins of interest to us contain 4 pyrroles in the
combinations below:
o Uropophyrin- 4AP (most soluble)
o Coproporphyrin-4 MP
o Protoporphyrin-2MP, 2MV (least soluble)
It is possible to draw a number of isomers of these
porphyrins depending on how different the pyrroles are put
together
o Type III is the naturally occurring isomer or
uroporyphyrin and coproporphyrin (APAPAPPA
and MPMPMPPM respectively)
o Protoporphyrin IX is the naturally occurring isomer
(MVMVMPPM)
These compounds can occur with methane or methylene
bridges
PROPERTIES OF PORPHYRINS
o Color
usually a shade of red or brown with a distinctive light absorption
spectrum
major absorption is at 400nm (the Soret band) with several
smaller bands between 500-630nm
metal-free porphyrins emit an intense red fluorescence
when excited by light at ~400nm, but heme does not
Porphyrin-sensitized injury aka photosensitivity-the
fluorescence in vivo of porphyrins in the presence of O damages
cells
Based on injury to plasma and lysosomal membranes
Photosensitization occurs when abnormally large quantities
of porphyrins are present either by experimentation or by a
derangement in porphyrins synthesis called porphyria
o Symptoms=itching, edema, erythema and
ulceration
Another problem due to elevated porphyrins levels is in regards to
water solubility
In general, porphyrins solubility increases as the number of
carboxylic acid side chains increases
o Uro and coproporphyrins can be excreted in urine
whereas protoporphyrin is so insoluble it must be
secreted in feces via bile
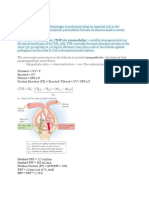
THE PATHWAY OF HEME SYNTHESIS
o The cells of most tissues can synthesize heme compounds but the liver and
bone marrow produce the most
In bone marrow, heme is inc. into Hb, the liver normally produces
15% of the total heme synthesized in the body and the bone
marrow does the rest
In rats, 65% of heme synthesized in the liver is used for the
formation of microsomal cytochrome P450 and 6% for
mitochondrial cytochromes
o The porphyrins ring is synthesized entirely from glycine and succinate (as
succinyl CoA)
2 key intermediates=aminolevulini acid ALA and porphobilinogen
PBG
o #1-8 enzymes are involved from glycine; the first and last three are in the
mitochondria requiring the cooperation of the mitochondria and cytostolic
compartments for heme synthesis
ALA IS FORMED IN THE FIRST STEP
PBG (the pyrrole precursor of heme) IS FORMED IN
THE SECOND
o 4 molecules of PBG are condensed in the
pathway to heme
o #2-ALA synthase, the first enzyme of the pathway, is dependent on
pyridoxal phosphate for activity
This coenzyme functions to form a Schiff base with the glycine
before is reats with succinyl CoA
o #3-ALA is the initial intermediate=committed to heme synthesis and
rate controlling step!
o #4-from PBG until protoporphyrin IX, the precursors of heme are
synthesized reactions involving reduced ridge Cs of the porphyrinogen
type
Only the final enzyme, ferrochelatase, uses a porphyrins instead of
a porphyrinogen
o #5-Reduced iron (Fe2+) is incorporated into heme by ferrochelatase
o #6-PBG deaminase catalyzes the condensation of PBG molecules to form
a linear tetrapyrrole
When PBG acts in the presence of a 2nd enzyme, uroporphyrinogen
III cosynthase, proper closure of the tetrapyrrole occurs
In the absence of this enzyme, type I is formed which is unusable
o #7-uroporphyrinogen III is the first porphyrinogen formed in the
biosynthesis of heme
It is converted to protoporyphyrin IX by enzymatic reaction of its
side chains
4 acetic groups methly groups
2 proprionic acid groupsvinyl groups
REGULATION OF HEME BIOSYNTHESIS
o In liver:
ALA synthase (the 1st enzyme of the pathway) catalyzes the ratelimiting step and is the major point of regulation of the pathway in
the liver (but not in erythroid cells)
level of ALA synthase is regulated by the concentration of
free heme, the product of the pathway, and the
autooxidation product hemin (heme with Fe+3)
o There are 3 mechanisms which might regulate the rate of heme synthesis
1. Feedback inhibition of the isolated enzyme by hemin
Does not seem to be physiologically important
2. Hemin represses the synthesis of ALA synthase
3. Hemin inhibits the transport of cytosolic ALA synthase into the
mitochondria
THEREFORE, HIGH LEVELS OF HEME PREVENT THE
SYNTHESIS OF HEME!
o Many compounds increase ALA synthase and result in overproduction of
liver ALA, PBF and porphyrins
Ie. Phenobarbital-related barbiturates, naturally occurring
steroids (including estrogens) and other drugs
o There are tissue-specific ALA synthase isozymes which are encoded by 2
separate genes
ALAS1-housekeeping ALA synthase gene expressed in liver and
non-erythyroid tissues
ALAS2-expressed only in erythroid cells
o In erythroid cells:
The major regulation of heme synthesis in these cells is no via the
repression of ALA synthesis by heme as in the liver
In fact, hemin stimulates heme formation in several types of
erythroid cells
There is an erythroid-specific ALA synthase gene which has a
distinct mode of regulation tied to cell differentiation
ALAS-E is not inducible by the drugs that induce ALAS1
PORPHYRIA
o A group of related but distinct diseases, they are inherited and acquired
diseases characterized by:
Defects in specific enzymes in heme biosynthesis
Increased accumulation and excretion of intermediates of the
pathway
Most dramatic clinical symptoms=neurological disorders and
cutaneous photosensitivity
Excess ALA and PBF=peripheral neuritis, abdominal pain
and intestinal spasms
Excess porphyrins in plasma(rather than in blood
cells)=skin lesions
o Porphyrins transported by albumin
CLASSIFICATION OF PORPHYRIAS
o Classified as erythropoietic or hepatic, symptoms are essentially the same
bc overproduced porphyrins can enter plasma from both sources
COMMENTS ON PORPHYRIAS
o Erythropoeitic porphyrias
Congenital erythropoietic porphyria (CEP) is very rare
Severe photosensitivity, red cell hemolysis
Erythropoietic porphyria protoporphyria (EPP)
Usually exhibits milder clinical symptoms with little or no
hemolysis
o Hepatic Porphyrias
5-aminolevulinic acid dehydratase-deficient porphyria (ADP),
acute intermittent porphyria (AIP), porphyria cutanea tarta (PCT),
hereditary coproporphyria (HCP) and variegate porphyria (VP)
Variable degree of clinical expression
Hormonal, drug and nutritional factors predispose patients
to full expression of the disease
o These agents precipitate clinical expression in
asymptomatic patients carrying the genetic defect
o Most act by inducing higher levels of ALA synthase
o PCT is fairly common
More frequent in males over 40 who drink
and women on birth control
o ADP is extremely rare!
One would expect in some cases the accumulation of the gens in
the urine, but porphyrinogens oxidize spontaneously to the
porphyrins form during and after excretion; thus, a urine specimen
will gradually darken if left standing
The enzymatic defect in various porphyrias is genetic
Enzymatic activity is REDUCED
LEAD POISONING
o Usually occurs from the ingestion of peeling leaded paints or other
exposure
o Symptoms are similar to those AIP
o Lead tends to concentrate in erythroid cells
o Erythrocyte ALA dehydratase and ferrochelatase are particularly sensitive,
resulting in a kind of acquired porphyria
o Inhibits erythrocyte heme synthesis more than liver heme synthesis
o Excess excretion of ALA and proto IX and Copro III
o Possible photosensitiviy and noticeable anemia
o Abodominal and neurological problems
Anda mungkin juga menyukai
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDari EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifePenilaian: 4 dari 5 bintang4/5 (5794)
- Shoe Dog: A Memoir by the Creator of NikeDari EverandShoe Dog: A Memoir by the Creator of NikePenilaian: 4.5 dari 5 bintang4.5/5 (537)
- Generalized Anxiety Disorder - 124169 PDFDokumen8 halamanGeneralized Anxiety Disorder - 124169 PDFblackcatBelum ada peringkat
- Helping Children Cope With DisastersDokumen8 halamanHelping Children Cope With DisastersBrett FieldsBelum ada peringkat
- Im So Stressed OutDokumen2 halamanIm So Stressed OutBrett FieldsBelum ada peringkat
- I'm So Stressed InfographicDokumen1 halamanI'm So Stressed InfographicBrett FieldsBelum ada peringkat
- Things You Should Know About StressDokumen2 halamanThings You Should Know About StressBrett FieldsBelum ada peringkat
- What Is Borderline Personality Disorder?Dokumen8 halamanWhat Is Borderline Personality Disorder?aagBelum ada peringkat
- Zen Den - EditDokumen3 halamanZen Den - EditBrett FieldsBelum ada peringkat
- Brainactivitybook PDFDokumen17 halamanBrainactivitybook PDFBrett FieldsBelum ada peringkat
- Depression Basic NIMHDokumen6 halamanDepression Basic NIMHanastashaBelum ada peringkat
- Advanced Airway ControlDokumen1 halamanAdvanced Airway ControlBrett FieldsBelum ada peringkat
- Scene 5 - 2nd RevisionDokumen3 halamanScene 5 - 2nd RevisionBrett FieldsBelum ada peringkat
- Notification of Housing AllowanceDokumen1 halamanNotification of Housing AllowanceBrett FieldsBelum ada peringkat
- Depresie - Brosura 1Dokumen2 halamanDepresie - Brosura 1Diana TeodorovaBelum ada peringkat
- Academic Paper - FieldsDokumen8 halamanAcademic Paper - FieldsBrett FieldsBelum ada peringkat
- Environmental PathologyDokumen16 halamanEnvironmental PathologyBrett FieldsBelum ada peringkat
- DS82 Complete PDFDokumen6 halamanDS82 Complete PDFAnonymous FQPDtx6Belum ada peringkat
- HHE ItineraryDokumen6 halamanHHE ItineraryBrett FieldsBelum ada peringkat
- Nephrin (Protein in Slit Diaphragm of Podocytes) Plays An Essential Role in TheDokumen3 halamanNephrin (Protein in Slit Diaphragm of Podocytes) Plays An Essential Role in TheBrett FieldsBelum ada peringkat
- Nephrin (Protein in Slit Diaphragm of Podocytes) Plays An Essential Role in TheDokumen3 halamanNephrin (Protein in Slit Diaphragm of Podocytes) Plays An Essential Role in TheBrett FieldsBelum ada peringkat
- Slow Jam Hold On Riff Procrastiplayers Wednesday Night Jam Amazon G Jam I Wanna I Ain't Got It Grooveman Nothing But The Funk I Wanna Get in ItDokumen1 halamanSlow Jam Hold On Riff Procrastiplayers Wednesday Night Jam Amazon G Jam I Wanna I Ain't Got It Grooveman Nothing But The Funk I Wanna Get in ItBrett FieldsBelum ada peringkat
- Common Drugs ListDokumen3 halamanCommon Drugs ListBrett FieldsBelum ada peringkat
- HormonesDokumen48 halamanHormonesBrett FieldsBelum ada peringkat
- Lymphoproliferative DisordersDokumen36 halamanLymphoproliferative DisordersBrett FieldsBelum ada peringkat
- CAT - Critical AppraisalDokumen1 halamanCAT - Critical AppraisalBrett FieldsBelum ada peringkat
- Boardcommittee Meeting AgendaDokumen1 halamanBoardcommittee Meeting AgendaBrett FieldsBelum ada peringkat
- Urology Smart PhrasesDokumen1 halamanUrology Smart PhrasesBrett FieldsBelum ada peringkat
- Optogenetics UC 2014Dokumen6 halamanOptogenetics UC 2014Brett FieldsBelum ada peringkat
- JAMA 1998 Fontanarosa 1307 9Dokumen3 halamanJAMA 1998 Fontanarosa 1307 9Brett FieldsBelum ada peringkat
- Behaviors (Evidenced by The Analysis Results Below)Dokumen2 halamanBehaviors (Evidenced by The Analysis Results Below)Brett FieldsBelum ada peringkat
- The Yellow House: A Memoir (2019 National Book Award Winner)Dari EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Penilaian: 4 dari 5 bintang4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDari EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RacePenilaian: 4 dari 5 bintang4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDari EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersPenilaian: 4.5 dari 5 bintang4.5/5 (344)
- The Little Book of Hygge: Danish Secrets to Happy LivingDari EverandThe Little Book of Hygge: Danish Secrets to Happy LivingPenilaian: 3.5 dari 5 bintang3.5/5 (399)
- Grit: The Power of Passion and PerseveranceDari EverandGrit: The Power of Passion and PerseverancePenilaian: 4 dari 5 bintang4/5 (588)
- The Emperor of All Maladies: A Biography of CancerDari EverandThe Emperor of All Maladies: A Biography of CancerPenilaian: 4.5 dari 5 bintang4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDari EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaPenilaian: 4.5 dari 5 bintang4.5/5 (266)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDari EverandNever Split the Difference: Negotiating As If Your Life Depended On ItPenilaian: 4.5 dari 5 bintang4.5/5 (838)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDari EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryPenilaian: 3.5 dari 5 bintang3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealDari EverandOn Fire: The (Burning) Case for a Green New DealPenilaian: 4 dari 5 bintang4/5 (73)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDari EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FuturePenilaian: 4.5 dari 5 bintang4.5/5 (474)
- Team of Rivals: The Political Genius of Abraham LincolnDari EverandTeam of Rivals: The Political Genius of Abraham LincolnPenilaian: 4.5 dari 5 bintang4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDari EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyPenilaian: 3.5 dari 5 bintang3.5/5 (2259)
- Rise of ISIS: A Threat We Can't IgnoreDari EverandRise of ISIS: A Threat We Can't IgnorePenilaian: 3.5 dari 5 bintang3.5/5 (137)
- The Unwinding: An Inner History of the New AmericaDari EverandThe Unwinding: An Inner History of the New AmericaPenilaian: 4 dari 5 bintang4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDari EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You ArePenilaian: 4 dari 5 bintang4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Dari EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Penilaian: 4.5 dari 5 bintang4.5/5 (120)
- Her Body and Other Parties: StoriesDari EverandHer Body and Other Parties: StoriesPenilaian: 4 dari 5 bintang4/5 (821)
- Acute GastroenteritisDokumen8 halamanAcute Gastroenteritisleizel_seranBelum ada peringkat
- C 13Dokumen11 halamanC 13Jo100% (1)
- November 2008 NLE Practice IIDokumen19 halamanNovember 2008 NLE Practice IIEJ Cubero, R☤N100% (1)
- Food Safety Practices Among Street Food Vendors in Mayantoc PlazaDokumen5 halamanFood Safety Practices Among Street Food Vendors in Mayantoc PlazaHACKER WiFI100% (2)
- Matthew J. Friedman (Editor), Paula P. Schnurr (Editor), Terence M. Keane (Editor), Chadi Abdallah (Editor), Garrett B. Aikens (Editor) - Handbook of PTSD - Science and Practice-Guilford Press (2021)Dokumen691 halamanMatthew J. Friedman (Editor), Paula P. Schnurr (Editor), Terence M. Keane (Editor), Chadi Abdallah (Editor), Garrett B. Aikens (Editor) - Handbook of PTSD - Science and Practice-Guilford Press (2021)CESAR DARIO FALCON LIZANO100% (8)
- Neuroscience of GeniusDokumen222 halamanNeuroscience of GeniusGareth Thomas100% (7)
- Spinal SurgeryDokumen19 halamanSpinal SurgerySalomeSibashviliBelum ada peringkat
- Cerebral PalsyDokumen96 halamanCerebral PalsyRahini PaniBelum ada peringkat
- Case Taking in Mental IllnessDokumen38 halamanCase Taking in Mental IllnessDrnarendra KumarBelum ada peringkat
- English Task The Summery of Health Illness, Part of The Body, Medical Practicioners, Oncology and X-Ray & CT ScanDokumen7 halamanEnglish Task The Summery of Health Illness, Part of The Body, Medical Practicioners, Oncology and X-Ray & CT ScanYudha HupayantiBelum ada peringkat
- Abdominal Pains in Children Under 12Dokumen5 halamanAbdominal Pains in Children Under 12clubsanatateBelum ada peringkat
- 04 - Typhoid FeverDokumen35 halaman04 - Typhoid Feversoheil100% (1)
- Gestational HypertensionDokumen71 halamanGestational HypertensionDevikha PeremelBelum ada peringkat
- Mental Health AwarenessDokumen13 halamanMental Health AwarenessMaxine AguilarBelum ada peringkat
- Liver Function TestsDokumen16 halamanLiver Function TestsAnjali Devi100% (1)
- AgniDokumen12 halamanAgniAbhishek SharmaBelum ada peringkat
- Quarter ApplicationDokumen1 halamanQuarter ApplicationSouvik DasBelum ada peringkat
- Interpretasi ElektrokardiografiDokumen60 halamanInterpretasi ElektrokardiografiAditya Praja'schBelum ada peringkat
- Restrictive Lung DiseasesDokumen31 halamanRestrictive Lung DiseasesAnn FrancisBelum ada peringkat
- Edge Booty WorkbookDokumen102 halamanEdge Booty WorkbookEricaBerger94% (16)
- The Patient History An Evidence-Based Approach To Differential DiagnosisDokumen4 halamanThe Patient History An Evidence-Based Approach To Differential DiagnosisISAYK13% (8)
- Measles: Late Winter, Early Spring and Rainy SeasonDokumen10 halamanMeasles: Late Winter, Early Spring and Rainy SeasonReah Jayzel AlkuinoBelum ada peringkat
- Fungal Infections Are More Common Caused byDokumen28 halamanFungal Infections Are More Common Caused byPrashun Shekhar SrivastavaBelum ada peringkat
- Test Bank Chapter 11: End-of-Life and Palliative Care: Lewis: Medical-Surgical Nursing, 7 EditionDokumen7 halamanTest Bank Chapter 11: End-of-Life and Palliative Care: Lewis: Medical-Surgical Nursing, 7 EditionBriseidaSolisBelum ada peringkat
- Clinical Applications of Neuroimaging in PsychiatryDokumen10 halamanClinical Applications of Neuroimaging in PsychiatryJujuBelum ada peringkat
- PericarditisDokumen3 halamanPericarditisKhalid Mahmud Arifin0% (1)
- Bio Lab 3Dokumen4 halamanBio Lab 3Nkosi Jupiter100% (1)
- Lab Manual VPE311Dokumen40 halamanLab Manual VPE311Friends ZoneBelum ada peringkat
- Slide Chronic Pain-Rudy Hidayat-Iai PDFDokumen30 halamanSlide Chronic Pain-Rudy Hidayat-Iai PDFAsri ArrachmanBelum ada peringkat
- Ampicillin Sodium - Sulbactam Sodium Drug StudyDokumen1 halamanAmpicillin Sodium - Sulbactam Sodium Drug StudyMelissa Marie Custodio100% (3)