Predicting Acid/Base Strength from Structure
Diunggah oleh
judyli990%(1)0% menganggap dokumen ini bermanfaat (1 suara)
79 tayangan15 halamanpowerpoint
Judul Asli
Acids & Bases Strength Prediction
Hak Cipta
© © All Rights Reserved
Format Tersedia
PDF, TXT atau baca online dari Scribd
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen Inipowerpoint
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PDF, TXT atau baca online dari Scribd
0%(1)0% menganggap dokumen ini bermanfaat (1 suara)
79 tayangan15 halamanPredicting Acid/Base Strength from Structure
Diunggah oleh
judyli99powerpoint
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PDF, TXT atau baca online dari Scribd
Anda di halaman 1dari 15
Predicting the Strength of Acid / Base
The stronger the acid, the weaker will be its conjugate base.
(The larger the pKa of the conjugate acid, the stronger the base)
Can we predict the relative basicity of two bases ?
Predicting the Strength of Acid / Base
The stronger the acid, the weaker will be its conjugate base.
(The larger the pKa of the conjugate acid, the stronger the base)
Can we predict the direction of equilibrium of the following reactions?
Predicting the Strength of Acid / Base
The stronger the acid, the weaker will be its conjugate base.
(The larger the pKa of the conjugate acid, the stronger the base)
The relationship between structure (size) and acidity
FHO-
Cl-
s
e
s
a
re
c
In
y
it
il
b
ta
S
Br-
B
a
HS-
I-
HSe-
The extent of electron delocalization (overall size) of the
corresponding conjugate bases may correlate with the acidity.
Predicting the Strength of Acid / Base
The stronger the acid, the weaker will be its conjugate base.
(The larger the pKa of the conjugate acid, the stronger the base)
The relationship between electronegativity and acidity
Predicting the Strength of Acid / Base
The stronger the acid, the weaker will be its conjugate base.
(The larger the pKa of the conjugate acid, the stronger the base)
The relationship between structure / electronegativity and acidity
Predicting the Strength of Acid / Base
The stronger the acid, the weaker will be its conjugate base.
(The larger the pKa of the conjugate acid, the stronger the base)
The relationship between structure / hybridization and acidity
*The higher the s-character, the higher is the carbons electronegativity.
H
H
C C
H C C H
H
H
H
C C
H
Acidity Increases
H
H C C
C C
H
Basicity Increases
C C
H
The Strength of Acid / Base: pKa
The Strength of Acid / Base: Inductive Effect
Inductive Effect: propagation of bond polarity through -bond
network (decrease with the distance)
The Strength of Acid / Base: Resonance Effect
requires energy
for the change
endothermic
Resonance Effect: stabilization of the system by having resonance
structures (increase with the # of equivalent resonance structures)
pKa vs. G (Standard Free-Energy Change)
requires energy
for the change
endothermic
G = RT ln Keq
pKa = log Ka
G = 2.303RT (log Ka)
= 2.303 (8.314x10-3 kj/molK)
x (273+25)K (log Ka)
= (5.7 kj/mol) x 4.75
= 27.1 kj/mol
Resonance Effect: stabilization of the system by having resonance
structures (increase with the # of equivalent resonance structures)
Organic Compounds as Bases
carboxylic and and alcohol as acids
carboxylic as a base
H
O
O
H
H3C
Cl
H
H3C
Cl-
The Strength of Acid / Base: pKa
Mechanism for Organic Reactions
Why acidbase?
Step 1
Many reactions in organic chemistry are:
either acidbase reaction themselves
or involves acidbase reaction at some stage
Mechanism for Organic Reactions
Why acidbase?
Many reactions in organic chemistry are:
either acidbase reaction themselves
or involves acidbase reaction at some stage
Step 1
Step 2
Step 3
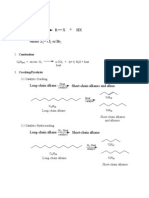
Acid / Base in Nonaqueous Solution
Leveling Effect: No stronger base than OH- can exist in water.
Synthesis of Tritium-labeled Compound
Anda mungkin juga menyukai
- Alcohols, Phenols and EthersDokumen28 halamanAlcohols, Phenols and EthersDnyanesh Shinde100% (1)
- Functional Groups - Organic ChemistryDokumen61 halamanFunctional Groups - Organic ChemistryYoAmoNYCBelum ada peringkat
- Alkenes and AlkynesDokumen2 halamanAlkenes and AlkynesLewis AlfonsoBelum ada peringkat
- Ch 16 Aromatic Compounds NotesDokumen10 halamanCh 16 Aromatic Compounds NotesVirendra Singh Rajput100% (1)
- Aromaticity With Huckle's RuleDokumen7 halamanAromaticity With Huckle's RuleSk ZBelum ada peringkat
- CHEM 4341 Syllabus Fall 2016 PDFDokumen5 halamanCHEM 4341 Syllabus Fall 2016 PDFCarmen FloresBelum ada peringkat
- Aromaticity Tutorial: Pi BondsDokumen15 halamanAromaticity Tutorial: Pi BondsAlex-Mihai Ciubara100% (2)
- Chemistry in BiologyDokumen15 halamanChemistry in BiologyJohn OsborneBelum ada peringkat
- How To Find No of Structural Isomers by S.K.sinha See Chemistry Animations at HTTP://WWW - Openchemistry.inDokumen2 halamanHow To Find No of Structural Isomers by S.K.sinha See Chemistry Animations at HTTP://WWW - Openchemistry.inmyiitchemistry81% (16)
- Thermochemistry Answers RemovedDokumen11 halamanThermochemistry Answers Removedapi-327309463Belum ada peringkat
- 3.2.1 Enthalpy ChangesDokumen9 halaman3.2.1 Enthalpy ChangesSofia YBelum ada peringkat
- Organic Chemistry - Chapter 15 Benzene & Aromatic CompoundsDokumen9 halamanOrganic Chemistry - Chapter 15 Benzene & Aromatic CompoundsSairille ManejaBelum ada peringkat
- An Organic Chemistry Might Study Organic Reactions, The Structure and Properties of Organic Molecules, Polymers, Drugs, or FuelsDokumen1 halamanAn Organic Chemistry Might Study Organic Reactions, The Structure and Properties of Organic Molecules, Polymers, Drugs, or FuelsRyan Jerico PamplonaBelum ada peringkat
- Water Carbon Nitrogen: Presented by Aditya KumarDokumen16 halamanWater Carbon Nitrogen: Presented by Aditya KumarAditya kumarBelum ada peringkat
- Org Chem Sem 3 Paper 2Dokumen15 halamanOrg Chem Sem 3 Paper 2Rohit DeshmukhBelum ada peringkat
- C 1141 - 01 QzexndeDokumen4 halamanC 1141 - 01 QzexndeEnrique Alejandro Ovando100% (1)
- Organic ChemistryDokumen13 halamanOrganic ChemistryErroel Rodel SaquilabonBelum ada peringkat
- Heme SynthesisDokumen32 halamanHeme SynthesisHari PrasathBelum ada peringkat
- SterioisomerismDokumen43 halamanSterioisomerismCistus AbenBelum ada peringkat
- Resonance and Mesomeric EffectDokumen13 halamanResonance and Mesomeric Effectapoorva singhBelum ada peringkat
- Electron Delocalization and ResonanceDokumen9 halamanElectron Delocalization and ResonanceMariana LizethBelum ada peringkat
- Reactions of AlkanesDokumen1 halamanReactions of AlkanesnofacejackBelum ada peringkat
- Aldehydes Ketones HaccDokumen66 halamanAldehydes Ketones HaccRammohan VaidyanathanBelum ada peringkat
- Surface TensionDokumen28 halamanSurface Tensionpurvesh23184Belum ada peringkat
- Interhalogen Compounds Structure and PropertiesDokumen14 halamanInterhalogen Compounds Structure and PropertiesAndriati RahayuBelum ada peringkat
- The S-Block Elements: The Elements in Which Last Electron Enters The S-Subshell Are Called As S-Block ElementsDokumen36 halamanThe S-Block Elements: The Elements in Which Last Electron Enters The S-Subshell Are Called As S-Block ElementstheDarknight2050 (Karthik Sunil)Belum ada peringkat
- Intro To Organic ChemDokumen91 halamanIntro To Organic ChemMiguel Marquez GelacioBelum ada peringkat
- Mesomeric EffectDokumen21 halamanMesomeric EffectAwais Arshad100% (2)
- Inductive and Resonance EffectDokumen20 halamanInductive and Resonance EffectNehalPatelBelum ada peringkat
- A Brief History of ChemistryDokumen27 halamanA Brief History of ChemistryRaymond BeligoloBelum ada peringkat
- Resonance and Induction Tutorial: Jack DeruiterDokumen19 halamanResonance and Induction Tutorial: Jack DeruiterDanish AhmedBelum ada peringkat
- Reaction of Alkenes and Alkynes For StudentsDokumen53 halamanReaction of Alkenes and Alkynes For StudentsGlen MangaliBelum ada peringkat
- Multi-Step Organic SynthesisDokumen6 halamanMulti-Step Organic SynthesisPhạm Thị Thùy NhiênBelum ada peringkat
- Retrosynthetic Analysis PDFDokumen6 halamanRetrosynthetic Analysis PDFNoleBelum ada peringkat
- Course Title: Organic Chemistry-I Course Code: Chm-553, Chm-507 Semester: MSC 1, Bs 5Dokumen18 halamanCourse Title: Organic Chemistry-I Course Code: Chm-553, Chm-507 Semester: MSC 1, Bs 5Mian Naveed AhmedBelum ada peringkat
- 12 August Mock TestDokumen65 halaman12 August Mock Testapi-26674800Belum ada peringkat
- 11chemistry OMEGA PDFDokumen96 halaman11chemistry OMEGA PDFChirAgBelum ada peringkat
- Aromatic Compounds and Uses of BenzeneDokumen44 halamanAromatic Compounds and Uses of BenzenePaarijat DubeyBelum ada peringkat
- The Structure and Reactions of Aromatic CompoundsDokumen25 halamanThe Structure and Reactions of Aromatic CompoundsElizabeth Vivar100% (1)
- Comparing The SN1 and SN2 Reactions - Master Organic ChemistryDokumen5 halamanComparing The SN1 and SN2 Reactions - Master Organic Chemistryprince ranaBelum ada peringkat
- Organometallic CompoundsDokumen66 halamanOrganometallic CompoundsJon Ho100% (1)
- Ugi ReactionsDokumen36 halamanUgi ReactionsHarjinder Singh Bhatia100% (1)
- Cy 101 Uv-Vis and Ir NewDokumen66 halamanCy 101 Uv-Vis and Ir NewSomesh MohapatraBelum ada peringkat
- Organometallic CompoundsDokumen40 halamanOrganometallic CompoundsHalida SophiaBelum ada peringkat
- Chapter 16: Benzene - Electrophilic Aromatic Substitution: Chem231 Study Notes On McmurryDokumen20 halamanChapter 16: Benzene - Electrophilic Aromatic Substitution: Chem231 Study Notes On McmurrykjjkimkmkBelum ada peringkat
- Bioinorganic HandoutDokumen63 halamanBioinorganic HandoutAL__52Belum ada peringkat
- Redox RxnsDokumen30 halamanRedox RxnsJolaine ValloBelum ada peringkat
- Essential Organic Chemistry: Aromaticity: Reactions of Benzene and Substituted BenzenesDokumen75 halamanEssential Organic Chemistry: Aromaticity: Reactions of Benzene and Substituted BenzeneschurvaloooBelum ada peringkat
- Electrochemical CellsDokumen5 halamanElectrochemical Cellsyanyilu100% (1)
- Organic Chemistry 2Dokumen298 halamanOrganic Chemistry 2arielBelum ada peringkat
- First Effective Chemotherapeutic AgentsDokumen19 halamanFirst Effective Chemotherapeutic AgentsPraveen T MBelum ada peringkat
- Resonance StructuresDokumen4 halamanResonance StructuresSmyra100% (1)
- 01 Aromatic Hydrocarbons BenzeneDokumen116 halaman01 Aromatic Hydrocarbons BenzeneHamid Hussain HamidBelum ada peringkat
- Practical Organic Chemistry (I) : October 2017Dokumen44 halamanPractical Organic Chemistry (I) : October 2017NUR ALOMBelum ada peringkat
- Chapter 05 Wade 8thDokumen66 halamanChapter 05 Wade 8thanupamgupta112Belum ada peringkat
- Lecture 1501 - Introduction To Chemical EquilibriumDokumen15 halamanLecture 1501 - Introduction To Chemical Equilibriumapi-196433526Belum ada peringkat
- Physics Brochure PDFDokumen12 halamanPhysics Brochure PDFKatrina PanaliganBelum ada peringkat
- The Transition Metals, The Lanthanides and The AntinidesDokumen21 halamanThe Transition Metals, The Lanthanides and The AntinidesApril CruzBelum ada peringkat
- Organometallic ChemistryDokumen31 halamanOrganometallic ChemistrySadiaKhan100% (1)
- Organic Functional Group Analysis: Theory and DevelopmentDari EverandOrganic Functional Group Analysis: Theory and DevelopmentBelum ada peringkat
- Final Test Series For Neet-2022Dokumen21 halamanFinal Test Series For Neet-2022sneha sahaBelum ada peringkat
- Question Bank - 102 - 2013-14 (English)Dokumen2 halamanQuestion Bank - 102 - 2013-14 (English)RICHA VERMABelum ada peringkat
- Nuevo Solvente Asfalten Con Hse FavorableDokumen16 halamanNuevo Solvente Asfalten Con Hse FavorableHebert FerneynesBelum ada peringkat
- Chapter 8 - Ion Exchange PDFDokumen29 halamanChapter 8 - Ion Exchange PDFAli AimranBelum ada peringkat
- Grade 12 Acids and BasesDokumen32 halamanGrade 12 Acids and BasesvfdfdBelum ada peringkat
- AOD Chapter12 PDFDokumen4 halamanAOD Chapter12 PDFNguyễn Văn ThanhBelum ada peringkat
- Lett 2c03388Dokumen5 halamanLett 2c03388Vũ LưuBelum ada peringkat
- Biomolecules and Cells Notes SummaryDokumen81 halamanBiomolecules and Cells Notes SummaryLily RobinsonBelum ada peringkat
- Series 4100 Translucent Calendered Film: Applications & FeaturesDokumen2 halamanSeries 4100 Translucent Calendered Film: Applications & Featuresgerardo_medina_92Belum ada peringkat
- Life Cycle Analysis of Star Soap and Detergents FactoryDokumen15 halamanLife Cycle Analysis of Star Soap and Detergents Factorymarion tesfaBelum ada peringkat
- 0653 m19 QP 22 PDFDokumen16 halaman0653 m19 QP 22 PDFmaria paula peña narvaezBelum ada peringkat
- Water Supply and Treatment Engineering - 18CV46 - Notes-Module-2Dokumen39 halamanWater Supply and Treatment Engineering - 18CV46 - Notes-Module-2Laxmi MahanteshBelum ada peringkat
- Combined Science: Paper 0653/12 Multiple Choice (Core)Dokumen20 halamanCombined Science: Paper 0653/12 Multiple Choice (Core)equakeroatsBelum ada peringkat
- CarbohydratesDokumen31 halamanCarbohydratesMarlene GonsalvezBelum ada peringkat
- MCB 7200: Molecular BiologyDokumen27 halamanMCB 7200: Molecular BiologytasniyanBelum ada peringkat
- DNA Damage and RepairDokumen32 halamanDNA Damage and RepairNashmiya SaheerBelum ada peringkat
- Micrasil TDSDokumen2 halamanMicrasil TDSAmmarBelum ada peringkat
- Catalase Enzyme Lab ReportDokumen5 halamanCatalase Enzyme Lab ReportSaraBelum ada peringkat
- Utilization of Fly Ash For Sustainable Environment ManagementDokumen9 halamanUtilization of Fly Ash For Sustainable Environment ManagementEL Hassania EL HERRADIBelum ada peringkat
- GENIUS 2016 Finalist ListDokumen36 halamanGENIUS 2016 Finalist ListUmair MoinBelum ada peringkat
- A Simple Method For Determining Activation Energies of Organic Reactions From DSC CurvesDokumen5 halamanA Simple Method For Determining Activation Energies of Organic Reactions From DSC CurvesYahaya Umar BalarabeBelum ada peringkat
- Introduction to Textile FibresDokumen56 halamanIntroduction to Textile FibresMayank LaroiaBelum ada peringkat
- 1 Year Chemistry Practical Course (Ch110), 2017Dokumen29 halaman1 Year Chemistry Practical Course (Ch110), 2017Belal AhmedBelum ada peringkat
- Corrosion Control - Chemical Conversion Coatings On Aluminum AlloysDokumen5 halamanCorrosion Control - Chemical Conversion Coatings On Aluminum AlloysbenBelum ada peringkat
- Nwanonenyi, S.C, Madufor, I. C, Chukwujike, I.C, Arinze, V.CDokumen14 halamanNwanonenyi, S.C, Madufor, I. C, Chukwujike, I.C, Arinze, V.CIheoma Chukwujike NwuzorBelum ada peringkat
- Hydrogen Hydrides Properties UsesDokumen7 halamanHydrogen Hydrides Properties UsesNitishBelum ada peringkat
- 0620 w12 QP 13 PDFDokumen16 halaman0620 w12 QP 13 PDFNgoc Quang NguyenBelum ada peringkat
- Inject STC - Resin CatalystDokumen2 halamanInject STC - Resin CatalystRaju ShresthaBelum ada peringkat
- La Metalografia A ColorDokumen81 halamanLa Metalografia A ColorLENDBelum ada peringkat