2010 Lecture 19
Diunggah oleh
Des MshenguJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
2010 Lecture 19
Diunggah oleh
Des MshenguHak Cipta:
Format Tersedia
Computational Fluid Dynamics I
http://users.wpi.edu/~gretar/me612.html!
Computational Fluid Dynamics I
Examples:!
Multiphase Flow Modeling!
Grtar Tryggvason !
Spring 2010!
Spray drying!
Pollution control!
Pneumatic transport!
Slurry transport!
Fluidized beds!
Spray forming!
Plasma spray coating!
Abrasive water jet cutting!
Pulverized coal fired furnaces!
Solid propellant rockets!
Fire suppression and control!
Computational Fluid Dynamics I
Computational Fluid Dynamics I
Disperse flow!
Single component
Multicomponent!
Solid-liquid: Slurries, quicksand, sediment transport!
Single
phase
water flow
Nitrogen flow
air flow!
emulsions!
Solid-air: dust, fluidized bed, erosion!
Multiphase
Steam-water flow
Freon-Freon
vapor flow!
air-water flow!
slurry flow!
Computational Fluid Dynamics I
Flow in pipes!
Stratified!
Slugs!
Mixed!
Dispersed!
Liquid-air: sprays, rain!
Air-liquid: bubbly flows!
Computational Fluid Dynamics I
This figure shows schematically one
of several different configurations of a
circulating fluidized bed loop used in
engineering practice. The particles
flow downward through the aerated
standpipe, and enter the bottom of a
fast fluidized bed riser. The particles
are centrifugally separated from the
gas in a train of cyclones. In this
diagram, the particles separated in the
primary cyclone are returned to the
standpipe while the fate of the
particles removed from the secondary
cyclone is not shown. !
From: Computational Methods for
Multiphase Flow, Edited by
A.Prosperetti and G.Tryggvason!
Computational Fluid Dynamics I
Need model equations to predict flow rates, pressure
drop, slip velocities, and void fraction!
Mixture models: one averaged phase!
Two-fluid models: two interpenetrating continuum!
Computational Fluid Dynamics I
Euler/Euler approach!
All phases are treated as interpenetrating continuum!
The dispersed phase is averaged over each control volume!
Each phase is governed by similar conservation equations!
Modeling is needed for!
!interaction between the phases!
!turbulent dispersion of particles!
!collision of particles with walls!
A size distribution requires the solution of several sets of
conservation equations !
Numerical diffusion at phase boundaries may result in errors!
This approach is best suited for high volume fraction of the
dispersed phase!
Computational Fluid Dynamics I
Euler/Lagrange approach!
The fluid flow is found by solving the Reynolds-averaged
Navier-Stokes equations with a turbulence model.!
The dispersed phase is simulated by tracking a large number of
representative particles.!
A statistically reliable average behavior of the dispersed phase
requires a large number of particles!
The point particles must be much smaller than the grid spacing!
Modeling is needed for!
!collision of particles with walls!
!particle/particle collisions and agglomeration!
!droplet/bubble coalescence and breakup!
A high particle concentration may cause convergence problems!
Computational Fluid Dynamics I
Although commercial codes will let you model relatively
complex multiphase flows, it is really only in the limit of
dispersed and dilute flows where we can expect reasonable
accuracy!
To treat systems like this, the
two-fluid model is usually
used. The continuous phase
is almost always used in an
Eularian way where the
continuity, momentum, and
energy equations are solved
on a fixed grid. !
Computational Fluid Dynamics I
The void fraction p describes how much
of the region is occupied by phase p.!
Obviously,!
Similarly, the effective
density of phase p is!
= p p
=1
Computational Fluid Dynamics I
While the averaging is similar to turbulent flows,
here we must account for the different phases!
1 inside phase p
p =
0 otherwise
The void fraction is found by!
p =
1
V
p dv
Computational Fluid Dynamics I
Computational Fluid Dynamics I
Averages are found by!
The total mass of phase p in a control volume is!
1
p =
pV
p dv
Where the volume
V goes to zero in
some way!
1
pV
p p dv
And the mass conservation equation can be
averaged to yield!
The velocity is found by!
u p =
p p + ( p p u p ) = m p
t
u p p dv
The averages can also be interpreted as time or
ensemble averages!
Here!
=0
Since a mass that leaves one phase
must add to another phase!
Computational Fluid Dynamics I
Computational Fluid Dynamics I
The conservation of momentum equation becomes!
( p pu p ) + ( p pu pu p ) = ppp
t
+ ( p p D p ) + p p g + ( p p < uu >) + Fint
Reynolds
stresses!
interfacial
forces!
In addition to the Reynolds stresses, it is now necessary
to model the interfacial forces. The kinetic energy is
often neglected, even though the fluctuations are nonzero in laminar flow!
Computational Fluid Dynamics I
However, the dispersed phase is not all that continuous
and an other approach is to explicitly tract
(representative) particles by solving!
du
= Fp
dt
If the particles have no influence on the fluid: one way
coupling!
If the particles exert a force on the fluid: Two way
coupling!
If there is no mass transfer m=0 and F is the force that
one phase exerts on the other!
=0
In principle the conservation equations can be
solved for both the continuous and the dispersed
phase (Euler/Euler approach).!
Computational Fluid Dynamics I
Usually the force is written:!
Fp = kD (u u p ) + g
Drag
force!
D
+ Fother
Gravity!
buoyancy!
Other forces due
to added mass,
pressure, lift, etc!
Computational Fluid Dynamics I
For the drag:!
where!
Fp = k ( ur u p )
k=
and!
Computational Fluid Dynamics I
The force allows us to find the particle
velocity by integrating:!
(ur up )
3
CDr q
4
dr
du p
= Fp
dt
CD = CD (Re)
Find particle trajectories by solving!
dx p
= up
dt
is obtained from experimental correlations, such as!
24
CD =
(1+ 0.15Re 0.687 ) Re < 103
Re
For solid particles!
Re based on slip velocity!
Computational Fluid Dynamics I
Turbulent flow!
Usually a large number of particles is used to get a
well converged particle distribution!
Set particle velocity!
u p + u'
Computational Fluid Dynamics I
Random velocity fluctuations
from !
k p = u' u'
Notice that almost all the interactions (particles/flow)
particle/particle, particle/wall) are highly empirical!
Particles can
accumulate here!
This allows particles to cross streamlines
as they do in turbulent flow!
Computational Fluid Dynamics I
Similar approach can be taken for the temperature
and the size of a particle (heat and mass transfer)!
m pc p
dTp
= hA p (Tf Tp ) + p A p (T4 Tp4 )
dt
dm p
= m p
dt
Mass transfer due
to evaporation, for
example!
For dilute flows this does work reasonably well if
the initial or inlet conditions are knows!
Computational Fluid Dynamics I
Turbulent in the continuous phase!
Either ignore the contributions of the dispersed phase
when computing the flow, or use a k- model!
Solve for k and in the liquid and kp. Called k kp models.!
The k equation is!
Dk
= + < U Fp >
Dt
< U Fp >=
< u f (u f u p ) >= (< u f u f > < u f u p >)
This term can lead to both reduction and increase in the
turbulence in the liquid!
Computational Fluid Dynamics I
Computational Fluid Dynamics I
The full two-fluid model suffers from several
problems, in addition to uncertainties about the
various closure assumptions:!
Modeling of Laminar Flow
in a Vertical Channel!
The major one is that the full equations are ill-posed
and one cannot expect a fully converged solution
under grid refinement!
One possible way around this is to use the drift flux
approximation where the particle velocity is assumed
to be a given function of the local conditions.!
Computational Fluid Dynamics I
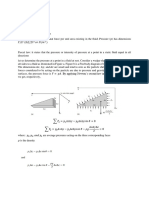
Bubbly flow in a vertical channel!
Need to know!
The bubble distribution!
The velocity profile and
the flow rate!
Computational Fluid Dynamics I
Comparison with a two-fluid model!
Simple two-fluid model for laminar multiphase flow
dp
3
+ g gy =
C D lU r U r
Bubble vertical momentum!
dy
8 Rb
dpl
2v l 3
Liquid vertical
(1 )
+ (1 ) l gy = (1 )l 2 +
C D lU r U r
momentum!
dy
x
8 Rb
Assume that the flow is
independent of y, so!
y
Flow! Gravity!
x
S.P. Antal, R.T. Lahey and J.E.
Flaherty. Intl. J. Multiphase Flow
17 (1991), 635-652.!
= 0 but!
y
pl
is given!
y
Computational Fluid Dynamics I
Comparison with a two-fluid model!
Comparison with experimental results. Graph from: S.P. Antal, R.T. Lahey and
J.E. Flaherty. Intl. J. Multiphase Flow 17 (1991), 635-652.!
U r
x 5
(1 ) = CLU r
ul
R U 2
Cw1 + Cw 2 b r
x
s Rb
Wall repulsion!
(away from wall or zero)!
Lift!
CD =
24
(1+ 0.1Re0.75)
Re
( x ) =
1
L
dx,
0
Bubble
horizontal
momentum!
Re =
2Rb l Ur
ul (0) = ul ( H ) = 0
m =
l
1
dpg dpl dp
=
=
dy
dy dy
Computational Fluid Dynamics I
Modeling of multiphase flows is still a very immature area.
Interpret the results with care!!
For more information about computing multiphase flow, see:!
Computational Fluid Dynamics I
http://users.wpi.edu/~gretar/me612.html!
Computational Fluid Dynamics I
Examples!
Gas combustion!
!Gasoline engines, !
!gas burners!
Combustion Modeling!
Spray combustion!
!Jet engines, !
!diesel engines!
Combustion of solids!
!coal, !
!wood, !
!polymers!
Grtar Tryggvason !
Spring 2010!
Computational Fluid Dynamics I
Diffusion flames!
Most burners, candle!
Flame stays at the
boundary between the
fuel and the oxidizer!
Computational Fluid Dynamics I
Premixed flames!
Some burners!
Hazards!
IC engines!
Diffusion Flames!
The flame separates
unburned and burned
mixture of fuel and oxidizer!
Computational Fluid Dynamics I
Diffusion flames!
Fuel, CH4
(methane), for
example!
Oxidizer, O2!
Computational Fluid Dynamics I
Diffusion flames!
The thickness of the
flame depends on
the ratio of the
reaction rates to the
diffusion times
(Dahmkler number)!
Slow Reaction!
Interim
species!
O2!
CH4!
Fast Reaction!
CH4!
Interim
species!
O2!
The thickness of the
flame depends on
the ratio of the
reaction rates to the
diffusion times
(Dahmkler number)!
Computational Fluid Dynamics I
Computational Fluid Dynamics I
In turbulent flows the
flame sheet usually
folds in complex ways!
In general, the combustion is a very complex
process involving O(100) species and reaction rates.
These are reasonably well known for a number of
reaction, but still an open research field in general.!
P = 4,
!inj =
0.1
P = 4,
!inj =
0.3
P = 4,
!inj =
0.5
Computational Fluid Dynamics I
For the full problem it is necessary to track the mass
fraction of every species along with the momentum,
mass, and energy conservation equations!
mi + umi = J + R
t
c i + uc i = R
t
R = T n AE E k / RT c i
i
Arrenius reaction rates!
Only a handful of computations of the full problem
have been done so far!
Computational Fluid Dynamics I
For a simple one step reaction it can be shown that it is
sufficient to follow one variable, called the mixture fraction!
f =
mf
m f + mo
This is a conserved variable that is simply advected with
the flow!
( f ) + ( fu) = Df
t
Given f, we can find each species fraction by!
i = i ( f )
For combustion of natural gas, GRIMech, for
example!
Computational Fluid Dynamics I
For realistic situations, the problem must be simplified!
Diffusion Flames!
Use one-step (overall) reaction rates !
(if Da -> , Burke Shuman limits)!
Use a reduced set of chemical reactions!
Can work very well for laminar flames. For
turbulent flows the reaction rates have to be
modified to account for stretching and folding of
flame sheets.!
Computational Fluid Dynamics I
The function can be constructed either assuming infinitely
fast reactions (flame sheets) or equilibrium. The library is
constructed once only. If the system is non-adiabatic, is a
function of the enthalpy also!
The chemical system must be a diffusion flame and
consists of a fuel and an oxidizer!
The Lewis number must be unity (all diffusion
coefficients equal)!
Only one fuel type (can be a mixture)!
Only one type of oxidizer (can be a mixture)!
Incompressible turbulent flow!
Computational Fluid Dynamics I
For turbulent flows we solve for both f and the fluctuations of
f and use those to determine the species fraction!
( f ')
pdf!
Computational Fluid Dynamics I
Constructing the pdf from measurements!
pdf!
f!
Determines how f
is distributed and
therefore how the
species are
distributed!
smaller!
f!
In the actual code a 2D look up table is first
constructed, given the shape of the pdf!
2
= f , ( f ')
t!
Computational Fluid Dynamics I
f!
Computational Fluid Dynamics I
Premixed flames!
The flame speed is found experimentally
or by detailed computations !
G<0!
Premixed Flames!
G + u G = U f G
t
Uf
G<0!
Flame is marked
by G=0!
Motion
due to
fluid
flow!
Motion
due to
burning!
For turbulent flow the flame speed is different from
laminar flow due to wrinkling!
Computational Fluid Dynamics I
Other combustion models!
Droplets!
Solid particles!
Evaporation!
Gasification!
Burning
(usually)!
Burning!
Computational Fluid Dynamics I
As for multiphase flows, many issues are still unresolved in
modeling of combustion and these models should be
applied with care!
Anda mungkin juga menyukai
- Extraction of Rare Earths From Iron-Rich Rare Earth DepositsDokumen9 halamanExtraction of Rare Earths From Iron-Rich Rare Earth DepositsDes MshenguBelum ada peringkat
- 1 s2.0 S0038092X16304169 MainDokumen19 halaman1 s2.0 S0038092X16304169 MainDes MshenguBelum ada peringkat
- 1 s2.0 S0038092X15001206 MainDokumen9 halaman1 s2.0 S0038092X15001206 MainDes MshenguBelum ada peringkat
- Well stirredScrubberResultsatatmosphericpressure Univ - Ofnewcastle 1 PDFDokumen62 halamanWell stirredScrubberResultsatatmosphericpressure Univ - Ofnewcastle 1 PDFDes MshenguBelum ada peringkat
- Nuclear Engineering and TechnologyDokumen8 halamanNuclear Engineering and TechnologyDes MshenguBelum ada peringkat
- Yehn 022Dokumen209 halamanYehn 022MuzzamilBelum ada peringkat
- Sasol Secunda Waste Disposal SiteDokumen29 halamanSasol Secunda Waste Disposal SiteDes MshenguBelum ada peringkat
- Rare Earth Cracking and Leaching ReviewDokumen14 halamanRare Earth Cracking and Leaching ReviewDes MshenguBelum ada peringkat
- 159 Yeboah 2015Dokumen13 halaman159 Yeboah 2015Des MshenguBelum ada peringkat
- High-Temperature Thermal Plasma Treatment of Monazite Followed by Aqueous DigestionDokumen6 halamanHigh-Temperature Thermal Plasma Treatment of Monazite Followed by Aqueous DigestionDes MshenguBelum ada peringkat
- Evolução Dos ShampoosDokumen22 halamanEvolução Dos Shampoosjones32Belum ada peringkat
- Okoloetal.2015 Chemical StructuralpropertiesofSouthAfricanbituminouscoalsDokumen15 halamanOkoloetal.2015 Chemical StructuralpropertiesofSouthAfricanbituminouscoalsDes MshenguBelum ada peringkat
- PP Chem Sodium SulphateDokumen9 halamanPP Chem Sodium SulphateDes MshenguBelum ada peringkat
- Process Development To Recover Rare Earth MetalsDokumen14 halamanProcess Development To Recover Rare Earth MetalsKumar SanjayBelum ada peringkat
- Aarifnwu PCC 2016Dokumen25 halamanAarifnwu PCC 2016Des MshenguBelum ada peringkat
- Aarifnwu PCC 2016Dokumen25 halamanAarifnwu PCC 2016Des MshenguBelum ada peringkat
- A13v30n3 PDFDokumen12 halamanA13v30n3 PDFDes MshenguBelum ada peringkat
- Ramsaroop Bhaveshnee R 2013Dokumen166 halamanRamsaroop Bhaveshnee R 2013Des MshenguBelum ada peringkat
- Thiosulfate As An Oxidation Inhibitor in Flue Gas Desulfurization Processes A Review of R D ResultsDokumen10 halamanThiosulfate As An Oxidation Inhibitor in Flue Gas Desulfurization Processes A Review of R D ResultsDes MshenguBelum ada peringkat
- Paramest PDFDokumen37 halamanParamest PDFMouhamadou DiabyBelum ada peringkat
- Abdullah, 2010Dokumen26 halamanAbdullah, 2010Des MshenguBelum ada peringkat
- Rotor-Stator Spinning Disc ReactorDokumen145 halamanRotor-Stator Spinning Disc ReactorDes MshenguBelum ada peringkat
- PdematDokumen29 halamanPdematNandNBelum ada peringkat
- 5.3.4. Effect of Other Variables On Gas-Liquid Mass TransferDokumen2 halaman5.3.4. Effect of Other Variables On Gas-Liquid Mass TransferDes MshenguBelum ada peringkat
- Materials and DesignDokumen20 halamanMaterials and DesignDes MshenguBelum ada peringkat
- Abraham, 2015Dokumen15 halamanAbraham, 2015Des MshenguBelum ada peringkat
- Wang Et Al., 2010Dokumen6 halamanWang Et Al., 2010Des MshenguBelum ada peringkat
- Borgwardt, 1970Dokumen5 halamanBorgwardt, 1970Des MshenguBelum ada peringkat
- Solving Differential EquationsDokumen2 halamanSolving Differential EquationsDes MshenguBelum ada peringkat
- Solution Methods For The Incompressible Navier-Stokes EquationsDokumen86 halamanSolution Methods For The Incompressible Navier-Stokes EquationsTaewoo KimBelum ada peringkat
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDari EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifePenilaian: 4 dari 5 bintang4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingDari EverandThe Little Book of Hygge: Danish Secrets to Happy LivingPenilaian: 3.5 dari 5 bintang3.5/5 (399)
- Shoe Dog: A Memoir by the Creator of NikeDari EverandShoe Dog: A Memoir by the Creator of NikePenilaian: 4.5 dari 5 bintang4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDari EverandNever Split the Difference: Negotiating As If Your Life Depended On ItPenilaian: 4.5 dari 5 bintang4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDari EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RacePenilaian: 4 dari 5 bintang4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Dari EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Penilaian: 4 dari 5 bintang4/5 (98)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDari EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryPenilaian: 3.5 dari 5 bintang3.5/5 (231)
- Grit: The Power of Passion and PerseveranceDari EverandGrit: The Power of Passion and PerseverancePenilaian: 4 dari 5 bintang4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDari EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FuturePenilaian: 4.5 dari 5 bintang4.5/5 (474)
- On Fire: The (Burning) Case for a Green New DealDari EverandOn Fire: The (Burning) Case for a Green New DealPenilaian: 4 dari 5 bintang4/5 (73)
- Team of Rivals: The Political Genius of Abraham LincolnDari EverandTeam of Rivals: The Political Genius of Abraham LincolnPenilaian: 4.5 dari 5 bintang4.5/5 (234)
- The Emperor of All Maladies: A Biography of CancerDari EverandThe Emperor of All Maladies: A Biography of CancerPenilaian: 4.5 dari 5 bintang4.5/5 (271)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDari EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersPenilaian: 4.5 dari 5 bintang4.5/5 (344)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDari EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaPenilaian: 4.5 dari 5 bintang4.5/5 (266)
- Rise of ISIS: A Threat We Can't IgnoreDari EverandRise of ISIS: A Threat We Can't IgnorePenilaian: 3.5 dari 5 bintang3.5/5 (137)
- The Unwinding: An Inner History of the New AmericaDari EverandThe Unwinding: An Inner History of the New AmericaPenilaian: 4 dari 5 bintang4/5 (45)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDari EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyPenilaian: 3.5 dari 5 bintang3.5/5 (2219)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDari EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You ArePenilaian: 4 dari 5 bintang4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Dari EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Penilaian: 4.5 dari 5 bintang4.5/5 (120)
- Her Body and Other Parties: StoriesDari EverandHer Body and Other Parties: StoriesPenilaian: 4 dari 5 bintang4/5 (821)
- 00 - Orientation LessonDokumen32 halaman00 - Orientation LessonRekha ShahBelum ada peringkat
- Special Report 205 Chemical Weekly 1 4-B PDFDokumen7 halamanSpecial Report 205 Chemical Weekly 1 4-B PDFsifrmoroBelum ada peringkat
- Syllabus For TR Supervisors Quality AssurnaceDokumen67 halamanSyllabus For TR Supervisors Quality Assurnaceadnanlibra194Belum ada peringkat
- To 1 SMPN 210 JKT Tahun 2016-2017Dokumen11 halamanTo 1 SMPN 210 JKT Tahun 2016-2017Sugeng Supoyo Pawit DigdotriratnaBelum ada peringkat
- Fiitjee: All India Internal Test SeriesDokumen16 halamanFiitjee: All India Internal Test SeriesAditya KashyapBelum ada peringkat
- Villamor Test - 2 PDFDokumen18 halamanVillamor Test - 2 PDFLocrian IonianBelum ada peringkat
- 2021 SC Hurricane GuideDokumen16 halaman2021 SC Hurricane GuideWMBF News100% (1)
- Public List Programs by Specialty 460Dokumen9 halamanPublic List Programs by Specialty 460Enta SyngBelum ada peringkat
- Company ProfileDokumen10 halamanCompany Profile1anupma0Belum ada peringkat
- To Study The Open Circuit/Core Losses of Single Phase TransformerDokumen5 halamanTo Study The Open Circuit/Core Losses of Single Phase TransformerTanzeel UR RehmanBelum ada peringkat
- Unit 1 Purva Swadhyaya QuestionsDokumen4 halamanUnit 1 Purva Swadhyaya QuestionsSurrender to Krishna AlwaysBelum ada peringkat
- Sizing PSV Broady Valvitalia-R4Dokumen18 halamanSizing PSV Broady Valvitalia-R4Anggun RushBelum ada peringkat
- 2) Inequality GraphsDokumen14 halaman2) Inequality Graphsmanobilli30Belum ada peringkat
- BrinjalDokumen2 halamanBrinjalGajananBelum ada peringkat
- An Overview of Muga and Eri Silk Industry in The North Eastern Region of India: An Empirical Study Dr. Jatindra Nath SaikiaDokumen11 halamanAn Overview of Muga and Eri Silk Industry in The North Eastern Region of India: An Empirical Study Dr. Jatindra Nath Saikiajatin_saikiaBelum ada peringkat
- Jyotish - A Manual of Hindu Astrology - B.v.raman - 1992Dokumen149 halamanJyotish - A Manual of Hindu Astrology - B.v.raman - 1992Ram RamBelum ada peringkat
- KFS Ut U3Dokumen5 halamanKFS Ut U3Ana DuarteBelum ada peringkat
- Chennai Public School: CH 3 Coordinate Geometry Multiple AssessmentDokumen3 halamanChennai Public School: CH 3 Coordinate Geometry Multiple Assessment11F10 RUCHITA MAARANBelum ada peringkat
- Steven Churchill, Jack Reynolds (Eds.) - Jean-Paul Sartre - Key Concepts-Routledge (2014)Dokumen257 halamanSteven Churchill, Jack Reynolds (Eds.) - Jean-Paul Sartre - Key Concepts-Routledge (2014)Leonardo Grana100% (3)
- Mathematics JEE Main Chapter Wise DPPDokumen513 halamanMathematics JEE Main Chapter Wise DPPMurali100% (1)
- DPS Nashik Class 12 Science SyllabusDokumen18 halamanDPS Nashik Class 12 Science SyllabusSayali Morwal-KumawatBelum ada peringkat
- Rev Quiz All PDFDokumen46 halamanRev Quiz All PDFNota Belz82% (17)
- Table Napkin Folding TechniquesDokumen6 halamanTable Napkin Folding TechniquesMiguel BaricuatroBelum ada peringkat
- F 25 Sensor Family Miniature Sensors for Precision DetectionDokumen34 halamanF 25 Sensor Family Miniature Sensors for Precision DetectionAlex GarduñoBelum ada peringkat
- Wood Turning NotesDokumen36 halamanWood Turning Notestheodrosyo0% (1)
- Chapter 2Dokumen21 halamanChapter 2mlyth1668Belum ada peringkat
- Light Source OP950Dokumen262 halamanLight Source OP950samiBelum ada peringkat
- Fluosilic Acid MsdsDokumen5 halamanFluosilic Acid MsdsVuglaShemmanBelum ada peringkat
- Structural Modelling and Analysis using BIM ToolsDokumen137 halamanStructural Modelling and Analysis using BIM ToolsManuel Solís López100% (1)
- India's Power Industry History and FutureDokumen11 halamanIndia's Power Industry History and FutureMinesh Chand MeenaBelum ada peringkat