Dew Bubble
Diunggah oleh
Jorge HuertaHak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Dew Bubble
Diunggah oleh
Jorge HuertaHak Cipta:
Format Tersedia
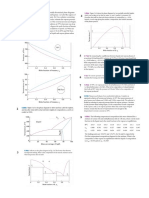
Calculas las composiciones de vapor y liquido y graficas los diagramas de fase para
1) propano
2) n-butano
a)
Temperatura constante de 240K
b)
A presin constante de 1 bar
Parmetros de Antoine
A
B
C
P=bar
log()=/(+
propano
n-butano
propano
n-butano
3.98292
3.85002
819.296
909.65
-24.417
-36.146
T=K
Presiones de vapor
1.522460838 bar
0.244206822 bar
T
240 K
x1
P
y1
0
0.244206822
0
0.05
0.308119523 0.24705686
0.1
0.372032224 0.409228217
0.15
0.435944925 0.523848571
0.2
0.499857625 0.609157792
0.25
0.563770326 0.675124588
0.3
0.627683027 0.727657483
0.35
0.691595728 0.770480892
0.4
0.755508429 0.806058956
0.45
0.819421129 0.836087028
0.5
0.88333383 0.861769801
0.55
0.947246531 0.883986833
0.6
1.011159232 0.903395305
0.65
1.075071932 0.920496122
0.7
1.138984633 0.935677757
0.75
1.202897334 0.949246121
0.8
1.266810035 0.961445392
0.85
1.330722735 0.972472836
0.9
1.394635436 0.982489558
0.95
1.458548137 0.991628428
1
1.522460838
1
T
1.6
1.4
1.2
1
P
0.8
0.6
0.4
0.2
0
0
c)
0.1
cuando:
y1
x1=
P
0.2
0.3
0.4
gramas de fase para
c)
d)
Calcular la presin de roco para mezcla equimolar a T=240K
Calcular la temperatura de roco para mezcla equimolar a P=1 bar
log()=/(+)
Para calcular T, usar T-x-y.ees
=11+(11)2
1=11/
T=240K
burbuja
X
Y
roco
0.2
0.3
0.4
0.5
0.6
0.7
x,y
0.5
0.1382
0.4209 bar
calculado con EES
dew_P.ees
0.8
0.9
1.1
x1
0
0.05
0.1
0.15
0.2
0.25
0.3
0.35
0.4
0.45
0.5
0.55
0.6
0.65
0.7
0.75
0.8
0.85
0.9
0.95
1
1=.5=11/(11+(11)
calcular T, usar T-x-y.ees
P
T
272.4
267.7
263.6
260
256.8
253.9
251.3
249
246.8
244.8
243
241.3
239.7
238.3
236.9
235.6
234.4
233.2
232.1
231.1
230.1
1=.5=11/
1 bar
Pvap1
4.776179321
4.123381684
3.610156715
3.200234428
2.866104689
2.586653946
2.3541021
2.161938534
1.989543753
1.842125768
1.716743307
1.604447936
1.504008372
1.420168022
1.339988114
1.268718583
1.205576079
1.144904039
1.091401861
1.044474404
0.999137636
y1
0
0.206169084
0.361015671
0.480035164
0.573220938
0.646663487
0.70623063
0.756678487
0.795817501
0.828956596
0.858371654
0.882446365
0.902405023
0.923109214
0.93799168
0.951538937
0.964460863
0.973168433
0.982261675
0.992250684
0.999137636
P=1 b
280
270
260
250
240
230
220
210
200
burbuja
x,y
d)
cuando:
y1
x1
T
calculado con EES
1/(11+(11)2)
de aqu se saca x1
para luego:
11/
se despeja P
P=1 bar
roco
X
Y
x,y
0.5
0.1598
259.3 K
ulado con EES
dew_T.ees
Anda mungkin juga menyukai
- Cationes AnionesDokumen2 halamanCationes AnionesJorge HuertaBelum ada peringkat
- El-DíaDokumen4 halamanEl-DíaJorge HuertaBelum ada peringkat
- Primer Ejercicio NomenclaturaDokumen1 halamanPrimer Ejercicio NomenclaturaJorge HuertaBelum ada peringkat
- Comparative Between Brasil Vs Mexico in ScavengersDokumen28 halamanComparative Between Brasil Vs Mexico in ScavengersJorge HuertaBelum ada peringkat
- 1er ParcialDokumen1 halaman1er ParcialJorge HuertaBelum ada peringkat
- Extra 2do Parcial (Química)Dokumen1 halamanExtra 2do Parcial (Química)Jorge HuertaBelum ada peringkat
- ExtraDokumen1 halamanExtraJorge HuertaBelum ada peringkat
- Fertilizantes Uncle AlbertDokumen2 halamanFertilizantes Uncle AlbertJorge HuertaBelum ada peringkat
- Resumen de Articulo CHADokumen4 halamanResumen de Articulo CHAJorge HuertaBelum ada peringkat
- Extra 3er Parcial (Química) - Jorge HuertaDokumen1 halamanExtra 3er Parcial (Química) - Jorge HuertaJorge HuertaBelum ada peringkat
- 3er Parcial Química (Extra) ContestadoDokumen2 halaman3er Parcial Química (Extra) ContestadoJorge HuertaBelum ada peringkat
- Problemas de Tarea Fisicoquímica 2o ParcialDokumen1 halamanProblemas de Tarea Fisicoquímica 2o ParcialJorge HuertaBelum ada peringkat
- Extra 3er Parcial (Química) - Jorge HuertaDokumen1 halamanExtra 3er Parcial (Química) - Jorge HuertaJorge HuertaBelum ada peringkat
- Problema ClaseDokumen3 halamanProblema ClaseJorge HuertaBelum ada peringkat
- Segundo ParcialDokumen1 halamanSegundo ParcialJorge HuertaBelum ada peringkat
- Calculador D RDokumen2 halamanCalculador D RJorge HuertaBelum ada peringkat
- Temario Química ExplicadoDokumen1 halamanTemario Química ExplicadoJorge HuertaBelum ada peringkat
- Cónicas 1o Bachillerato: Circunferencias, elipses, hipérbolas y parábolasDokumen64 halamanCónicas 1o Bachillerato: Circunferencias, elipses, hipérbolas y parábolasYola FernandezBelum ada peringkat
- Guía pago derechos pasaporte SREDokumen1 halamanGuía pago derechos pasaporte SREManuel RamosBelum ada peringkat
- HW#2 Cinética AvanzadaDokumen6 halamanHW#2 Cinética AvanzadaJorge HuertaBelum ada peringkat
- Disoluciones Ionicas Problemas ResueltosDokumen7 halamanDisoluciones Ionicas Problemas ResueltosJoab Li Ac100% (1)
- Atractor de LorenzDokumen2 halamanAtractor de LorenzJorge HuertaBelum ada peringkat
- MAv 14B-T09 PDFDokumen2 halamanMAv 14B-T09 PDFJorge HuertaBelum ada peringkat
- MAv 14B-T08 PDFDokumen2 halamanMAv 14B-T08 PDFJorge HuertaBelum ada peringkat
- MAv 14B-T07 PDFDokumen2 halamanMAv 14B-T07 PDFJorge HuertaBelum ada peringkat
- MM4Dokumen2 halamanMM4Jorge HuertaBelum ada peringkat
- Tarea 9.5 COMSOLDokumen4 halamanTarea 9.5 COMSOLJorge HuertaBelum ada peringkat
- Art 08Dokumen8 halamanArt 08yhormanjustoBelum ada peringkat