Annealing Temp
Diunggah oleh
Baranigha CjDeskripsi Asli:
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Annealing Temp
Diunggah oleh
Baranigha CjHak Cipta:
Format Tersedia
Materials Science in Semiconductor Processing 24 (2014) 193199
Contents lists available at ScienceDirect
Materials Science in Semiconductor Processing
journal homepage: www.elsevier.com/locate/mssp
Band gap reduction of titania thin films using
graphene nanosheets
Haroon Mahmood n, Amir Habib, Mohammad Mujahid, Muhammad Tanveer,
Sofia Javed, Asif Jamil
School of Chemical and Materials Engineering, National University of Sciences and Technology, Islamabad, Pakistan
a r t i c l e i n f o
abstract
Available online 5 April 2014
Titania possesses a band gap of ! 3.2 eV, whereas the work function of graphene ! 4.5 eV
exists near the lowest unoccupied molecular orbital (LUMO) of titania. The photoexcitation of
electrons in titania (TiO2) is possible only under the ultraviolet radiation. If such conversion is
required in visible light range, band gap has to be lowered below 3.1 eV. This research work
hence aims at lowering the band gap of titania thin films by incorporation of graphene
nanosheets. Graphene was synthesized using modified Hummer's method. Oxidation of
graphite was confirmed by X-ray diffraction (XRD) revealing a peak at 11.81 with interplanar
distance of 7.5 and energy dispersive X-ray spectroscopy (EDS) analysis. Atomic force
microscopy (AFM) analysis revealed exfoliation of single layer graphene oxide nanosheets
having thickness of 0.61.0 nm. Fourier transform infrared spectroscopy (FT-IR) analysis
confirmed the reduction of graphene oxide by revealing removal of carbonyl group (CQO).
Titania sol was obtained using hydrolysis and condensation of titanium tetraisopropoxide. The
TiO2graphene composite was prepared by liquid phase dispersion of graphene oxide into
titania sol and reduction of deposited films was carried out by exposing hydrazine hydrate
vapors. In the second method, nanoparticles of reduced graphene oxide were dispersed in
titania sol; thin films were deposited on indiumtin oxide (ITO) coated glass using spin coating
and subsequently heat treated. A scanning electron microscope (SEM) was used to reveal
dispersion of graphene nanosheets among homogenously distributed titania nanoparticles of
uniform size distribution (! 3050 nm). Surface roughness of about 16 nm was observed by
AFM topographic images. The highest occupied molecular orbital (HOMO) and LUMO levels
were calculated by cyclic voltammetry and subsequent band gap was calculated which came
out to be as low as 2.9 eV. TiOC chemical bonding between titania and graphene sheets
resulted in enhanced electron transport in the obtained composite films.
& 2014 Elsevier Ltd. All rights reserved.
Keywords:
Titania thin film
Graphene
Band gap
Photovoltaics
1. Introduction
Titania, either in anatase or rutile phase, is considered to
be one of important materials for various applications on
account of its property of a photoexcitation phenomenon.
n
Correspondence to: SCME, National University of Sciences and
Technology (NUST), Sector H-12, Islamabad, Pakistan.
Tel.: 92 51 90855204.
E-mail address: haroon88@gmail.com (H. Mahmood).
http://dx.doi.org/10.1016/j.mssp.2014.03.038
1369-8001/& 2014 Elsevier Ltd. All rights reserved.
Among the applications, efficiency of photocatalytic devices
and photovoltaic solar cells could be enhanced if the band
gap of the films made up of titania may be narrowed down
[1,2]. Till now, several methods have been reported in
which doping of titania is done to reduce the band gap
and enhance the photocurrent by preventing photoexcitons recombination [35].
Carbon nanostructures have superior electrical properties and hence they can provide a medium for transport
of electrons in semiconductor thin films. Use of carbon
194
H. Mahmood et al. / Materials Science in Semiconductor Processing 24 (2014) 193199
nanotubes in TiO2, CdS and CdSe thin films has resulted in
increase in the efficiency of photocurrent in such structures
[68]. Graphene, a 2D carbon nanostructure, has high
electron mobility (exceeding 2000 cm2/V). Incorporation
of graphene has shown enhancement in the charge transfer
rate of electrons. This increased charge transfer rate
resulted in increase in the photocurrent by preventing
recombination of photo-generated electronhole pair
because of interactions of graphene [911].
Originally, titania possesses a band gap of ! 3.2 eV.
Lowering of band gap is essential for maximum harvesting
of sunlight and thus improvement of efficiency of devices
hence produced. Graphene, having a work function of
4.5 eV, is explored in this research work for its effect on
the band gap of titania thin films and also on photocurrent
generated in such structures. In the past, much of the
research have been performed for enhancement of photocatalytic activity of titania using graphene nanosheets,
such as preparing composites through thermal reaction
[5], solvothermal reaction [9], hydrothermal method
[10,12,13], solgel method [14] etc. These methods have
shown success in increment of photocatalytic activity by
the composite; however none of the work actually
reported the effect of graphene on the band gap of the
semiconductor material. This work was hence developed
to report the change in electrical properties of titania by
incorporation of graphene nanosheets in two forms, i.e.,
graphene oxide (GO) and reduced graphene oxide (RGO).
Solgel route was employed to obtain titania sol and
graphene was dispersed in the sol. Thin films were
obtained using spin-coating composite sol on the glass
and ITO substrates. After heat treatment of the films,
characterization and testing were performed.
2. Experimental work
Graphene oxide was synthesized from flake graphite
(average100 mesh (Z75% min), SigmaAldrich) by a
modified Hummers method reported elsewhere [15]. All
other chemicals were of analytical reagent grade and used
without further purification. Sonication of the resultant
solid was carried out in water to exfoliate graphene oxide
nanosheets. Chemical reduction of GO was carried out by
adding hydrazine hydrate (1.00 ml, 32.1 mmol) (Merck) in
the exfoliated GO dispersion and the solution was heated
at 100 1C for 24 h under a water-cooled condenser for 24 h.
The reduced GO slowly precipitated out as a black solid.
The subsequent nanoparticles were washed with water
and methanol and finally vacuum dried.
For thin film fabrication, titania sol was prepared by
hydrolysis and condensation of 97% titanium tetraisopropoxide (TTIP) (Aldrich) in the acidic mixtures of glacial
acetic acid and 37% hydrochloric acid. The solution, whose
composition is shown in Table 1, was heated at 60 1C
under vigorous stirring for 2 h [16].
Two approaches are used for composite thin film
processing. In one method, graphene oxide was mixed
with various concentrations (Table 2) in titania sol and
resulting spin coated composite films were exposed to an
atmosphere that has been saturated with vapor of hydrazine hydrate at 100 1C for 2 min. In another approach,
Table 1
Titania sol composition.
Chemical
Amount (ml)
TTIP
Glacial acetic acid
HCl
Water
10
5
1.4
83.6
Table 2
TitaniaGO composite sols.
Precursor
composite sol
Titania
sol (ml)
GO (5 mg ml $ 1)
(ml)
TGO-1
TGO-2
TGO-3
TGO-4
TGO-5
50
50
50
50
50
0
2
4
6
8
already reduced graphene oxide was mixed in various
amounts (Table 3) in titania sol and films were prepared
using a spin coating method. In both cases, after addition
of graphene precursor in titania sol, the solution was
sonicated for 30 min before film deposition. Films were
pre-annealed at 80 1C for 2 min followed by annealing at
450 1C for 2 h at 2 1C/min of heating rate.
3. Results and discussion
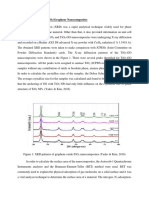
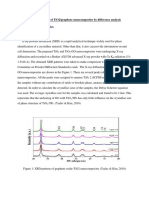
Fig. 1 shows a comparison of the X-ray diffractograms
of precursor graphite and graphite oxide. Pristine graphite
shows a high degree of crystallinity with a sharp diffraction peak at 226.471, that has an interplanar distance of
3.37 of (002) diffraction plane. However the degree of
crystallinity of graphite oxide is very low with a minor
broad-diffraction peak at 11.81, which is due to a conserved
graphene-like honeycomb lattice in graphite oxides [17].
Hence the shift of the diffraction peak position from 26.471
to 11.81 is due to increase in the interplanar distance from
3.37 to 7.5 . This is confirmed from Braggs equation
which inversely relates interplanar distance d to the
diffraction angle 2 [18]. The increase in interplanar
distance is due to trapping of oxygen atoms between the
graphene sheets in graphite structure [19].
Scanning electron microscope (SEM) images (Fig. 2a
and b) reveal exfoliation of graphite oxide into individual
(single or multi-layer) graphene sheets. A closely packed
tilting of graphene oxide platelets having an even contrast was detected on the substrate. The lateral dimensions
of the sheets ranged from several hundred nanometers to
several micrometers. Fig. 2c shows the thickness of exfoliated graphene oxide sheets investigated by atomic force
microscopy in a tapping mode. The graphene oxide height
profiles typically ranged between 0.6 nm and 1 nm which
is reported to be single layer graphene oxide sheet [20].
From the AFM topographical image (Fig. 3), graphene
nanosheets can be seen lying on glass slide. Overall, roughness (RMS) is about 0.5 nm indicating that the features are
very small in dimension along the z axis. The only factor
H. Mahmood et al. / Materials Science in Semiconductor Processing 24 (2014) 193199
contributing to the roughness values is intrinsic height of
carbon sheets and their lifting up against the base plane due
to crossovers of neighboring flakes. Line plots and their
Table 3
TitaniaRGO composite sols.
Precursor composite sol
Titania sol (ml)
RGO (mg)
TRGO-1
TRGO-2
TRGO-3
TRGO-4
50
50
50
50
0
5
10
15
Fig. 1. Comparison of XRD diffractographs of graphite and graphite oxide.
195
analysis support the above statement. Three lines have very
small difference in roughness values due to difference in
regions selected for the analysis. Red line has maximum
roughness of 0.5 nm and has maximum height feature of
about 3.4 nm. It is because the line has passed over two
flakes and got lifted upward due to many crossovers and
buckling. While some of the portion of green line indicates
that it has passed over single sheet or some bilayers as
evident from the line plot and corresponding z values. More
interesting part is the blue line which has only passed over a
monolayer graphene flake and has minimum roughness
value of 0.2 nm. The maximum height feature is observed
to be 1.12 nm relative to mean plane of overall image, while
the z plot of this line indicates that the height of flake is
even less than 1 nm.
Reduction of graphene oxide was confirmed using FT-IR
and Raman Spectroscopy. Fig. 4a shows FT-IR spectra
which indicates that the attached H2O molecules and
much of the oxide groups (Carbonyl CQO) of GO are
detached after chemical reduction by hydrazine hydrate.
Nevertheless, hydroxyl group (OH) and cyanide (CN)
remain on GO after chemical reductions due to poor
reactivity of hydrazine hydrate with these groups. Fig. 4b
shows Raman spectrum of the reduced GO which contains
both G and D bands (at 1589 and 1325 cm $ 1, respectively).
G band is related with stretching of bond of the sp2 carbon
pairs in both rings and chains, whereas the D band is
because of the breathing mode of aromatic rings with
dangling bonds in plane terminations. Furthermore, the
intensity ratio of the D band to the G band is usually
employed to determine the disorder degree of structure
for carbon. High D/G ratios obtained in Raman spectra of
GO indicate a high degree of exfoliation/disorder [21].
Fig. 2. (a and b) SEM images of single layer and multi-layer graphene oxide and (c) AFM image with height profile of graphene oxide nanosheets.
196
H. Mahmood et al. / Materials Science in Semiconductor Processing 24 (2014) 193199
Fig. 3. AFM topographical image of graphene nanosheets. (For interpretation of the references to color in this figure, the reader is referred to the web
version of this article.)
Fig. 4. (a) FTIR spectra of graphene oxide (GO) and reduced graphene oxide (RGO) and (b) Raman spectra of reduced graphene oxide.
Fig. 5a shows the scanning electron microscope (SEM)
images of 110125 nm thick graphenetitania composite
thin films. SEM images (Fig. 5b and c) reveal embedding of
titania nanoparticles of size ranging from 30 to 50 nm
embedded on graphene nanosheets. The film produced
has homogeneity of titania nanoparticles to appreciable
lengths. Some pinholes and agglomerated nanoparticles
have been observed on films as well.
AFM analysis confirms the dispersion of titania nanoparticles and embedding of titania nanoparticles on
H. Mahmood et al. / Materials Science in Semiconductor Processing 24 (2014) 193199
197
Fig. 5. (a) Thickness of graphenetitania composite thin film, and (b) low magnification image of graphene embedded in titania thin film. (c) High
magnification image of titania particles with size ranging from 30 nm to 50 nm.
graphene nanosheets (Fig. 6). Homogenous structure as
seen in SEM images is confirmed by AFM analysis of titania
nanoparticles with height profile ranging 1525 nm.
Using cyclic voltammetry (CV), HOMO/LUMO levels
were evaluated by the standard formula EHOMO $ Eonset
ox
$4:8 eV; ELUMO $Eonset
red $ 4:8 eV which gives us the
band gap information. The device structure for the CV
measurement consists of three electrodes: a working electrode which is the test sample, a counterelectrode made of a
platinum wire while saturated calomel as the reference
electrode. The range of current was set to auto while voltage
was varied from $2.5 V to 2.5 V. An assessment of the
obtained band gap and current of synthesized films is shown
in Fig. 7a and b.
The films containing reduced graphene oxide (RGO) in
titania matrix exhibited enhanced reduction of band gap
as compared to films developed using dispersion of
graphene oxide (GO) in titania sol. This can be related to
the higher conductivity of reduced graphene oxide as
compared to graphene oxide reduced after film fabrication. The lowest band gap found to be 2.90 eV for TRGO-4.
The overall trend for both types of composites films (GO
and RGO embedded films) is linear, apart from couple of
exceptions i.e. TGO-8 and TRGO-3. The distinctiveness
could be either because of defects in the thin films or
improper embedment of graphene nanosheets. Moreover
in both graphs (Fig. 7a and b), it is also clear that electron
mobility or current increases linearly with increasing
amount of graphene in the composite. This also verifies
the hypothesis that with increasing graphene amount, the
movement of electron becomes easier due to electron
capture as well as higher conductivity of graphene.
Fig. 8 displays a model diagram which shows the
synergistic effect of graphene on the electron transport
mechanism generated by titania nanoparticle. The work
function of graphene ranges between 4.2 and 4.5 eV [22].
After getting excited, the electron jumps from HOMO to
LUMO level within titania. Since LUMO (3.23.5 eV) of
titania is close to work function of graphene, the excited
electron in the LUMO level finds an intermediate energy
level and gets trapped hence causing charge separation.
This prevents recombination of electron and holes. Thus
effective electron transfer from lower band gap film
increases the current generation. This complements the
results obtained by cyclic voltammetry as shown above.
4. Conclusion
In this study, graphenetitania composite thin films were
synthesized using a spin coating technique. The dispersion of
graphene nanosheets in titania thin films, homogeneity of
films, HOMO/LUMO levels and band gap were evaluated. XRD
results indicated the oxidation of graphite while AFM analysis
revealed the exfoliation of graphite oxide into single or
multilayer graphene oxide nanosheets. Using SEM, the length
of graphene nanosheets ranged between several hundred
nanometers to several micrometers. Raman Spectroscopy
revealed higher D/G ratio suggesting high degree of exfoliation/disorder of graphene nanosheets. Reduction of graphene
oxide nanosheets was confirmed by FTIR displaying removal
of functional groups containing oxygen. Using SEM, graphenetitania composite thin films were analyzed which
revealed dispersion of graphene nanosheets. Single layers
graphene was fully embedded in the titania matrix while
198
H. Mahmood et al. / Materials Science in Semiconductor Processing 24 (2014) 193199
Fig. 6. AFM images of titania thin film with graphene nanosheets.
Fig. 7. Comparison of band gap of composite films: (a) titaniagraphene oxide and (b) titania-reduced graphene oxide.
H. Mahmood et al. / Materials Science in Semiconductor Processing 24 (2014) 193199
199
Fig. 8. Model energy diagram effect of graphene on electron transport.
multilayered graphene or irregular shaped graphene did not
fully embed in the film due to having feature size greater than
film thickness. Both SEM and AFM confirmed the titania
nanoparticles size in the range of 3050 nm. Cyclic voltammetry confirmed the effect of graphene on reduction of band
gap and enhancement of current by titania thin film. The
lowest band gap obtained was 2.9 eV of graphenetitania
composite thin film as compared to pure titania thin film
having 3.18 eV. The work function of graphene being closer to
LUMO energy level of titania and superior electrical properties
of graphene nanosheets resulted in the trapping of electrons
generated by titania nanoparticles which resulted in increased
efficiency of electrons generation and lowering of band gap.
References
[1]
[2]
[3]
[4]
A.L. Linsebigler, G. Lu, J.T. Yates Jr., Chem. Rev. 95 (1995) 735758.
B. ORegan, M. Gratzel, Nature 353 (1991) 737740.
X. Chen, S.S. Mao, Chem. Rev. 107 (2007) 28912959.
G. Liu, L. Wang, H.G. Yang, H.-M. Cheng, J. Mater. Chem. 20 (2010)
831843.
[5] Y. Zhang, C. Pan, J. Mater. Sci. 46 (2011) 26222626.
[6] A. Kongkanand, R.M. Dominguez, P.V. Kamat, Nano Lett. 7 (2007)
676680.
[7] F. Vietmeyer, B. Seger, P.V. Kamat, Adv. Mater. 19 (2007) 29352940.
[8] M. Wang, S. Kumar, A. Lee, N. Felorzabihi, L. Shen, F. Zhao, et al.,
J. Am. Chem. Soc. 130 (2008) 94819491.
[9] K. Zhou, Y. Zhu, X. Yang, X. Jiang, C. Li, New J. Chem. 35 (2011)
353359.
[10] H. Zhang, X. Lv, Y. Li, Y. Wang, J. Li, ACS Nano 4 (2009) 380386.
[11] Y.H. Ng, I.V. Lightcap, K. Goodwin, M. Matsumura, P.V. Kamat, J. Phys.
Chem. Lett. 1 (2010) 22222227.
[12] S.D. Perera, R.G. Mariano, K. Vu, N. Nour, O. Seitz, Y. Chabal, et al.,
ACS Catal. 2 (2012) 949956.
[13] Y. Zhang, Z.-R. Tang, X. Fu, Y.-J. Xu, ACS Nano 4 (2010) 73037314.
[14] X.-Y. Zhang, H.-P. Li, X.-L. Cui, Y. Lin, J. Mater. Chem. 20 (2010)
28012806.
[15] D.C. Marcano, D.V. Kosynkin, J.M. Berlin, A. Sinitskii, Z. Sun,
A. Slesarev, et al., ACS Nano 4 (2010) 48064814.
[16] W.S. Tung, W.A. Daoud, S.K. Leung, J. Colloid Interface Sci. 339 (2009)
424433.
[17] G. Wang, J. Yang, J. Park, X. Gou, B. Wang, H. Liu, et al., J. Phys. Chem.
C 112 (2008) 81928195.
[18] B.D. Cullity, S.R. Stock, Elements of X-ray Diffraction, Prentice-Hall,
Upper Saddle River; NJ, 2001.
[19] A. Lerf, H. He, M. Forster, J. Klinowski, J. Phys. Chem. B 102 (1998)
44774482.
[20] G. Eda, G. Fanchini, M. Chhowalla, Nat. Nanotechnol. 3 (2008)
270274.
[21] H. Wang, Y.H. Hu, Ind. Eng. Chem. Res. 50 (2011) 61326137.
[22] R. Czerw, B. Foley, D. Tekleab, A. Rubio, P. Ajayan, D. Carroll, Phys.
Rev. B 66 (2002) 033408.
Anda mungkin juga menyukai
- Surface Plasmon Enhanced, Coupled and Controlled FluorescenceDari EverandSurface Plasmon Enhanced, Coupled and Controlled FluorescenceBelum ada peringkat
- 3ftfbsdi "Sujdmf 4zouiftjt Boe $ibsbdufsj (Bujpo PG (Sbqifof 5ijo 'JMNT CZ $ifnjdbm 3fevdujpo PG &ygpmjbufe Boe Oufsdbmbufe (Sbqijuf 0yjefDokumen7 halaman3ftfbsdi "Sujdmf 4zouiftjt Boe $ibsbdufsj (Bujpo PG (Sbqifof 5ijo 'JMNT CZ $ifnjdbm 3fevdujpo PG &ygpmjbufe Boe Oufsdbmbufe (Sbqijuf 0yjefXana RodriguesBelum ada peringkat
- High-Quality Thin Graphene Films From Fast Electrochemical ExfoliationDokumen8 halamanHigh-Quality Thin Graphene Films From Fast Electrochemical ExfoliationelectroternalBelum ada peringkat
- LIS 2022 New 1-82-87Dokumen6 halamanLIS 2022 New 1-82-87Lppm Politeknik JambiBelum ada peringkat
- Sonochemical Synthesis of Tio2 Nanoparticles On Graphene For Use As PhotocatalystDokumen9 halamanSonochemical Synthesis of Tio2 Nanoparticles On Graphene For Use As PhotocatalystBryanda ReyesBelum ada peringkat
- GO-TiO2 2016Dokumen5 halamanGO-TiO2 2016Rafif QuthronadaBelum ada peringkat
- 1 s2.0 S000862231100621X MainDokumen9 halaman1 s2.0 S000862231100621X MainŞebnem İlarslanBelum ada peringkat
- Covalent Attaching Protein To Graphene Oxide Via Diimide-Activated AmidationDokumen4 halamanCovalent Attaching Protein To Graphene Oxide Via Diimide-Activated AmidationClaramaria RodriguezBelum ada peringkat
- Manikandan 2019Dokumen10 halamanManikandan 2019dilla zainudinBelum ada peringkat
- Graphene Effect On Efficiency of Tio - Based Dye Sensitized Solar Cells (DSSC)Dokumen8 halamanGraphene Effect On Efficiency of Tio - Based Dye Sensitized Solar Cells (DSSC)Ihsan PranataBelum ada peringkat
- 3221 - Deposition of TiO2 and Ag TiO2 Thin Films by The Polymeric Precursor MethodDokumen8 halaman3221 - Deposition of TiO2 and Ag TiO2 Thin Films by The Polymeric Precursor MethodAttila BartiBelum ada peringkat
- Saito 2019Dokumen11 halamanSaito 2019juanlopzBelum ada peringkat
- Nainani 2016Dokumen10 halamanNainani 2016Rafif QuthronadaBelum ada peringkat
- 1 s2.0 S000862231200111X MainDokumen10 halaman1 s2.0 S000862231200111X MainŞebnem İlarslanBelum ada peringkat
- 19moshgi Et. Al.Dokumen14 halaman19moshgi Et. Al.Divya BharathiBelum ada peringkat
- SSRN Id3844936Dokumen27 halamanSSRN Id3844936Rahul SahaBelum ada peringkat
- Photoluminescence and Photocatalytic Activity of Spin Coated Ag+ Doped Anatase TiO2 Thin FilmsDokumen14 halamanPhotoluminescence and Photocatalytic Activity of Spin Coated Ag+ Doped Anatase TiO2 Thin FilmsJasielRuizDesalesBelum ada peringkat
- Synthesis and Characterization of Tio /graphene Oxide NanocompositeDokumen7 halamanSynthesis and Characterization of Tio /graphene Oxide NanocompositeTHƯ NGUYỄN MINHBelum ada peringkat
- 5ungureanu PDFDokumen5 halaman5ungureanu PDFsuganyaBelum ada peringkat
- Materials Science in Semiconductor Processing: Rahul Kumar, Parag BhargavaDokumen6 halamanMaterials Science in Semiconductor Processing: Rahul Kumar, Parag BhargavaMohammad IrwanBelum ada peringkat
- 2012 Park - Graphene As Transparent Conducting Electrodes in OrganicDokumen8 halaman2012 Park - Graphene As Transparent Conducting Electrodes in Organicheri rustamajiBelum ada peringkat
- Graphene From Fundamental-From Fundamental To Future ApplicationsDokumen40 halamanGraphene From Fundamental-From Fundamental To Future ApplicationsMustaque Ali100% (2)
- Photo-Oxidation of Polypropylene-Graft-Maleic Anhydride - Clay NanocompositesDokumen10 halamanPhoto-Oxidation of Polypropylene-Graft-Maleic Anhydride - Clay NanocompositesDaniel GomesBelum ada peringkat
- Sahu2021 - Copper TiO2 GrapheneDokumen10 halamanSahu2021 - Copper TiO2 Grapheneenna hasnaBelum ada peringkat
- Functionalized Single Graphene Sheets Derived Form Splitting Graphite OxideDokumen5 halamanFunctionalized Single Graphene Sheets Derived Form Splitting Graphite OxidehumayunBelum ada peringkat
- Advance Material Draft Version 3.0Dokumen16 halamanAdvance Material Draft Version 3.0Yz LowBelum ada peringkat
- Materials Chemistry A: Journal ofDokumen7 halamanMaterials Chemistry A: Journal ofBhabani Sankar SwainBelum ada peringkat
- Restoring The Electrical Conductivity of Graphene Oxide Films by UV Light Induced Oxygen DesorptionDokumen11 halamanRestoring The Electrical Conductivity of Graphene Oxide Films by UV Light Induced Oxygen DesorptionFamiloni LayoBelum ada peringkat
- Optical and Electrical Properties of TiO2 Doped Fe2O3 Thin Film Prepared by Spray Paralysis TechniqueDokumen7 halamanOptical and Electrical Properties of TiO2 Doped Fe2O3 Thin Film Prepared by Spray Paralysis TechniqueIjaems JournalBelum ada peringkat
- Fiber Reinforced Composites Mallick Solution Manual PDFDokumen6 halamanFiber Reinforced Composites Mallick Solution Manual PDFGECJ0% (5)
- 19 Fe2O3 - CNTDokumen9 halaman19 Fe2O3 - CNTraj mallickBelum ada peringkat
- Nanostructured Cobalt Zinc Ferrite Thin Films For Gas Sensor ApplicationDokumen10 halamanNanostructured Cobalt Zinc Ferrite Thin Films For Gas Sensor ApplicationInternational Journal of Innovative Science and Research TechnologyBelum ada peringkat
- 8649 24199 2 PBDokumen6 halaman8649 24199 2 PBAliy Kanza Al GhiffariBelum ada peringkat
- Advance Material Draft Version1.0Dokumen16 halamanAdvance Material Draft Version1.0Yz LowBelum ada peringkat
- Kavya PaperDokumen5 halamanKavya PaperNAVEEN KUMAR S NBelum ada peringkat
- Ag TiO2 SintesiDokumen8 halamanAg TiO2 SintesiLuca AccorsiBelum ada peringkat
- Preparation and Characterization of Graphite Nano SheetsDokumen7 halamanPreparation and Characterization of Graphite Nano SheetssygabrielaBelum ada peringkat
- Journal of Physics and Chemistry of Solids: 2 Amir Farzaneh, Mousa Javidani, Mehdi D. Esrafili, Omer MermerDokumen11 halamanJournal of Physics and Chemistry of Solids: 2 Amir Farzaneh, Mousa Javidani, Mehdi D. Esrafili, Omer Mermergta gtaBelum ada peringkat
- Graphene Oxide-Tio Composite Filtration Membranes and Their Potential Application For Water PurificationDokumen7 halamanGraphene Oxide-Tio Composite Filtration Membranes and Their Potential Application For Water PurificationSyed Ahmad Ali ShahBelum ada peringkat
- Band Gap Studies of Anatase Tio2 NanoparticlesDokumen7 halamanBand Gap Studies of Anatase Tio2 Nanoparticlesnarayanan1701_318170Belum ada peringkat
- Preprints202008 0325 v1Dokumen8 halamanPreprints202008 0325 v1Ulisses OliveiraBelum ada peringkat
- s10854 017 6943 1Dokumen9 halamans10854 017 6943 1karthik tvkBelum ada peringkat
- Synthesis of Visible-Light Responsive Graphene Oxide-TiO2 Composites With P-N HeterojunctionDokumen8 halamanSynthesis of Visible-Light Responsive Graphene Oxide-TiO2 Composites With P-N HeterojunctionFamiloni LayoBelum ada peringkat
- Crystals: The Investigation For Coating Method of Titanium Dioxide Layer in Perovskite Solar CellsDokumen9 halamanCrystals: The Investigation For Coating Method of Titanium Dioxide Layer in Perovskite Solar CellsThanakrit SintiamBelum ada peringkat
- Applied Surface ScienceDokumen9 halamanApplied Surface ScienceŞebnem Gül İlarslanBelum ada peringkat
- Effect of Sonication Time On The Production of Graphene by Electrochemical Exfoliation MethodDokumen6 halamanEffect of Sonication Time On The Production of Graphene by Electrochemical Exfoliation MethodYe Zar Ni HtweBelum ada peringkat
- Electrochemically Exfoliated Graphene Production by Using Pure Graphite and Pencil CoreDokumen8 halamanElectrochemically Exfoliated Graphene Production by Using Pure Graphite and Pencil CoreErhan KaracaBelum ada peringkat
- Corrosion Protection of AZ31 Magnesium Alloy by A TiO2 Coating Prepared by LPD MethodDokumen4 halamanCorrosion Protection of AZ31 Magnesium Alloy by A TiO2 Coating Prepared by LPD MethodRahul MadanBelum ada peringkat
- Synthesis of Ethanol-Soluble Few-Layer Graphene Nanosheets For Flexible and Transparent Conducting Composite FilmsDokumen9 halamanSynthesis of Ethanol-Soluble Few-Layer Graphene Nanosheets For Flexible and Transparent Conducting Composite FilmsNOrma690Belum ada peringkat
- TiO Rgo Metilen Blue 2016Dokumen7 halamanTiO Rgo Metilen Blue 2016Rafif QuthronadaBelum ada peringkat
- New Route To Synthesize of Graphene Nano Sheets: Oriental Journal of Chemistry February 2018Dokumen7 halamanNew Route To Synthesize of Graphene Nano Sheets: Oriental Journal of Chemistry February 2018maryamBelum ada peringkat
- Blair 2016Dokumen5 halamanBlair 2016khabanjar0Belum ada peringkat
- J Jallcom 2015 09 241Dokumen8 halamanJ Jallcom 2015 09 241THƯ NGUYỄN MINHBelum ada peringkat
- 2 s2.0 85030846253Dokumen7 halaman2 s2.0 85030846253ARSALAN AHMADBelum ada peringkat
- Tio Nanocrystals Grown On Graphene As Advanced Photocatalytic Hybrid MaterialsDokumen5 halamanTio Nanocrystals Grown On Graphene As Advanced Photocatalytic Hybrid MaterialsAdi Primanto ShevaBelum ada peringkat
- Thong Le BaDokumen28 halamanThong Le BaAsif Abdur RehmanBelum ada peringkat
- Franc 2006Dokumen6 halamanFranc 2006Xiongyu LuoBelum ada peringkat
- Investigating The Thermo Mechanical Properties of Aluminum-Graphene Nano Platelets Composites Developed by Friction StirDokumen8 halamanInvestigating The Thermo Mechanical Properties of Aluminum-Graphene Nano Platelets Composites Developed by Friction StirMohammed GamilBelum ada peringkat
- tmp6E7F TMPDokumen6 halamantmp6E7F TMPFrontiersBelum ada peringkat
- The 243 Steps of Making Photonic Integrated Circuits in InpDokumen5 halamanThe 243 Steps of Making Photonic Integrated Circuits in InpBaranigha CjBelum ada peringkat
- Annual Safety Report 2015Dokumen31 halamanAnnual Safety Report 2015Baranigha CjBelum ada peringkat
- MusicDokumen28 halamanMusicBaranigha CjBelum ada peringkat
- Kin 61a JL Spring 2015Dokumen10 halamanKin 61a JL Spring 2015Baranigha CjBelum ada peringkat
- SONGSDokumen19 halamanSONGSBaranigha CjBelum ada peringkat
- 200W SyllabusDokumen9 halaman200W SyllabusBaranigha CjBelum ada peringkat
- Electronic Properties of Engineering MaterialsDokumen330 halamanElectronic Properties of Engineering MaterialsBaranigha Cj100% (6)
- Codigos de SMD CapasitoresDokumen26 halamanCodigos de SMD CapasitoresHugo Roberto RibeiroBelum ada peringkat
- EDC MAUALdDokumen98 halamanEDC MAUALdsrinivaskaredlaBelum ada peringkat
- Power BJTDokumen14 halamanPower BJTJahangeer SoomroBelum ada peringkat
- Flyback Converter With LeakageDokumen34 halamanFlyback Converter With LeakageVinodBelum ada peringkat
- 5 KVA UPS Docs HI REL PDFDokumen53 halaman5 KVA UPS Docs HI REL PDFAbdelwhab ElsaftyBelum ada peringkat
- K2761 FujiElectricDokumen2 halamanK2761 FujiElectricCampuran CHANNELBelum ada peringkat
- High-Voltage, High-Power, ZVS, Full-Bridge PWM Converter Employing An Active SnubberDokumen8 halamanHigh-Voltage, High-Power, ZVS, Full-Bridge PWM Converter Employing An Active Snubberdr psBelum ada peringkat
- Silicon Bridge Rectifiers KBP2005 - KBP210Dokumen3 halamanSilicon Bridge Rectifiers KBP2005 - KBP210Emad Ali Abu HassnaienBelum ada peringkat
- BY251P Thru BY255P: FeaturesDokumen2 halamanBY251P Thru BY255P: Featureskwagga125Belum ada peringkat
- NCP12400 Eval BoardDokumen11 halamanNCP12400 Eval Board3degreesBelum ada peringkat
- 1 s2.0 S2211379723001274 MainDokumen7 halaman1 s2.0 S2211379723001274 MainArunBelum ada peringkat
- Rutu Gate PDFDokumen2 halamanRutu Gate PDFRuturaj MendhegiriBelum ada peringkat
- Presentation: On Light DetectorDokumen14 halamanPresentation: On Light Detectorxeem conceptsBelum ada peringkat
- Fundamental STADokumen47 halamanFundamental STAsatBelum ada peringkat
- CXD1265 - CCD Camera Timing GeneratorDokumen24 halamanCXD1265 - CCD Camera Timing Generator40818248Belum ada peringkat
- THAT 4301 DatasheetDokumen12 halamanTHAT 4301 DatasheetkelvinBelum ada peringkat
- 21 Safety Rules For Working With Electrical EquipmentDokumen6 halaman21 Safety Rules For Working With Electrical EquipmentSumer LandBelum ada peringkat
- Service Manual: Color TelevisionDokumen74 halamanService Manual: Color TelevisionRichard HerBelum ada peringkat
- JKM270PP-60-A: Poly Crystalline Module 255-270 WattDokumen2 halamanJKM270PP-60-A: Poly Crystalline Module 255-270 Wattnelson_grandeBelum ada peringkat
- Applications of SCR Diac Triac and Converter FamilyDokumen38 halamanApplications of SCR Diac Triac and Converter FamilyFrancis GambolBelum ada peringkat
- CHAPTER 2. Physics f5Dokumen45 halamanCHAPTER 2. Physics f5Normawarni HasanBelum ada peringkat
- IcasesyDokumen40 halamanIcasesyPrem SagarBelum ada peringkat
- Operation of Vcsel at Pulsed ConditionsDokumen10 halamanOperation of Vcsel at Pulsed Conditionsvijila vargheseBelum ada peringkat
- Physics Semiconductors Project PDFDokumen25 halamanPhysics Semiconductors Project PDFAman BindBelum ada peringkat
- AIC Selection GuideDokumen7 halamanAIC Selection GuideWerner LouwBelum ada peringkat
- CD4027BC Dual J-K Master/Slave Flip-Flop With Set and Reset: General Description FeaturesDokumen6 halamanCD4027BC Dual J-K Master/Slave Flip-Flop With Set and Reset: General Description FeaturesGoodLookingPirateBelum ada peringkat
- DSU ManualDokumen204 halamanDSU ManualCarlos Alberto Rueda100% (1)
- MKW41Z512RMDokumen2.425 halamanMKW41Z512RMКомпания ЕЛАМЕД АО Елатомский приборный заводBelum ada peringkat
- Multisim Tutorial Using Bipolar Transistor Circuit." N.P., N.D. WebDokumen1 halamanMultisim Tutorial Using Bipolar Transistor Circuit." N.P., N.D. Webmax pyneBelum ada peringkat
- Slide Deck 1-DC AnalysisDokumen6 halamanSlide Deck 1-DC AnalysisPrithviraj NetkeBelum ada peringkat