00007
Diunggah oleh
Eugenio Daniel Martinez HurtadoJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
00007
Diunggah oleh
Eugenio Daniel Martinez HurtadoHak Cipta:
Format Tersedia
REGIONAL ANESTHESIA AND ACUTE PAIN
ORIGINAL ARTICLE
Ultrasound-Guided Interscalene Block
Reevaluation of the Stoplight Sign and Clinical Implications
Carlo D. Franco, MD* and James M. Williams, PhD
Background and Objectives: The stoplight sign is a frequently
described image during ultrasound-guided interscalene block, referring to 3 hypoechoic structures found between the anterior and middle scalene muscles.
This study was designed to establish the ultrasound-anatomy correlation of this sign and to find any other anatomical features within the
roots that could help with the interpretation of the ultrasound images
obtained at the interscalene level.
Methods: We performed 20 dissections of the brachial plexus in 10
embalmed human cadavers and systematically analyzed and measured
the roots of C5 to C7 and then correlated these findings with ultrasonographic images on file.
Results: We found that the C5 root is significantly smaller than either C6
or C7 (P < 0.0001). We also found that C6 and C7, but not C5, frequently
present macroscopic evidence of intraroot splitting visible to the naked eye.
We also found that the roots of C5 and C6, but not of C7, present frequent
variations in their relationship with the scalene muscles.
Conclusions: Our results provide the anatomic basis to define the
stoplight sign as one made of, from cephalad to caudal, the root of
C5, the upper fascicle(s) of C6, and the lower fascicle(s) of C6 without
contribution from C7. The important clinical implication is that an
injection attempted between what is commonly perceived as the
gap between C6 and C7 would indeed be an intraneural injection
at C6, which could potentially spread toward the neuraxial space.
(Reg Anesth Pain Med 2016;41: 452459)
he interscalene brachial plexus block was introduced by Winnie1 in 1970 and has since become the approach of choice
for anesthesia and analgesia of the shoulder.2 Ultrasound-guided
interscalene block, a more recent modality,3,4 is now widely practiced around the world.512
Ultrasound-guided interscalene block requires imaging the
brachial plexus at the most cephalad portion of the interscalene
space (usually referred to as interscalene groove in anesthesia literature). This space, triangular in shape,13 is bound by the anterior
and middle scalene muscles that diverge from each other as they
approach their insertion on the first rib (Fig. 1). Ultrasound imaging of the plexus around the apex of the interscalene space usually
reveals 3 hypoechoic round structures individually delineated
by thin hyperechoic halos14 and arranged from cranial to caudal in between the anterior and middle scalene muscles. This
From the *Department of Anesthesiology, John H. Stroger Jr. Hospital of Cook
County; and Department of Anatomy and Cell Biology, Section of Clinical
Anatomy, Rush University Medical Center, Chicago, IL.
Accepted for publication February 24, 2016.
Address correspondence to: Carlo D. Franco, MD, Regional Anesthesia, John
H. Stroger Jr. Hospital of Cook County, 1901 West Harrison St, Suite 5670,
Chicago, IL 60612 (email: carlofra@aol.com).
Only intradepartmental funds were used.
The authors declare no conflict of interest.
This study has not been presented either in part or as a whole anywhere nor was
it submitted for consideration to any other journal.
Copyright 2016 by American Society of Regional Anesthesia and Pain
Medicine
ISSN: 1098-7339
DOI: 10.1097/AAP.0000000000000407
452
ultrasonographic image has been referred to as the stoplight
sign.1518 Despite our familiarity with this image, there is no
consensus as to what the hypoechoic nodules represent and
where to inject.19 The majority of authors think that they represent the most proximal roots (C5-C7) of the brachial
plexus,7,12,14,16,17,2022 whereas others believe that they correspond to the 3 brachial plexus trunks.5,15,23,24
In our experience, tracing these hypoechoic structures proximally under ultrasound reveals that not every hypoechoic structure corresponds with a single nerve. This is especially the case
of the C6 root that is frequently imaged as a bifascicular or
multifascicular structure that converges into a single nerve as
the root approaches the intervertebral foramen (Fig. 2, A and B).
Although this impression is shared by other practitioners through
personal communications, it is only rarely mentioned in the literature.2528 In addition to this, our experience in the anatomy
laboratory has demonstrated that the root of C5 is usually significantly smaller than C6 and that C6 frequently shows macroscopic evidence, to the naked eye, of some degree of bifurcation.
Based on this experience, we decided to perform a series
of dissections to analyze the macroscopic appearance and relative sizes of the proximal roots (C5-C7) of the brachial plexus,
with the aim of determining the anatomical basis of the stoplight sign as well as finding any other macroscopic features
within the roots that could correlate with ultrasound images obtained at the interscalene level.
METHODS
A total of 20 brachial plexuses in 10 embalmed human cadavers (5 males and 5 females) were dissected. The dissections
were performed at the anatomy laboratory of Rush University
Medical Center in Chicago using regular dissecting tools and
the unaided eye. The cadavers were obtained from the Anatomical
Gift Association of Illinois, and their procurement and handling
were in accordance to the policies and procedures of Rush University as well as state and federal laws and regulations.
The specimens were lightly embalmed, as done for medical
anatomical research. Their ages at the time of death ranged from
61 to 95 years. After partial removal of the prevertebral fascia,
the anterior and middle scalene muscles were exposed, as well
as the nerves of the brachial plexus, with special attention to the
roots of C5, C6, and C7. The trajectory of the roots and trunks
with respect to the scalene muscles was verified and noted. Notes
were also made concerning the appearance of the roots looking for
any evidence of macroscopic splitting or bifurcation.
The roots were then excised as close to the distal end of the
transverse process proximally and at the level of the respective
trunk distally. A digital caliper (Fisher Scientific, Pittsburgh,
Pennsylvania) was used to measure (in millimeters) the diameter of the roots at midpoint of the available length by a single
observer (C.F.). After measurements and macroscopic examination, some roots underwent careful removal of epineurial
layers to better reveal their structural arrangement. Notes were
made and photographs were taken throughout.
Simple descriptive statistics were used to summarize root
location, diameter, and presence of anatomical variation. To
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
Reevaluation of the Stoplight Sign
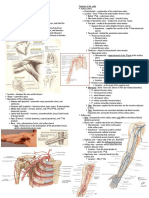
FIGURE 1. Dissection of left brachial plexus, medial view as seen from the shoulder, to show the diverging scalene muscles and the resulting
triangular space in-between. The roots and divisions are long compared with the short trunks. A, anterior division upper trunk; AS, anterior
scalene; MS, middle scalene; P, posterior division upper trunk; SA, subclavian artery; SS, suprascapular nerve; TRAP, trapezius; UT, upper trunk.
assess diameter size differences between root levels, we performed
a 1-way analysis of variance. To adjust for multiple comparisons, a
post hoc analysis was performed using the Tukey highly significant
difference test.
to the anterior scalene muscle. Although C5 and C6 roots
showed some variability in this respect, C7 was found consistently between the scalene muscles. Table 1 summarizes these
findings. An asymmetric pattern between both sides was found
in 4 (40%) of 10 specimens.
RESULTS
The dissection findings were categorized as 1) root trajectory
and symmetry, 2) nerve root diameter, and 3) macroscopic signs
of intraroot splitting.
Root Trajectory and Symmetry
The trajectory of the C5 to C7 roots with respect to the scalene muscles was noted to follow 3 different patterns: the root
was located between the anterior and middle scalene muscles,
passed through the anterior scalene muscle, or traveled anterior
Root Diameter
From the dissection of 20 brachial plexuses, 60 individual
roots (C5-C7) were collected for measurement. The root of C5
was clearly the smallest root (P < 0.0001) in 19 of 20 cases (Fig. 3),
whereas C6 and C7 roots were almost equally larger. The diameters of C5 to C7 roots in all specimens are shown in Table 2.
The mean diameters of C5 to C7 roots in millimeters, with
standard deviations, were as follows: C5, 3.2 0.9; C6, 5.1
0.6; and C7, 5.3 0.7. Statistical analysis revealed a significant
FIGURE 2. Ultrasound tracing of the root of C6, with 2A showing C6 root bifurcated and, 2B, the same root seems monofascicular as it
approaches the intervertebral foramen. AS, anterior scalene; MS, middle scalene.
2016 American Society of Regional Anesthesia and Pain Medicine
453
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
Franco and Williams
TABLE 1. Trajectory of C5 to C7 With Respect to Scalene
Muscles
Between
Through
Anterior
Total (n)
TABLE 2. Diameter of C5 to C7 Roots in All 20 Plexuses
C5 (n)
C6 (n)
C7 (n)
14
4
2
20
17
2
1
20
20
0
0
20
Between, in-between anterior and middle scalene muscles; through,
through anterior scalene muscle; anterior, anterior to anterior scalene
muscle.
difference between the size of C5 compared with both C6 and C7,
whereas no difference was found between the sizes of C6 and C7.
Size difference among the 3 roots with a 95% confidence interval
(CI):
C5 compared with C6: 95% CI, 1.33 to 2.45 mm, P < 0.0001;
C5 compared with C7: 95% CI, 1.58 to 2.70 mm, P < 0.0001;
C6 compared with C7: 95% CI, 0.31 to 0.81 mm, P = 0.53.
Macroscopic Appearance
The macroscopic examination of the roots revealed that C5
was significantly smaller than C6 and C7 and rarely showed any
macroscopic evidence of splitting, with 1 exception in which the
root of C5 was as large as C6 (4.87 vs 4.51 mm), and both were
split (Fig. 4). The root of C6 was not only larger than C5 but frequently showed evidence of splitting in situ (intact epineurium)
(Fig. 5). The evidence of fascicular splitting became more apparent after the outer layers of the epineurium were removed, as
shown in Figures 6, 7, and 8. The summary of these findings is
presented in Table 3.
DISCUSSION
This study was undertaken to better understand the correlation between the images commonly obtained during ultrasound-
No.
Sex
Side
C5, mm
C6, mm
C7, mm
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
Male
Male
Male
Male
Male
Male
Female
Female
Female
Female
Female
Female
Male
Male
Male
Male
Female
Female
Female
Female
Right
Left
Right
Left
Right
Left
Right
Left
Right
Left
Right
Left
Right
Left
Right
Left
Right
Left
Right
Left
2.21
1.72
3.25
3.69
4.53
3.88
2.93
3.15
2.18
1.80
2.08
2.45
3.94
3.36
3.84
3.70
4.87
3.62
3.46
3.11
4.84
4.58
6.16
5.79
5.52
5.54
4.43
4.79
4.28
4.68
5.34
4.36
5.78
5.26
5.38
5.57
4.51
4.51
5.52
4.70
5.44
4.83
5.75
5.82
6.11
6.11
4.14
4.38
4.26
5.35
5.31
4.79
5.98
6.33
4.52
5.16
4.90
5.04
6.36
6.11
guided interscalene block and the macroscopic anatomy of
the brachial plexus at that level. The most important issues that
we tried to address were, first, the anatomical correlation of the
so-called stoplight sign and, second, the anatomical features
within the roots that could support the contention that not every
hypoechoic structure found at the interscalene level, specifically the root of C6, corresponds with a single nerve, as we
and others have noticed.2528 Two important findings were made.
First, we established that the root of C5 is significantly smaller
than the roots of either C6 or C7. Second, we found that the roots
of C6 and C7, but not of C5, frequently present at least 1 visible
FIGURE 3. Comparative sizes of C5 to C7 roots. MT, middle trunk; UT, upper trunk.
454
2016 American Society of Regional Anesthesia and Pain Medicine
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
Reevaluation of the Stoplight Sign
FIGURE 4. A case of C5 splitting. C5 and C6 are shown to be about equal sizes, and both demonstrate evidence of macroscopic splitting.
Shown after partial removal of epineurium.
FIGURE 5. Dissection of left brachial plexus (different specimen from that of Fig. 1). Frontal view, after removal of prevertebral fascia and most
of connective tissue to show bare nerves and muscles. C5 is clearly the smallest root, and C6 shows some evidence of macroscopic splitting
(arrow) with intact epineurium. MS, middle scalene; Ph ner, phrenic nerve; SA, subclavian artery.
2016 American Society of Regional Anesthesia and Pain Medicine
455
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Franco and Williams
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
FIGURE 6. C5 to C6 roots forming upper trunk (UT), right side. A, With intact epineurium. B, After partial removal of the epineurium.
FIGURE 7. C5 to C6 roots forming upper trunk (UT), left side. A, With intact epineurium. B, After partial removal of the epineurium.
FIGURE 8. C5 to C6 roots forming the upper trunk (UT), left side. The C6 root shows macroscopic evidence of double splitting (arrows).
456
2016 American Society of Regional Anesthesia and Pain Medicine
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
TABLE 3. Macroscopic Appearance of Roots C5 to C7
C5
C6
C7
No Visible Split (n)
At Least 1 Visible Split (n)
19
1
2
1
19
18
sign of splitting within their structure. This was even more apparent in those specimens that had some layers of epineurium
removed. These 2 findings provide, in our view, solid evidence
for the stoplight ultrasound sign to be interpreted as the crosssectional image of C5 and C6 roots, without participation of
C7. Of the 3 components of the sign, the superior 1, or the red
light, would correspond with C5; the middle component, or the
yellow light, would be the upper fascicle(s) of C6; and the distal
component, or the green light, would be the lower fascicle
(s) of C6. The fact that C5 was found to be significantly smaller
than C6, and the splitting of a larger C6, correlates well with
this sign being usually composed of 3 hypoechoic structures
of similar size. The anatomy/ultrasound correlation is shown
in panels A and B of Figure 9.
However, we believe that even more important than establishing a credible correlation between the ultrasound images
and the anatomy of the interscalene block are the clinical implications of these findings. The macroscopic bifurcation of C6
into 2 fascicles or 2 groups of fascicles, frequently mistaken
as being 2 separate roots (C6 and C7), could lead to an unintended intraneural injection29 at this level. We have not failed
to notice30 that such a bifurcation provides an entrance path
into the neuraxis and could help explain some catastrophic outcomes associated with interscalene blocks.3135 The spread into
the neuraxial space from an interscalene injection has been usually explained as an injection within the dural sleeve of a spinal
nerve. However, because this sleeve rarely extends beyond the
intervertebral foramen,3638 our findings seem to provide a
more likely mechanism for such spread. Furthermore, it seems
fair to speculate that an unintended intraneural injection at C6
could also be associated with the relatively high incidence of
dysesthesias observed after interscalene blocks,3943 for which
no credible explanation has ever been offered. An injection
Reevaluation of the Stoplight Sign
between the fascicles of C6, indeed intraneural but extrafascicular,
could be accompanied by low opening pressures44 (false-negative).
The possibility that the stoplight sign could instead correspond to the cross-sectional image of the trunks seems highly
unlikely for many reasons. First, the usual insonation for an
interscalene block is too cephalad and angled too medial to
be able to show all 3 trunks in 1 image12; second, it is unlikely
that a cross-sectional image of the lower trunk would not demonstrate in the same image the subclavian artery in its immediate vicinity; third, the trunks are indeed more multifascicular4547 than
the roots as they start to take the appearance of more peripheral
nerves with more abundant connective tissue.22
Our extensive literature review found supportive anatomical
and clinical evidence for C5 being the smallest root, especially
when compared with C6 and C7.4751 However, although strong
when present, the evidence is surprisingly not as pervasive as it
would be expected considering the significant difference in root
size that we and others have documented. Regarding the macroscopic bifurcation of C6, Kerr's48 comprehensive 1918 publication on the human brachial plexus presents a diagram depicting
a C5 root significantly smaller than C6 and the latter showing a
structure that seems to suggest cephalad and caudal groups of fascicles. Unfortunately, no explicit statement to that effect is made.
To our knowledge, other than this cryptic diagram, the literature
does not explicitly describe the roots of the plexus as exhibiting
any macroscopic evidence of septation that would betray their
multifascicular nature. Thus, we believe that our anatomical findings, which nicely correlate with known ultrasonographic images,
are indeed novel.
The ultrasound image of a split C6 converging into a single
hypoechoic structure when traced proximally is supported by the
literature that shows the ventral branch of the spinal nerve as being
monofascicular,52 whereas the C5 to T1 roots are multifascicular
(27 fascicles).52 Our study suggests that at least C6 and C7 roots
not only are multifascicular but part of that arrangement is usually
visible to the naked eye and likely to ultrasound.
Although we did not intend to study brachial plexus variations or asymmetry, our findings are in accordance with the literature.47 That lack of symmetry involves the size and structure of
individual nerves, as well as their relationship with the scalene
muscles. Whatever their importance, these findings are only incidental to our study.
FIGURE 9. Ultrasound-anatomy correlation of the stoplight sign. A, Depiction of a short segment of roots of C5 and C6 as they converge to
form the upper trunk (UT). The splitting of C6 is clear after removal of the epineurium. B, Ultrasound image depicting the stoplight sign
formed by 3 hypoechoic dots between the anterior scalene (AS) and the middle scalene (MS) muscles.
2016 American Society of Regional Anesthesia and Pain Medicine
457
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
Franco and Williams
In light of our findings, and considering that the brachial
plexus contribution to the innervation of the shoulder comes almost exclusively from C5 and C6, with only marginal contribution from C7,27,53 our advice is that, during an interscalene
block, the single target of injection be the space between C5
and C6. No attempt should be made to inject or place a catheter
distal to this level.54 It would also be advisable that, when in
doubt, the operator traces the hypoechoic structure(s) proximally to ascertain whether they correspond to a splitting of a
single nerve or indeed to 2 separate roots.
Our findings do not support the concept of common epineurium between 2 different roots, as some have suggested.55 It seems
clear from the literature that every single root exits through a particular intervertebral foramen surrounded by its own dural sleeve,
which at the level of the intervertebral foramen becomes continuous with the epineurium of that root.13,3638 The multifascicular
arrangement of the root does not contradict this fact, as all these
fascicles form 1 nerve and 1 single epineurium individualizes it.
Our study has some limitations. The use of lightly embalmed
cadavers could be questioned. However, the main findings: relative
(not absolute) size of nerves and macroscopic evidence of fascicular
arrangement within the roots should not be affected by it. In fact, we
have found similar features in fresh cadavers. The evidence that C5
is the smallest root among C5 to C7 is overwhelming. Another limitation is that we did not study the histological features of the roots
so we could not establish the exact number of fascicles per root nor
their course and disposition. We believe that the absence of this information at this time does not diminish the anatomical-ultrasound
correlation we have established and the potentially relevant
clinical implications.
In summary, we found that C5 is frequently the smallest root
of the brachial plexus and that C6 is both one of the largest and
frequently exhibits macroscopic signs of splitting. These anatomical features provide the anatomical basis for the stoplight sign to
correspond with the cross-sectional image of C5 and both (or
more) fascicles of C6. This stoplight sign usually has 3 lights,
but they are made of only 2 nerves.
REFERENCES
1. Winnie AP. Interscalene brachial plexus block. Anesth Analg. 1970;49:
455466.
2. Singelyn FJ, Lhotel L, Fabre B. Pain relief after arthroscopic shoulder
surgery: a comparison of intraarticular analgesia, suprascapular nerve
block, and interscalene brachial plexus block. Anesth Analg. 2004;99:
589562.
3. Jandrasits O, Likar R, Marhofer P, Weinstabl C, Kapral S. The use of
ultrasonography for regional anesthetic techniques: upper extremity
blockades. Acta Anaesthesiol Scand. 1998;42:4851.
4. Chan VW. Applying ultrasound imaging to interscalene brachial plexus
block. Reg Anesth Pain Med. 2003;28:340343.
5. De Andrs J, Sala-Blanch X. Ultrasound in the practice of brachial plexus
anesthesia. Reg Anesth Pain Med. 2002;27:7789.
6. Sinha SK, Abrams JH, Weller RS. Ultrasound-guided interscalene needle
placement produces successful anesthesia regardless of motor stimulation
above or below 0.5 mA. Anesth Analg. 2007;105:848852.
7. Marhofer P, Greher M, Kapral S. Ultrasound guidance in regional
anaesthesia. Br J Anaesth. 2005;94:717.
8. Kapral S, Greher M, Huber G, et al. Ultrasonographic guidance improves
the success rate of interscalene brachial plexus blockade. Reg Anesth Pain
Med. 2008;33:253258.
9. Neal JM, Brull R, Chan VW, et al. The ASRA evidence-based medicine
assessment of ultrasound-guided regional anesthesia and pain medicine:
Executive summary. Reg Anesth Pain Med. 2010;35:S1S9.
458
10. Blaivas M, Adhikari S, Lander L. A prospective comparison of procedural
sedation and ultrasound-guided interscalene nerve block for shoulder
reduction in the emergency department. Acad Emerg Med. 2011;18:
922927.
11. Antonakakis JG, Ting PH, Sites B. Ultrasound-guided regional anesthesia
for peripheral nerve blocks: an evidence-based outcome review.
Anesthesiol Clin. 2011;29:179191.
12. Lapegue F, Faruch-Bilfeld M, Demondion X, et al. Ultrasonography of the
brachial plexus, normal appearance and practical applications. Diagn Interv
Imaging. 2014;95:259275.
13. Gray H, Davies DV, Coupland RE. Gray's Anatomy Descriptive and
Applied. 34th ed. London: Longmans; 1969.
14. Brown D. Atlas of Regional Anesthesia. 4th ed. Philadelphia, PA: Saunders
Elsevier; 2010.
15. Moore CL. Ultrasound-guided procedures in emergency medicine.
Ultrasound Clin. 2011;6:277289.
16. Hill J, Singh VM, Sen SK. Anesthesia for upper extremity surgery. In:
Chang J, Neligan P (eds). Plastic Surgery. 3rd ed. Philadelphia, PA: Elsevier
Saunders; 2012:92105.
17. Mackenzie CR, Cornell CN, Memtsoudis SG (eds). Perioperative Care of
the Orthopedic Patient. New York, NY: Springer; 2014.
18. Waldman SD. Brachial plexus block: interscalene approach. In: Atlas of
Interventional Pain Management. 4th ed. Philadelphia, PA: Elsevier; 2015:
203210.
19. Spence BC, Beach ML, Gallagher JD, Sites BD. Ultrasound-guided
interscalene blocks: understanding where to inject the local anaesthetic.
Anaesthesia. 2011;66:509514.
20. Marhofer P, Harrop-Griffiths W, Willschke H, Kirchmair L. Fifteen years of
ultrasound guidance in regional anesthesia: part 2recent developments in
block techniques. Br J Anaesth. 2010;104:673683.
21. Demondion X, Herbinet P, Boutry N, Fontaine C, Francke JP, Cotten A.
Sonographic mapping of the normal brachial plexus. Am J Neuroradiol.
2003;24:13031309.
22. Van Geffen GJ, Moayeri N, Bruhn J, Scheffer GJ, Chan VW, Groen GJ.
Correlation between ultrasound imaging, cross-sectional anatomy, and
histology of the brachial plexus: a review. Reg Anesth Pain Med.
2009;34:490497.
23. Yang WT, Chui PT, Metreweli C. Anatomy of the normal brachial plexus
revealed by sonography and the role of sonographic guidance in anesthesia
of the brachial plexus. Am J Roent. 1998;171:16311636.
24. Popovic J, Morimoto M, Wambold D, Blanck TJ, Rosenberg AD. Current
practice of ultrasound-assisted regional anesthesia. Pain Pract. 2006;6:
127134.
25. Filip P. Complex arithmetic at the brachial plexus roots (letter).
Reg Anesth Pain Med. 2016;34:7980.
26. Gonzales J, Auyong D, Grant S. Upper limb ultrasound guided regional
anesthesia. In: Grant SA, Auyong DB (eds). Ultrasound Guided
Regional Anesthesia. New York, NY: Oxford University Press;
2012:3181.
27. Burckett-St Laurent D, Chan V, Chin KJ. Refining the ultrasound-guided
interscalene brachial plexus block: the superior trunk approach. Can J
Anaesth. 2014;61:10981102.
28. Grant SA, Smith EC. Ultrasound of cervical roots and brachial plexus in
neonates. Muscle Nerve. 2015;51:626.
29. Franco CD. Connective tissues associated with peripheral nerves.
Reg Anesth Pain Med. 2012;37:363365.
30. Watson JD, Crick FHC. Molecular structure of nucleic acids. A structure
for deoxyribose nucleic acid. Nature. 1953;171:737738.
31. Tetzlaff JE, Yoon HJ, Dilger J, Brems J. Subdural anesthesia as a
complication of an interscalene brachial plexus block. Case report.
Reg Anesth. 1994;19:357359.
2016 American Society of Regional Anesthesia and Pain Medicine
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Regional Anesthesia and Pain Medicine Volume 41, Number 4, July-August 2016
32. Passannante AN. Spinal anesthesia and permanent neurologic deficit after
interscalene block. Anesth Analg. 1996;82:873874.
33. Benumof JL. Permanent loss of cervical spinal cord function associated
with interscalene block performed under general anesthesia.
Anesthesiology. 2000;93:15411544.
34. Ala-Kokko TI, Lppnen A, Alahuhta S. Two instances of central
nervous system toxicity in the same patient following repeated
ropivacaine-induced brachial plexus block. Acta Anaesthesiol Scand.
2000;44:623626.
35. Yanovski B, Gaitini L, Volodarski D, Ben-David B. Catastrophic
complication of an interscalene catheter for continuous peripheral nerve
block analgesia. Anaesthesia. 2012;67:11661169.
36. Mancall EL, Brock DG. Spinal cord and nerve roots. In: Mancall EL,
Brock DG (eds). Gray's Clinical Neuroanatomy: The Anatomical Basis for
Clinical Neuroscience. Philadelphia, PA: Elsevier Saunders; 2011:
117148.
37. Louis-Ugbo J, Pedlow FX Jr, Heller JG. Anatomy of the cervical spine. In:
Benzel EC (ed). The Cervical Spine. 5th ed. Philadelphia, PA: Lippincott
Williams & Wilkins; 2012:133.
38. Ombregt L. Applied Anatomy of the Cervical Spine. A System of
Orthopaedic Medicine. 3rd ed. Churchill Livingstone Elsevier:
Edinburgh; 2013.
39. Borgeat A, Ekatodramis G, Kalberer F, Benz C. Acute and nonacute
complications associated with interscalene block and shoulder surgery:
a prospective study. Anesthesiology. 2001;95:875880.
40. Candido KD, Sukhani R, Doty R Jr, et al. Neurologic sequelae after
interscalene brachial plexus block for shoulder/upper arm surgery: the
association of patient, anesthetic, and surgical factors to the incidence
and clinical course. Anesth Analg. 2005;100:14891495.
41. Brull R, McCartney CJ, Chan VW, El-Beheiry H. Neurological
complications after regional anesthesia: contemporary estimates of risk.
Anesth Analg. 2007;104:965974.
42. Liu SS, Zayas VM, Gordon MA, et al. A prospective, randomized,
controlled trial comparing ultrasound versus nerve stimulator guidance
for interscalene block for ambulatory shoulder surgery for postoperative
neurologic symptoms. Anesth Analg. 2009;109:265271.
2016 American Society of Regional Anesthesia and Pain Medicine
Reevaluation of the Stoplight Sign
43. Liu SS, Yadeau JT, Shaw PM, Wilfred S, Shetty T, Gordon M. Incidence of
unintentional intraneural injection and post-operative neurological
complications with US-guided interscalene and supraclavicular nerve
blocks. Anaesthesia. 2011;66:168174.
44. Altermatt FR, Cummings TJ, Auten KM, Baldwin MF, Belknap SW,
Reynolds JD. Ultrasonographic appearance of intraneural injections in
the porcine model. Reg Anesth Pain Med. 2010;35:203206.
45. Moayeri N, Bigeleisen PE, Groen GJ. Quantitative architecture of
the brachial plexus and surrounding compartments and their
possible significance for plexus blocks. Anesthesiology. 2008;108:
299304.
46. Martinoli C, Bianchi S, Santacroce E, Pugliese F, Graif M, Derchi LE.
Brachial plexus sonography: a technique for assessing the root level.
AJR Am J Roentgenol. 2002;179:699702.
47. Won SJ, Kim BJ, Park KS, Kim SH, Yoon JS. Measurement of crosssectional area of cervical roots and brachial plexus trunks. Muscle Nerve.
2012;46:711716.
48. Kerr AT. The brachial plexus of nerves in man, the variations in its
formation and branches. Am J Anat. 1918;23:285395.
49. Lee HY, Chung IH, Sir WS, et al. Variations of the ventral rami of the
brachial plexus. J Korean Med Sci. 1992;7:1924.
50. Takeuchi M, Wakao N, Kamiya M, et al. Morphological distinction of
cervical nerve roots associated with motor function in 219 healthy
volunteers: a multicenter prospective study. Spine (Phila Pa 1976). 2014;
39:E944E949.
51. Narakas A. Surgical treatment of traction injuries of the brachial plexus.
Clin Orthop Relat Res. 1978;133:7190.
52. Kawai H, Kawabata H (eds). Anatomy of the brachial plexus. In: Brachial
Plexus Palsy. Singapore: World Scientific; 2000:124.
53. Gardner E. The innervation of the shoulder joint. Anat Rec. 1948;102:118.
54. Sites BD, Brull R. Ultrasound guidance in peripheral regional anesthesia:
philosophy, evidence-based medicine, and techniques. Curr Opin
Anaesthesiol. 2006;19:630639.
55. Orebaugh SL, McFadden K, Skorupan H, Bigeleisen PE. Subepineurial
injection in ultrasound-guided interscalene needle tip placement.
Reg Anesth Pain Med. 2010;35:450454.
459
Copyright 2016 American Society of Regional Anesthesia and Pain Medicine. Unauthorized reproduction of this article is prohibited.
Anda mungkin juga menyukai
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDari EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifePenilaian: 4 dari 5 bintang4/5 (5794)
- The Yellow House: A Memoir (2019 National Book Award Winner)Dari EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Penilaian: 4 dari 5 bintang4/5 (98)
- Variable Bleeding Manifestations Characterize Different Types of Surgery in Patients With Severe Factor XI Deficiency Enabling Parsimonious Use of Replacement Therapy Salomon2006Dokumen4 halamanVariable Bleeding Manifestations Characterize Different Types of Surgery in Patients With Severe Factor XI Deficiency Enabling Parsimonious Use of Replacement Therapy Salomon2006Eugenio Daniel Martinez HurtadoBelum ada peringkat
- 2022 American Society of Anesthesiologists Practice Guidelines For Management of The Difficult AirwayDokumen51 halaman2022 American Society of Anesthesiologists Practice Guidelines For Management of The Difficult AirwayCarolina Ormaza GiraldoBelum ada peringkat
- Focused Review: High-Flow Nasal Cannula Oxygen in Adults: An Evidence-Based AssessmentDokumen11 halamanFocused Review: High-Flow Nasal Cannula Oxygen in Adults: An Evidence-Based AssessmentNandhini SivakumarBelum ada peringkat
- PCR LAncetDokumen6 halamanPCR LAncetEugenio Daniel Martinez HurtadoBelum ada peringkat
- Basic Concepts in The Use of Thoracic and LungDokumen11 halamanBasic Concepts in The Use of Thoracic and LungEugenio Daniel Martinez HurtadoBelum ada peringkat
- Airway Management Guidance For The Endemic Phase of COVID-19Dokumen10 halamanAirway Management Guidance For The Endemic Phase of COVID-19Kamar Operasi Hermina BogorBelum ada peringkat
- Tracheal ExtubationDokumen15 halamanTracheal ExtubationEugenio Daniel Martinez HurtadoBelum ada peringkat
- IndianJAnaesth599606-8387767 231757 PDFDokumen12 halamanIndianJAnaesth599606-8387767 231757 PDFCindyPurnamaBelum ada peringkat
- Validation of Modified Mallampati Test With Addition of Thyromental Distance and Sternomental Distance To Predict Difficult Endotracheal Intubation in AdultsDokumen5 halamanValidation of Modified Mallampati Test With Addition of Thyromental Distance and Sternomental Distance To Predict Difficult Endotracheal Intubation in AdultsEugenio Daniel Martinez HurtadoBelum ada peringkat
- Aev371 PDFDokumen22 halamanAev371 PDFEugenio Daniel Martinez HurtadoBelum ada peringkat
- Airway Management in Obese Patient - Minerva 2014Dokumen11 halamanAirway Management in Obese Patient - Minerva 2014Eugenio Daniel Martinez HurtadoBelum ada peringkat
- Bibliografia Video LaryngosDokumen1 halamanBibliografia Video LaryngosEugenio Daniel Martinez HurtadoBelum ada peringkat
- Airway Management Using Transmylohyoid Oroendotracheal (Submental) Intubation in Maxillofacial Trauma - NatlJMaxillofacSurg52138-2466486 - 065104Dokumen4 halamanAirway Management Using Transmylohyoid Oroendotracheal (Submental) Intubation in Maxillofacial Trauma - NatlJMaxillofacSurg52138-2466486 - 065104Eugenio Daniel Martinez HurtadoBelum ada peringkat
- Challenges in Airway ManagementDokumen36 halamanChallenges in Airway ManagementEugenio Daniel Martinez HurtadoBelum ada peringkat
- Abstracts From The Difficult Airway Society Meeting 2014 Bja - Aev258.fullDokumen18 halamanAbstracts From The Difficult Airway Society Meeting 2014 Bja - Aev258.fullEugenio Daniel Martinez HurtadoBelum ada peringkat
- Uso de Guía Aintree para Intubación Con FibroscopioDokumen1 halamanUso de Guía Aintree para Intubación Con Fibroscopio1cucu0Belum ada peringkat
- BJA - Van Zundert Et Al. VLS Better Angle of ViewDokumen3 halamanBJA - Van Zundert Et Al. VLS Better Angle of ViewEugenio Daniel Martinez HurtadoBelum ada peringkat
- IE Visualisation Platform Brochure 496951001 V01 1113Dokumen7 halamanIE Visualisation Platform Brochure 496951001 V01 1113Eugenio Daniel Martinez HurtadoBelum ada peringkat
- Uso de Guía Aintree para Intubación Con FibroscopioDokumen1 halamanUso de Guía Aintree para Intubación Con Fibroscopio1cucu0Belum ada peringkat
- Mallampati 0 2001Dokumen3 halamanMallampati 0 2001Eugenio Daniel Martinez HurtadoBelum ada peringkat
- Br. J. Anaesth.-1998-Mason-305-7Dokumen3 halamanBr. J. Anaesth.-1998-Mason-305-7Eugenio Daniel Martinez HurtadoBelum ada peringkat
- Br. J. Anaesth. 2012 Abdulatif 1038 9Dokumen2 halamanBr. J. Anaesth. 2012 Abdulatif 1038 9Eugenio Daniel Martinez HurtadoBelum ada peringkat
- Difficult and Failed Intubation in 3430 Obstetric General AnestesiasDokumen4 halamanDifficult and Failed Intubation in 3430 Obstetric General AnestesiasEugenio Daniel Martinez HurtadoBelum ada peringkat
- Predicción de Laringoscopía Difícil. Puntuación de Mallampati Extendida Versus MMT, ULBT y RHTDMDokumen6 halamanPredicción de Laringoscopía Difícil. Puntuación de Mallampati Extendida Versus MMT, ULBT y RHTDMEugenio Daniel Martinez HurtadoBelum ada peringkat
- Practice Guidelines For Management of The Difficult Airway 2013Dokumen20 halamanPractice Guidelines For Management of The Difficult Airway 2013Anthony VasconezBelum ada peringkat
- Características Anatómicas Del Cuello Como Marcadores Predictivos de Dificultad para Laringoscopia Directa en Los Hombres y Las MujeresDokumen7 halamanCaracterísticas Anatómicas Del Cuello Como Marcadores Predictivos de Dificultad para Laringoscopia Directa en Los Hombres y Las MujeresEugenio Daniel Martinez HurtadoBelum ada peringkat
- Validation of Modified Mallampati Test With Addition of Thyromental Distance and Sternomental Distance To Predict Difficult Endotracheal Intubation in AdultsDokumen5 halamanValidation of Modified Mallampati Test With Addition of Thyromental Distance and Sternomental Distance To Predict Difficult Endotracheal Intubation in AdultsEugenio Daniel Martinez HurtadoBelum ada peringkat
- Ultrasonido Diafragmático Como Predictor de Extubación Exitosa de La Ventilación MecánicaDokumen6 halamanUltrasonido Diafragmático Como Predictor de Extubación Exitosa de La Ventilación MecánicaEugenio Daniel Martinez HurtadoBelum ada peringkat
- Obstetric Difficult Airway Guidelines - Difficult Airway Society 2015Dokumen9 halamanObstetric Difficult Airway Guidelines - Difficult Airway Society 2015Eugenio Daniel Martinez HurtadoBelum ada peringkat
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDari EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryPenilaian: 3.5 dari 5 bintang3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDari EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RacePenilaian: 4 dari 5 bintang4/5 (895)
- The Little Book of Hygge: Danish Secrets to Happy LivingDari EverandThe Little Book of Hygge: Danish Secrets to Happy LivingPenilaian: 3.5 dari 5 bintang3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeDari EverandShoe Dog: A Memoir by the Creator of NikePenilaian: 4.5 dari 5 bintang4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDari EverandNever Split the Difference: Negotiating As If Your Life Depended On ItPenilaian: 4.5 dari 5 bintang4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDari EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FuturePenilaian: 4.5 dari 5 bintang4.5/5 (474)
- Grit: The Power of Passion and PerseveranceDari EverandGrit: The Power of Passion and PerseverancePenilaian: 4 dari 5 bintang4/5 (588)
- The Emperor of All Maladies: A Biography of CancerDari EverandThe Emperor of All Maladies: A Biography of CancerPenilaian: 4.5 dari 5 bintang4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealDari EverandOn Fire: The (Burning) Case for a Green New DealPenilaian: 4 dari 5 bintang4/5 (74)
- Team of Rivals: The Political Genius of Abraham LincolnDari EverandTeam of Rivals: The Political Genius of Abraham LincolnPenilaian: 4.5 dari 5 bintang4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDari EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaPenilaian: 4.5 dari 5 bintang4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDari EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersPenilaian: 4.5 dari 5 bintang4.5/5 (344)
- Rise of ISIS: A Threat We Can't IgnoreDari EverandRise of ISIS: A Threat We Can't IgnorePenilaian: 3.5 dari 5 bintang3.5/5 (137)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDari EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyPenilaian: 3.5 dari 5 bintang3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDari EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You ArePenilaian: 4 dari 5 bintang4/5 (1090)
- The Unwinding: An Inner History of the New AmericaDari EverandThe Unwinding: An Inner History of the New AmericaPenilaian: 4 dari 5 bintang4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Dari EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Penilaian: 4.5 dari 5 bintang4.5/5 (121)
- Her Body and Other Parties: StoriesDari EverandHer Body and Other Parties: StoriesPenilaian: 4 dari 5 bintang4/5 (821)
- Acland Human AnatomyDokumen51 halamanAcland Human Anatomyjust for download matterBelum ada peringkat
- Shawnoma Del Plexo BraquialDokumen4 halamanShawnoma Del Plexo BraquialjohanesBelum ada peringkat
- Dermatomes of The Upper LimbDokumen55 halamanDermatomes of The Upper LimbGideon WillieBelum ada peringkat
- Brachial Plexus ExaminationDokumen7 halamanBrachial Plexus ExaminationMalvinder Singh DhillonBelum ada peringkat
- Anatomy Upper Limb & Brest (120Qs)Dokumen55 halamanAnatomy Upper Limb & Brest (120Qs)Salma Esam KambalBelum ada peringkat
- Matary Special Surgery 2013 (WWW - Afriqa Sat - Com)Dokumen419 halamanMatary Special Surgery 2013 (WWW - Afriqa Sat - Com)Ahmed Attia100% (1)
- ICD Code ChartDokumen1 halamanICD Code ChartbjpalmerBelum ada peringkat
- Phrenic Nerve Palsy: A Rare Cause of Respiratory Distress in NewbornDokumen5 halamanPhrenic Nerve Palsy: A Rare Cause of Respiratory Distress in NewbornIJAR JOURNALBelum ada peringkat
- For All of The Questions Upper LimpDokumen22 halamanFor All of The Questions Upper LimpKAAB MEDICS ZONEBelum ada peringkat
- Erb'S Palsy: Definition/DescriptionDokumen10 halamanErb'S Palsy: Definition/DescriptionGuhan SubramaniamBelum ada peringkat
- Quiz On The Upper LimbDokumen6 halamanQuiz On The Upper LimbNajah HanimBelum ada peringkat
- UPPER LIMB - 5 Course of Nerves and Vessels in The Upper Limb PDFDokumen10 halamanUPPER LIMB - 5 Course of Nerves and Vessels in The Upper Limb PDFDare DevilBelum ada peringkat
- UE 1 Clavicle FracturesDokumen46 halamanUE 1 Clavicle FracturesDavidBeatonComuladaBelum ada peringkat
- Tendon Transfers For Intrinsic Function in Ulnar Nerve PalsyDokumen21 halamanTendon Transfers For Intrinsic Function in Ulnar Nerve PalsyFernando Martín Arce AlvaBelum ada peringkat
- 1 ANT Anatomy of The Upper Limp TGDokumen129 halaman1 ANT Anatomy of The Upper Limp TGReath Gatkuoth DuothBelum ada peringkat
- Anatomi Fossa AxillarisDokumen56 halamanAnatomi Fossa AxillarisdoktergigikoeBelum ada peringkat
- Ortho Review 1Dokumen67 halamanOrtho Review 1mark tBelum ada peringkat
- Patient Positioning and AnaesthesiaDokumen24 halamanPatient Positioning and AnaesthesiaGrishma KothariBelum ada peringkat
- Thoracic Outlet SyndromeDokumen678 halamanThoracic Outlet Syndromejorgeroca2009Belum ada peringkat
- Our Journey Kylee's StoryDokumen12 halamanOur Journey Kylee's StoryKimberley Zammit GautreauBelum ada peringkat
- 40th Annual Percivall Pott Scientific MeetingDokumen26 halaman40th Annual Percivall Pott Scientific MeetingAlex Mulligan100% (1)
- A Clinical Study On Ayurvedic Approach in Prasav Kaleen Abhighat (Birth Injury) With Special Referance To Erb's PalsyDokumen6 halamanA Clinical Study On Ayurvedic Approach in Prasav Kaleen Abhighat (Birth Injury) With Special Referance To Erb's PalsyAnonymous CwJeBCAXpBelum ada peringkat
- Jakarta School of Prosthetics & Orthotics: Upper Limb Prosthetics Course Work Manual 2010Dokumen84 halamanJakarta School of Prosthetics & Orthotics: Upper Limb Prosthetics Course Work Manual 2010Adinda Dinda100% (1)
- Anatomy of The Thoracic Wall, Pulmonary Cavities, and MediastinumDokumen26 halamanAnatomy of The Thoracic Wall, Pulmonary Cavities, and Mediastinumearly intervention centre thudiylu trerBelum ada peringkat
- Elective EntropyDokumen21 halamanElective EntropyDavidroy IRITSBelum ada peringkat
- Brachial Plexus Anesthesia: A Review of The Relevant Anatomy, Complications, and Anatomical VariationsDokumen12 halamanBrachial Plexus Anesthesia: A Review of The Relevant Anatomy, Complications, and Anatomical VariationsLucille IlaganBelum ada peringkat
- 10 Peripheral Nervous System Coloring BookDokumen10 halaman10 Peripheral Nervous System Coloring BookArmy JmBelum ada peringkat
- Gross HSB A - Axillary RegionDokumen3 halamanGross HSB A - Axillary RegionAngelo BautistaBelum ada peringkat
- 2018 EDX-SAE Exam-Booklet PDFDokumen59 halaman2018 EDX-SAE Exam-Booklet PDFMuhammad Imran MirzaBelum ada peringkat
- Anatomical Variation of The Brachial PlexusDokumen9 halamanAnatomical Variation of The Brachial Plexusjustinsanz77Belum ada peringkat