Eddy Gey2
Diunggah oleh
Junior LeivaJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Eddy Gey2
Diunggah oleh
Junior LeivaHak Cipta:
Format Tersedia
EJEMPLO DE CALCULOS
a) Calculo de las Tensiones Superficiales de las muestras:
El Etanol 100 v/v%:
A 20C : Utilizamos como referencia la densidad terica a 20C para del
Etanol y la altura experimental medida ; adems lo relacionamos con la
densidad terica del agua a 20C , su tensin superficial a 20C y su
altura experimental a esa temperatura mediante la siguiente expresin:
EtOH ( experimental) 20 C hEtOH 20 C EtOH ( teorico) 20 C
=
H 2 O (teorico ) 20 C
h H 2O 20 C H 2 O (teorico ) 20 C
Reemplazando:
EtOH ( experimental) 20 C
71.75 Dinas /cm
2.7 cm 0.7910 g/cm 3
5.2 cm 0.99829 g /cm3
EtOH (experimental )20 C =29.51 Dinas /cm
Porcentaje de Error:
%Error=
valor experimentalvalor teorico
22.7529.51
100=
100=29.71 ( Por exceso)
valor teorico
22.75
A 40C: Del mismo modo que en el caso anterior para la temperatura
de 40C:
EtOH ( experimental) 40 C h EtOH 40 C EtOH (teorico ) 40 C
=
H 2 O (teorico ) 40 C
h H 2 O 40 C H 2 O (teorico ) 40 C
Reemplazando:
EtOH ( experimental) 40 C
2.3 cm0.7744 g /cm 3
=
69.56 Dinas /cm 4.1 cm 0.99225 g /cm 3
EtOH (experimental ) 40 C =30.43 Dinas/cm
Porcentaje de Error:
%Error=
30.4320.60
100=47.71 (Por exceso)
20.60
A 50C: Del mismo modo que en el caso anterior para la temperatura
de 50C:
EtOH ( experimental) 50 C hEtOH 50 C EtOH (teorico ) 50 C
=
H 2 O (teorico ) 50 C
h H 2O 50 C H 2O (teorico )50 C
Reemplazando:
EtOH ( experimental) 50 C
67.91 Dinas /cm
2.38 cm 0.7670 g/cm3
7.2cm 0.98802 g/cm3
EtOH (experimental )50 C =17.43 Dinas / cm
Porcentaje de Error:
%Error=
17.4319.80
100=11.97
19.80
Solucin de Etanol al 10 v/v%: Significa que en 100ml de solucin
habr 10ml de alcohol puro, por lo que el volumen de agua ser de
90ml. Para el clculo de la densidad del Etanol al 10v/v% a la
temperatura de 20C se utilizara la siguiente expresin:
H 2 O (teorico ) 20 C Vomuen H 2O +
EtOH (teorico )20 C VolumenEtOH
Volumen total
masa
de H 2 O+masa de EtOH
C
20
=
EtOH(10 %V /V )=
Volumen total
0.99829 g
3 0.7910 g
3
90 cm +
10 cm
3
3
cm
cm
20 C
EtOH(10 %V /V )=
3
100 cm
C
3
20
EtOH(10 %V /V )=0.9776 g/cm
A 20C:Se calcula la Tensin superficial de la solucin de Etanol
10%v/v a la temperatura de 20C siguiendo el procedimiento
anterior:
C
C
20 C
20
h20
EtOH (10%V / V )
EtOH (10 %V /V ) EtOH(10 %V /V )
=
H 2O (teorico ) 20 C h H 2O 20 C H 2 O (teorico ) 20 C
20 C
EtOH (10%V / V )
2.65 cm 0.9776 g/cm3
=
71.75 Dinas /cm 7.9 cm 0.99829 g /cm3
20 C
EtOH (10%V / V ) =23.57 Dinas/cm
Solucin de Etanol al 50 v/v%: Significa que en 100ml de solucin
habr 50ml de alcohol puro, por lo que el volumen de agua ser de
50ml. Para el clculo de la densidad del Etanol al 50v/v% a la
temperatura de 20C se utilizara la siguiente expresin:
H 2 O (teorico ) 20 C Vomuen H 2O +
EtOH (teorico )20 C VolumenEtOH
Volumen total
masa
de H 2 O+masa de EtOH
C
20
=
EtOH(50 %V /V )=
Volumen total
0.98802 g
0.7670 g
50 cm3 +
50 cm3
3
3
cm
cm
C
20
EtOH(50 %V /V )=
3
100 cm
C
3
20
EtOH(50 %V /V )=0.8775 g/cm
A 20C:Se calcula la Tensin superficial de la solucin de Etanol
50%v/v a la temperatura de 20C siguiendo el procedimiento
anterior:
C
C
20 C
20
h20
EtOH (50%V / V )
EtOH (50 %V /V ) EtOH(50 %V /V )
=
H 2O (teorico ) 20 C h H 2O 20 C H 2 O (teorico ) 20 C
C
20
EtOH (50%V / V )
2.70 cm 0.8775 g /cm3
=
71.75 Dinas /cm 7.9 cm 0.99829 g /cm3
20 C
EtOH (50%V / V ) =21.56 Dinas/cm
b) Calculo del radio capilar:
Utilizando la siguiente expresin:
hgr
2
de donde :r=
2
hg
Utilizando los datos del Etanol 100%v/v:
A 20C:
r=
2 18.00 g/ s2
=0.01856 cm
2.5 cm 0.7910 g / cm 3 981 cm/s2
A 40C:
2
r=
2 17.85 g /s
=0.01958 cm
2.4 cm 0.7744 g /cm3 981cm/ s2
A 50C:
2 17.43 g/ s2
r=
=0.01947 cm
3
2
2.38 cm 0.7670 g /cm 981 cm/s
Luego:
Radio del Capilar=Radio promedio =
0.01856+0.01958+0.01947
=0.01920cm
3
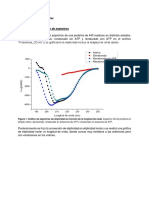
c) Calculo de valores para la grafica
2/ 3
( )
vs(t c 6 0t) para el
Etanol 100%v/v
t c =Temperatura criticadel Etanol=514 K , M =Peso molecular del Etanol=46,068 g/mol
A 20C:
2/ 3
( )
=( 18.00 )
46,068 3
=270 . 45 ( t c 60t ) =( 5146293 )=215 K
0.7910
K20C=270.45/215=1.26
A 40C:
2/ 3
( )
=( 17.85 )
46,068 32
=272. 02 ( t c 60 t ) =( 5146313 )=195 K
0.7744
K40C=272.02/195=1.39
A 50C:
2/ 3
( )
=( 17.43 )
46,068 23
=267 .32 ( t c 6 0t )=( 5146323 ) =185 K
0.7670
K50C=267.32/185=1.44
Anda mungkin juga menyukai
- Guía para La Prevención de Explosiones de Polvo de Carbón en Minería Subterránea y Limitación de Sus ConsecuenciasDokumen139 halamanGuía para La Prevención de Explosiones de Polvo de Carbón en Minería Subterránea y Limitación de Sus ConsecuenciasAnonymous Kvm4KxY4Belum ada peringkat
- Liter of Light México, Eval de Proy SocialesDokumen10 halamanLiter of Light México, Eval de Proy SocialesDavne Lopez OrozBelum ada peringkat
- PenduloDokumen11 halamanPenduloCésar Manuel Ramos SaldañaBelum ada peringkat
- HidroeléctricasDokumen7 halamanHidroeléctricasleoBelum ada peringkat
- Actividad 01 S3 Propiedades de Los Liquidos Ejercicios propuestos-TAREA 4Dokumen5 halamanActividad 01 S3 Propiedades de Los Liquidos Ejercicios propuestos-TAREA 4Ariel AlejandroBelum ada peringkat
- Calibraciones y Mapeos Térmicos - Laboratorio Nº48 Perteneciente A La Red INTI-SAC - Testo Argentina S.ADokumen3 halamanCalibraciones y Mapeos Térmicos - Laboratorio Nº48 Perteneciente A La Red INTI-SAC - Testo Argentina S.APepeBelum ada peringkat
- Mallas HHDokumen8 halamanMallas HHHector Quispe HuamanBelum ada peringkat
- Flujo LuminosoDokumen14 halamanFlujo LuminosoBetsy Karina Torres OrconBelum ada peringkat
- Incertidumbre CoriolisDokumen7 halamanIncertidumbre Coriolisjesus77777777777Belum ada peringkat
- Segundo GradoDokumen89 halamanSegundo GradoDilmer Hurtado100% (1)
- Conexiones A TierraDokumen15 halamanConexiones A TierraAshley CriolloBelum ada peringkat
- Contrato de ArrendamientoDokumen3 halamanContrato de ArrendamientoBaruchs BaruchsBelum ada peringkat
- Cap 33 Norma NFPA 704Dokumen3 halamanCap 33 Norma NFPA 704susana paliza uscapiBelum ada peringkat
- Cuadernillo CienciasNaturalesyEducacionAmbiental 9 1Dokumen14 halamanCuadernillo CienciasNaturalesyEducacionAmbiental 9 1Luis Alberto Cardozo GonzalezBelum ada peringkat
- TermoDokumen30 halamanTermoPauloCésarChoqueVasquezBelum ada peringkat
- Tarea 1. Transferencia de CalorDokumen9 halamanTarea 1. Transferencia de CalorOswaldo RiveraBelum ada peringkat
- Informe HojaDokumen11 halamanInforme Hojabrayan de hoyosBelum ada peringkat
- Resistencia Térmica Por Contacto 2Dokumen6 halamanResistencia Térmica Por Contacto 2David SalasBelum ada peringkat
- EXAMEN FINAL Adm Demanda y Plan de Compras ALCA PADILLADokumen42 halamanEXAMEN FINAL Adm Demanda y Plan de Compras ALCA PADILLAMishell Ap0% (1)
- Info Sheet Type 6 SpanishDokumen2 halamanInfo Sheet Type 6 SpanishRolando Antonio Cuenca PironaBelum ada peringkat
- Medina Samuel Diseño Protesis Tesis PDFDokumen123 halamanMedina Samuel Diseño Protesis Tesis PDFMiguel PintadoBelum ada peringkat
- Elvie-Pump Manual SpanishDokumen18 halamanElvie-Pump Manual SpanishAlejandro Gonzalez BenitezBelum ada peringkat
- Práctica 1 - RelevadoresDokumen6 halamanPráctica 1 - RelevadoresHand CorpBelum ada peringkat
- Pruebas A Transformadores de PotenciaDokumen16 halamanPruebas A Transformadores de PotenciaJean CarloBelum ada peringkat
- Anexo 7 Manual Tecnico Ptar Villa MartinDokumen12 halamanAnexo 7 Manual Tecnico Ptar Villa MartinEDUCAPP elevator peachBelum ada peringkat
- ColoidesDokumen17 halamanColoidesfrancisco alfonso galvanBelum ada peringkat
- CAP 3-Tipos de YacimientosDokumen8 halamanCAP 3-Tipos de YacimientosMayba MillaresBelum ada peringkat
- Tapa TapeiDokumen3 halamanTapa TapeiHarold EncantoBelum ada peringkat
- Dicroismo Circular s2018Dokumen8 halamanDicroismo Circular s2018Ariel Pinto MarchantBelum ada peringkat
- Diagramas de Ellingham para CO y CO2Dokumen5 halamanDiagramas de Ellingham para CO y CO2Jack QuispeBelum ada peringkat