Lipoidosis PDF
Diunggah oleh
Osiithaa CañaszJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Lipoidosis PDF
Diunggah oleh
Osiithaa CañaszHak Cipta:
Format Tersedia
www.medigraphic.org.
mx
Trabajo de revisin
Vol. 81, Nm. 4 Julio-Agosto 2014
pp 143-153
Enfoque simplificado para el diagnstico
de enfermedades lisosomales en nios
(Simplified diagnostic approach in the lysosomal storage diseases in children)
Luis Carbajal Rodrguez,* Raymundo Rodrguez Herrera,** Jorge Zarco Romn,**
Arturo Perea Martnez,** Alfonso Copto Garca***
RESUMEN
En el mundo se han identificado ms de 50 enfermedades por depsito lisosomal, las que aproximadamente ocurren como grupo
en 1 de 5,000 nacidos vivos. Muestran una patognesis comn, un defecto gentico en una o varias enzimas lisosomales espec-
ficas, dando lugar a una deficiente actividad enzimtica. Los lisosomas en los macrfagos tienen hidrolasas cidas que reducen las
macromolculas en pequeas partculas para ser recicladas o eliminadas del cuerpo; si no se eliminan cabalmente se acumulan
interfiriendo en la actividad celular normal a diferentes niveles, pudiendo ocasionar una posible muerte celular. Los tipos diferentes
de sustratos acumulados en diversas clulas del cuerpo producen alteraciones en diferentes rganos con gran variabilidad en la
expresin clnica. La evolucin de estas enfermedades es crnica y progresiva, depende de la cantidad de sustrato acumulado, as
como tambin su pronstico es con relacin al tiempo en que se hace el diagnstico y se inicia el tratamiento; es por eso necesario
que haya un entendimiento adecuado de estas enfermedades: consideradas raras, progresivas, incurables y mortales.
Palabras clave: Lisosomas, enzimas, lisosomales, depsito lisosomal.
SUMMARY
There are more than 50 lysosomal steerage disorder in the world. The incidence is approximately 1 in every 5,000 newborns. Lysosomal
steerage disorder shows a common pathogenetic pattern: a genetic defect in one or various specific lysosomal enzyme which leads
to deficient enzymatic activities. Lysosomas in macrophages contain hydrolase acids that act on macro molecules to reduce them to
small particles which are recycled or eliminated in the body. When the elimination of the substrate is not adequate, they are progres-
sively accumulated in the cell, thereby interfere with the normal cellular activities at different levels, the outcome of which may be
cell death. The different kinds of substrates accumulated in diverse cells of the body produce alterations in many organs with great
variability in their clinical expression. The evolution is chronic and progressive depending on the quantity of accumulated substrates as
well as its prognosis in relation to the time of its diagnosis and treatment. It is therefore necessary that there is adequate understand-
ing of these disorders considered rare, progressive, incurable, and mortal.
Key words: Lysosomes, enzymes, lysosomal, storage.
Hay actualmente identificadas en el mundo ms de 50 estas enfermedades sean conocidas en cualquiera de
enfermedades por depsito lisosomal, y stas ocurren las disciplinas mdicas, en nios y en adultos; es por
aproximadamente como grupo en 1 de 5,000 nacidos eso que el objetivo principal de este estudio es conocer
vivos (Cuadro 1), por lo que hay la necesidad de que estas enfermedades y de modo prctico estar prepara-
www.medigraphic.org.mx dos para hacer el diagnstico correcto y el tratamientos
oportuno de estos nios.
* Dpto. de Medicina Interna. Jefe de la Clnica de Enfermedades
En general, este grupo de enfermedades tiene un
por Depsito Lisosomal, Instituto Nacional de Pediatra. defecto gentico en una o varias de las enzimas liso-
** Adscrito al Dpto. de Medicina Interna, Instituto Nacional de Pe- somales especficas: protenas activadoras o protenas
diatra. de membrana, dando lugar a una deficiente actividad
*** Hospital ngeles Mxico.
enzimtica.
Este artculo puede ser consultado en versin completa en Los lisosomas en los macrfagos contienen hidro-
http://www.medigraphic.com/rmp lasas cidas, que transforman las macromolculas en
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 1. Enfermedades por depsito lisosomal.
LSDs: Clasificacin
Esfingolipidosis Glucogenosis
Gaucher (beta-glicosidasa) Pompe (tipo 2)
Krabbe (galactocerebrosidasa) Oligosacaridosis
Leucodistrofia met. (arisulfatasa A) Alfa-manosidosa
Niemann-Pick tipo A y B (esfingomielinasa) Beta-manosidosa
Fabry (alfa-galactosidasa) Fucosidosa
Gangliosidosis GM1 (beta-galactosidasa) Sialidosa
Farber (ceramidasa) Galactosialidosa
Tay-Sachs (hexosaminidasas B) Aspartilglucosaminuria
Sandhoff (hexosaminidasas A y B) cido silico
Schindler
Mucopolisacaridosis Mucolipidosis
Tipos I, II, III, IV, VI y VII Glucoproteinosis
CDG
pequeas partculas que pueden ser recicladas o elimi- Cuadro 2. Modalidades teraputicas.
nadas del cuerpo; si stas no son desechadas adecua-
damente, el sustrato se acumula de manera progresiva, Terapia de reemplazo enzimtico (Gaucher tipo I, MPS I,
interfiriendo en la actividad celular normal a diferentes II y VI, Fabry y Pompe)
niveles, dando como resultado la posible muerte celular
Terapia farmacolgica (enfermedad de Gaucher, cistinosis,
y una multiplicidad de signos y sntomas en el enfermo. enfermedad de Nieman-Pick tipo C)
La primera EDL fue informada en 1955 por De Duve
C y cols., quienes la dieron a conocer como enferme- Trasplante de clulas madre hematopoyticas (MPS I,
dad de Pompe o glucogenosis II; con el transcurso del II y VI)
tiempo se empezaron a identificar en grupos de enfer-
mos por su dficit enzimtico y su sustrato acumulado; Trasplante de rganos (enfermedad de Fabry)
ms tarde se describieron sus particularidades clnicas,
aunque no ha sido posible llegar a una estandarizacin Terapia de reemplazo gentico (No disponible)
de stas, a pesar de que comparten bases patognicas;
sin embargo, los diferentes tipos de sustratos acumula-
dos en las diversas clulas del cuerpo dan como resulta- enzima glucocerebrosidasa y el desarrollo de enfer-
do alteraciones en diferentes rganos y con una amplia medades mieloproliferativas, as como enfermedad de
variabilidad en la expresin clnica. En la gran mayora Parkinson; estos factores epigenticos y del medio am-
de estos nios la evolucin es crnica y progresiva, de- biente juegan un papel en el fenotipo, como es el caso
pendiendo de la cantidad de sustrato acumulado y su de las enfermedades de Gaucher y Fabry.
pronstico depender del tiempo de su diagnstico y Es necesario que haya entendimiento de estas ra-
tratamiento, ya que en un principio los datos clnicos ras enfermedades de curso progresivo, incurables y
pueden pasar inadvertidos o confundirse con otras en- mortales.1-16
fermedades.
En la pasada dcada ocurri un progreso significa- CLASIFICACIN
www.medigraphic.org.mx
tivo en cuanto al entendimiento de la fisiopatologa de
estas enfermedades, particularmente con relacin a las
Estas enfermedades se han clasificado en cuatro gran-
tcnicas de rastreo en el recin nacido, obteniendo des grupos, dependiendo de la alteracin en la macro-
en papel filtro una gota de sangre para identificar una molcula afectada, siendo reconocida como: I. Esfingo-
de estas enfermedades: Fabry, Gaucher, Niemann Pick, lipidosis, II. Mucopolisacaridosis, III. Glucogenosis y IV.
Pompe, Sandhoff y Tay-Sachs. Glucoproteinosis.
En algunos casos, como en la enfermedad de Gau- Se describen algunas de ellas con las que ya se cuen-
cher, hay una marcada correlacin genotipo fenotipo, ta con tratamiento de reemplazo enzimtico (Cuadros
adems de una estrecha unin entre mutaciones de la 2 a 9).
Rev Mex Pediatr 2014; 81(4); 143-153 144
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 3. Enfermedad de Gaucher.
Glucocerebrosidasa
Enzima deficiente
Glucosidasa cida (catabolismo de la glucosilceramida)
Macromolcula afectada Esfingolpidos (catabolismo de los glucocerebrsidos)
GBA + de 300 mutaciones
Gen mutado
N370S, L444P, 84GG y EIUS 2+1
Cromosoma 1 (q-21-31)
Tipo de herencia Autosmica recesiva
Tipo I Tipo II Tipo III
Etnia Pantnica Pantnica Pantnica
Judos ashkenazi polacos, suecos y rabes
Incidencia Judos ashkenazi 1:1,000 1:200,000 1:100,000
Judos ashkenazi
1:450 Poblacin general
Prevalencia
Poblacin general 1:100,000 1:50,000
1:40,000 a 1:60,000
Judos ashkenazi 1:14
Frecuencia de portadores
Poblacin general 1:500
No neuronoptico Neuronoptico agudo Neuronoptico subagudo
Tipos clnicos
(subtipos a, b, c)
% de presentacin 92% 1% 7%
rganos afectados Bazo, hgado, mdula sea, huesos y sistema nervioso central
Determinacin de la actividad enzimtica en leucocitos de sangre perifrica o en cultivo de fibro-
blastos (lecturas < de 305 de actividad)
Diagnstico Estudio molecular de las mutaciones as como de quitotriosidasa y fosfatasa cida
Diagnstico citomorfolgico en biopsias de mdula sea, ganglios o hgado (macrfagos cargados
con glucocerebrsidos)
Pronstico Tipo I: bueno Tipo II: malo Tipo III: reservado
Tratamiento
www.medigraphic.org.mx
Terapia de reemplazo enzimtico con imiglucerasa, velaglucerasa alfa o taligluerasa alfa, cada 15
das toda la vida
Referencias: 17-30
145 Rev Mex Pediatr 2014; 81(4); 143-153
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 4. Enfermedad de Fabry.
Enzima deficiente Alfa galactosidasa A
Macromolcula afectada Esfingolpidos (catabolismo de la globotriaosilceramida GL3)
Alfa galactosidasa A
Gen mutado
ms de 160 mutaciones
Cromosoma Xq 22
Tipo de herencia Recesivo ligado al cromosoma X
Etnia Pantnica
Hombres 1:40,000-476,000
Incidencia
Mujeres 1:100,000
Prevalencia 1:5,000
Los hijos varones tienen 50% de probabilidad de que padezcan la enfermedad y las hijas
Frecuencia de portadores
50% de ser portadoras
Tipos clnicos Manifestaciones tempranas <35 aos y tardas >35 aos
Este documento
Cerebroes elaborado
(accidente por Medigraphic
cerebrovascular)
Corazn (miocarditis, endocarditis)
rganos afectados
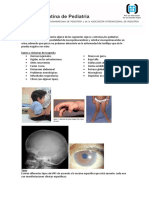
Piel (angioqueratomas, hipo o anhidrosis)
Ojos (opacidad corneal)
Determinacin de la actividad enzimtica en plasma, leucocitos u otros grupos celulares
Diagnstico o en gotas de sangre seca en papel filtro
Estudio molecular de las mutaciones
Pronstico Reservado a evolucin
Terapia de reemplazo enzimtico con tcnica de DNA recombinante agalsidasa alfa y/o
Tratamiento
beta, catorcenalmente toda la vida
Referencias: 31-48 www.medigraphic.org.mx
Rev Mex Pediatr 2014; 81(4); 143-153 146
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 5. Mucopolisacaridosis tipo I (enfermedad de Hurler).
Enzima deficiente L, iduronidasa
Mucopolisacridos o glicosaminoglicanos
Macromolcula afectada
(heparn y dermatn sulfato)
Gen mutado W402X q70X P533R
Cromosoma 4locus 4P 16.3
Tipo de herencia Autosmica recesiva
Etnia Pantnica
Forma severa Forma intermedia Forma atenuada 1:500,000
Incidencia
1:100,000 a 1: 280,000 recin nacidos vivos
Frecuencia de portadores 1/160
Forma severa Forma intermedia Forma atenuada
Tipos clnicos
Sndrome de Hurler Sndrome de Hurler-Scheie Sndrome de Scheie
% de presentacin 50-80% 20% 10%
Sistema nervioso central (retraso psicomotor con hidrocefalia, mielopata cervical progresiva,
con compresin medular)
Sistema nervioso perifrico (compresin de nervios y tendones)
Huesos (disostosis mltiple, miopata, talla baja)
rganos afectados Ojos (opacidad corneal, glaucoma atrofia ptica y ceguera)
ONG (obstruccin de la va area, otitis media, prdida de la audicin, SOAS)
Pulmones (atelectasias, cor pulmonale)
Corazn (displasia valvular, cardiomiopata, fibroelastosis)
Tubo digestivo (hepatoesplenomegalia, hernias)
Determinacin de la actividad enzimtica en leucocitos plasma y cultivo de fibroblastos. Estu-
Diagnstico
dio molecular de las mutaciones
Pronstico Reservado al tipo de presentacin clnica
Terapia de reemplazo enzimtico: L-iduronidasa recombinante cada semana toda la vida
Tratamiento
www.medigraphic.org.mx
Trasplante de mdula sea o de clulas de cordn umbilical
Referencias: 49-59
147 Rev Mex Pediatr 2014; 81(4); 143-153
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 6. Mucopolisacaridosis tipo II (enfermedad de Hunter).
Enzima deficiente Iduronato 2 sulfatasa
Macromolcula afectada Mucopolisacridos o glicosaminoglicanos (heparn, dermatn, keratn y condroitin sulfato)
Gen mutado 12s pseudogen 12S2, IQSP1
Cromosoma Xq28
Tipo de herencia Recesiva ligada al X
Etnia Pantnica
Incidencia 1:170,000 recin nacidos masculinos
Prevalencia 3.4 a 3.5: 100,000 en judos ashkenazi y sefardes es el doble
Forma severa Forma atenuada
Tipos clnicos
< 2 a 4 aos >4 aos
Sistema nervioso central (retraso psicomotor)
Musculoesqueltico: (facies tosca macrocefalea, disostosis mltiple)
Sistema nervioso: (compresin del nervio ptico). ONG: (obstruccin de la va area, prdida de
rganos afectados
la audicin)
Gastrointestinal: (hepatomegalia, hernias inguinales y umbilicales, diarrea crnica)
Cardiovascular: (valvulopata)
Determinacin enzimtica en cultivo de fibroblastos, leucocitos, plasma, lquido amnitico
Diagnstico Determinacin de glicosaminoglicanos en orina (pueden aparecer en MPS I y VII)
Anlisis de las mutaciones
Pronstico Dependiendo del tipo de presentacin
Tratamiento Terapia de reemplazo enzimtico con idursulfasa recombinante cada semana toda la vida
Referencias: 60-69
Cuadro 7. Mucopolisacaridosis VI (sndrome de Maroteaux-Lamy).
Enzima deficiente n-acetilgalactosamina-4-sulfatasa (arilsulfatasa B)
Macromolcula afectada Glicosaminoglicanos (dermatn sulfato)
Y210c (10%)
Gen mutado
80 mutaciones
Cromosoma Gen ARSB en cromosoma 5
Tipo de herencia Autosmica recesiva
Pantnica
Etnia
Brasil y Portugal
Incidencia 1:238-1:300,000
Tipos clnicos Aguda y crnica
Musculoesqueltico (disostosis mltiple, artropata crnica)
ONG (obstruccin de las vas areas superiores, otitis crnica, hipoacusia)
rganos afectados Gastrointestinal (hepatoesplenomegalia)
www.medigraphic.org.mx
Cardiovascular (enfermedad cardiopulmonar)
Sistema nervioso (compresin espinal, neuropatas, ceguera, funcin intelectual normal)
Determinacin de la enzima en leucocitos, fibroblastos
Diagnstico
Glicosaminoglicanos en orina, en cromatografa, en capa fina o en electroforesis
Pronstico Reservado a su evolucin
Terapia de reemplazo enzimtico con n-acetilgalactosamina 4 sulfatasa
Tratamiento
Cada semana toda la vida
Referencias: 70-75
Rev Mex Pediatr 2014; 81(4); 143-153 148
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 8. Glucogenosis tipo II (enfermedad de Pompe).
Enzima deficiente Alfa glucosidasa alfa o maltasa cida
Macromolcula
Glucgeno
afectada
IVS-1 en caucsicos
Asp645 glu en Taiwan
Gen mutado
Avg 854X en africanos
140 mutaciones
Cromosoma 17q25.2-q25.3
Tipo de herencia Autosmica recesiva
Etnia Pantnica
Incidencia 1:40,000-1:300,000
Prevalencia 5,000-10,000
Infantil clsico Infantil juvenil
Tipos clnicos (rpidamente Infantil no clsico (aparicin tarda)
progresivo) adulto
Corazn (cardiomiopata)
Musculoesqueltico (debilidad muscular proximal grave y progre-
rganos afectados siva)
Pulmn (insuficiencia respiratoria e infecciones)
Gastrointestinal (hepatoesplenomegalia, macroglosia)
Determinacin de la actividad enzimtica en fibroblastos, msculo,
leucocitos
Amniocentesis en vellosidades corinicas. Actividad de la enzima en
Diagnstico papel filtro y anlisis por fluorometra o espectrometra de masas
Anlisis de las mutaciones. Creatincinasa, aspartasa, alanino ami-
notransferasa, deshidrogenasa lctica y glucosa tetrasacrida, estn
elevadas
Pronstico Reservado a evolucin
Tratamiento www.medigraphic.org.mx
Terapia de reemplazo enzimtico con alfa glucosidasa cida huma-
na recombinante
Cada dos semanas toda la vida
Referencias: 76-89
149 Rev Mex Pediatr 2014; 81(4); 143-153
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Cuadro 9. Enfermedad de NIEMANN PICK Tipo C.
Enzima deficiente Alteracin intracelular en el transporte de lpidos
Macromolcula
Esfingolpidosis
afectada
NPC1 (95%)
Gen mutado
NPC2 (5%)
NPC1: 18q11-q12
Cromosoma
NPC2: 14q 24-3
Tipo de herencia Autosmica recesiva
Etnia Pantnica
Incidencia 1:150,000
Forma infantil temprana Forma tarda
Tipos clnicos Forma juvenil Forma adulta
<2 aos 2-6 aos
6-15 aos >16 aos
Hgado (colestasis neonatal, hepatoesplenomegalia)
rganos afectados Sistema nervioso (parlisis supranuclear de la mirada vertical, ataxia, disartria, trastornos cognitivos,
cataplexia, epilepsia mioclnica, distona, sntomas neurosiquitricos)
Bsqueda de acmulo de colesterol libre no esterificado en fibroblastos mediante tcnica de la Filipi-
Diagnstico
na. Secuencia de los genes
Pronstico Reservado a evolucin
Terapia de reduccin de sustrato, impidiendo su sntesis
Tratamiento
Miglustat
Referencias: 90-94
Referencias 8. Wenger DA, Coppola S, Liu SL. Insights into the diagnosis
and treatment of lysosomal storage diseases. Arch Neurol.
1. Meikle PJ, Ranieri E, Simonsen H. Newborn screening for 2003; 60: 322-328.
lysosomal storage disorders: clinical evaluation of a two-tier 9. Marsden D, Levy H. Newborn screening of lysosomal stora-
strategy. Pediatrics. 2004; 114: 909-916. ge disorders. Clin Chem. 2010; 56: 1071-1079.
2. Wilcox WR. Lysosomal storage disorders: the need for bet- 10. Futerman AH, van Meer G. The cell biology of lysosomal sto-
ter pediatric recognition and comprehensive care. J Pediatr. rage disorders. Nat Rev Mol Cell Biol. 2004; 5: 554-565.
2004; 144: S3-14. 11. Beck M. New therapeutic options for lysosomal storage
3. Zetina ME, Gonzlez NA. Enfermedades lisosomales heredi- disorders: enzyme replacement, small molecules and gene
tarias I. los resultados iniciales de un programa de diagnosti- therapy. Hum Genet. 2007; 121: 1-22.
4. www.medigraphic.org.mx
co en Mxico. Rev Invest Clin. 1989; 41: 319-326.
Zetina ME, Gonzlez Noriega A. Hereditary lysosomal disea-
12. Sidransky E, Nalls MA, Aasly JO. Multicenter analysis of glu-
cocerebrosidase mutations in Parkinsons disease. N Engl J
ses in Mexico II. Laboratory diagnosis of mucopolysacchari- Med. 2009; 361: 1651-1661.
dosis and mucolipidosis. Rev Invest Clin. 1990; 42: 165-173. 13. Costello R, OCallaghan T, Sbahoun G. Gaucher disease and
5. Meikle PJ, Hopwood JJ, Clague AE. Prevalence of lysosomal multiple myeloma. Leuk Lymphoma. 2006; 47: 1365-1358.
storage disorders. JAMA. 1999; 281: 249-254. 14. Barba-Romero MA, Deegan P, Giugliani R, Hughes D. Does
6. de Duve C, Pressman B, Gianetto R. Tissue fractionation stu- geographical location influence the phenotype of Fabry disea-
dies. Intracellular distribution patterns of enzymes in rat liver se in women in Europe? Clin Genet. 2010; 77: 131-140.
tissue. Biochem J. 1955; 60: 604-617. 15. Elstein D, Gellman A, Altarescu G. Disease severity in sibling
7. Hers HG. Alpha-glucosidase deficiency in generalized glycogens- pairs with type 1 Gaucher disease. J Inherit Metab Dis. 2010;
torage disease (Pompes disease). Biochem J. 1963; 86: 11-16. 33: 79-83.
Rev Mex Pediatr 2014; 81(4); 143-153 150
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
16. Beutler E, Grabowski G. Gaucher disease. In: Scriver C, Beau- 35. Alroy J, Sabnis S, Koop JB. Renal pathology in Fabry disease.
det A, Sly W, Valle D (eds). The metabolic and molecular bases J Am Soc Nephrol. 2002; 13: 134-138.
of inherited disease. 8th ed. New York, New York: McGraw 36. Branton M, Schiffmann R, Sabnis S, Murray GJ, Quirk JM, Al-
Hill; 2001: 3635-3668. tarescu G. Natural history of Fabry renal disease: influence of
17. Zimran A, Gelbart T, Westwood B. High frequency of the alpha-galactosidase activity and genetic mutations on clinical
Gaucher disease mutation at nucleotide 1,226 among Ashke- course. Medicine. 2002; 81: 122-138.
nazi Jews. Am J Hum Genet. 1991; 49: 855-859. 37. Ortiz A, Oliveira JP, Waldek S, Warnock DG, Cianciarus B,
18. Grabowski GA. Gaucher disease. Enzymology, genetics, and Wanner C. Fabry Registry. Nephropathy in males and fema-
treatment. Adv Hum Genet. 1993; 21: 377-441. les with Fabry disease: cross-sectional description of patients
19. Charrow J, Andersson HC, Kaplan P. Enzyme replacement before treatment with enzyme replacement therapy. Nephrol
therapy and monitoring for children with type 1. Gaucher Dial Transplant. 2008; 23: 1600-1607.
disease: consensus recommendations. J Pediatr. 2004; 144: 38. Ortiz A, Oliveira JP, Wanner C, Brenner BM, Waldek S, War-
112-120. nock DG. Recommendations and guidelines for the diagnosis
20. Maas M, Poll LW, Terk MR. Imaging and quantifying skeletal and treatment of Fabry nephropathy in adults. Nat Clin Pract
involvement in Gaucher disease. Br J Radiol. 2002; 75: A13- Nephrol [Revista en lnea]. 2008; 4: 327-336.
24. 39. Ruiz-de Gauna R, Minguela-Pesquera I, Ocharan-Corcuera
21. Kaplan P, Mazur A, Manor O. Acceleration of retarded J, Gimeno-Martin I, Chenna AA. El entorno social de los pa-
growth in children with Gaucher disease after treatment cientes en dilisis peritoneal. Nefrologa. 2008; 28: 133-136.
with alglucerase. J Pediatr. 1996; 129: 149-153. 40. Mitsias P, Levine SR. Cerebrovascular complications of
22. Zimran A, Altarescu G, Rudensky B. Survey of hematological Fabrys disease. Ann Neurol. 1996; 40: 8-17.
aspects of Gaucher disease. Hematology. 2005; 10: 151-156. 41. Sher NA. Letson RD, Desnick RJ. The ocular manifestations
23. Aker M, Zimran A, Abrahamov A. Abnormal neutrophil che- in Fabrys disease. Arch Opthalmol. 1979; 97: 671-676.
motaxis in Gaucher disease. Br J Haematol. 1993; 83: 187- 42. Nguyen TT, Gin Tm Nicholls K, Low M, Galanos J, Crawford
191. A. Ophthalmological manifestations of Fabry disease: a
24. Miller A, Brown LK, Pastores GM. Pulmonary involvement survey of patients at the Royal Melbourne Fabry Disease
in type 1 Gaucher disease: functional and exercise findings in Treatment Centre. Clin Experiment Ophthalmol. 2005; 33:
patients with and without clinical interstitial lung disease. Clin 164-168.
Genet. 2003; 63: 368-376. 43. Ries M, Gupta S, Moore DF, Sachdev V, Quirk JM, Murray GJ.
25. Alterescu G, Hill S, Wiggs E. The efficacy of enzyme re- Pediatric Fabry disease. Pediatrics. 2005; 115: 344-355.
placement therapy in patients with chronic neuronopathic 44. Palla A, Hegemann S, Widmer U, Straumann D. Vestibular and
Gauchers disease. J Pediatr. 2001; 138: 539-47. auditory deficits in Fabry disease and their response to enzy-
26. Campbell P, Harris C, Vellodi A. Deterioration of the auditory me replacement therapy. J Neurol. 2007; 254: 1433-1442.
brainstem response in children with type 3 Gaucher Disease. 45. Desnick RJ, Brady RO. Fabry disease in childhood. J Pediatr.
Neurology. 2004; 63: 385-7. 2004; 144: 20-26.
27. Bembi B, Zambito MS, Sidransky E. Gauchers disease with 46. Gelb MH, Turecek F, Scott CR, Chamoles NA. Direct multi-
Parkinsons disease: clinical and pathological aspects. Neuro- plex assay of enzymes in dried spots by tandem mass spec-
logy. 2003; 61: 99-101. trometry for the newborn screening of lysosomal storage
28. Tylki SA, Vellodi A, El Beshlawy A, Cole JA, Kolodny E. disorders. J Inherit Metab Dis. 2006; 29: 397-404.
Neuronopathic Gaucher disease: demographics and clini- 47. Garman SC, Garboczi DN. The molecular defect leading to
cal features in 141 patients enrolled in the ICGG Gaucher Fabry disease: structure of human alpha-galactosidase. J Mol
Registry. J Inherited Metabol Dis. 2010 Jan 19[Epub ahead of Biol. 2004; 337: 319-335.
print]. 48. Ries M, Clarke JT, Whybra C, Timmons M, Robinson C,
29. Beutler E, Kuhl W, Trinidad F. Beta-glucosidase activi- Schlaggar BL. Enzyme-replacement therapy with agalsidase
ty in fibroblasts from homozygotes and heterozygotes for alfa in children with Fabry disease. Pediatrics. 2006; 118: 924-
Gauchers disease. Am J Hum Genet. 1971; 23: 62-66. 932.
30. Carbajal RL, Voirol GA, Mora MI, Rodriguez HR, Zarco RJ. 49. Neufeld EF, Muenzer J. The Mucopolysaccharidoses. In: Scri-
Gaucher disease in Mexico. Epidemiologic overview. Act Pe- ver C, Beaudet A, Sly W, Valle D (eds). The metabolic and
diatr Mex. 2011; 32: 277-280. molecular bases of inherited disease. 8th ed. New York, New
31. Desnick R, Ioannou Y, Eng C. Alpha-Galactosidase A deficien- York: McGraw-Hill; 2001; 3421-3452.
cy: Fabry disease. In: Scriver CR BA, Sly WS, Valle D (eds). 50. Vijay S, Wraith JE. Clinical presentation and follow-up of pa-
The metabolic and molecular bases of inherited disease, 8th Ed. tients with the attenuated phenotype of mucopolysacchari-
New York, New York: McGraw-Hill; 2001. pp. 3733-3774. dosis type I. Acta Paediatr. 2005; 94: 872-877.
32. Radillo P. Grupo Mexicano de Especialistas en Enfermedad de 51. Grewal SS, Wynn R, Abdenur JE. Safety and efficacy of enzy-
Fabry. Gua para el diagnostico, evaluacin y tratamiento de me replacement therapy in combination with hematopoietic
505-514. www.medigraphic.org.mx
la enfermedad de Fabry. Rev Med Inst Mex Seg Soc. 2009; 5: stem cell transplantation in Hurler syndrome. Genet Med.
2005; 7: 143-146.
33. Desnick RJ, Brady R, Barranger J, Collins AJ, Germain DP, 52. Wraith JE. The first 5 years of clinical experience with laroni-
Goldman M. Fabry disease, an under-recognized multisyste- dase enzyme replacement therapy for mucopolysaccharido-
mic disorder: expert recommendations for diagnosis, mana- sis I. Expert Opin Pharmacother. 2005; 6: 489-506.
gement, and enzyme replacement therapy. Ann Intern Med. 53. Kachur E, Del Maestro R. Mucopolysaccharidoses and spinal
2003; 138: 338-346. cord compression: case report and review of the literature
34. Wang RY, Lelis A, Mirocha J, Wilcox WR. Heterozygous Fabry with implications of bone marrow transplantation. Neurosur-
women are not just carriers but have a significant burden of gery. 2000; 47: 223-228.
disease and impaired quality of life. Genet Med. 2007; 9: 34- 54. Neufeld EF, Muenzer J. The mucopolysaccharidosis. In: Scri-
45. ver GR, Beaudet AL, Sly WS, Valle D, Childs B, Kinzler KW,
151 Rev Mex Pediatr 2014; 81(4); 143-153
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
Vogelstein B (eds.). The metabolic and molecular bases of 73. Poorthuis B, Wevers R, Kleijer W. The frequency of lysoso-
inherited disease. 8th edition, Vol. III. McGrawHill. Medical mal storage diseases in the Netherlands. Hum Genet. 1999;
Publishing Division, 2001; 3421. 105: 151-156.
55. Clarke LA, Portigal C. Mucopolysaccharidosis type I. Genet 74. Whitley CB, Ridnour MD, Draper KA, Dutton CM, Neglia JP.
Reviews. 2002Available at www.genetests.org Diagnostic test for mucopolysaccharidosis. I. Direct method
56. Terlato NJ, Cox GF. Can mucopolysaccharidosis type I disea- for quantifying excessive urinary glycosaminoglycan excre-
se severity be predicted based on a patients genotype? A tion. Clin Chem. 1989; 35: 374-379.
comprehensive review of the literature. Genet Med. 2003; 5: 75. McGill JJ, Inwood AC, Coman DJ, Lipke ML, de Lore D,
286-294. Swiedler SJ et al. Enzyme replacement therapy for muco-
57. Hall CW, Liebaers I, Di Natale P. Enzymatic diagnosis of the polysaccharidosis VI from 8 weeks of age-a sibling control
genetic mucopolysaccharide storage disorders. Methods En- study. Clin Genet. 2010; 77(5): 492-498.
zymol. 1978; 50: 439-456. 76. DAncona GG, Wurm J, Croce CM. Genetics of type II glyco-
58. Terlato N, Cox G. Can mucopolysaccharidosis type I disea- genosis: assignment of the human gene for acid alpha-gluco-
se severity be predicted based on a patients genotype? A sidase to chromosome 17. Proc Natl Acad Sci USA. 1979; 76:
comprehensive review of the literature. Genet Med. 2003; 5: 4526-4529.
286-294. 77. Hirschhorn R, Reuser AJJ. Glycogen storage disease type II:
59. Clarke LA, MacFarland J. La Mucopolisacaridosis-I (MPS-I). acid alpha-glucosidase (acid maltase) deficiency. In: Scriver C,
In: Clarke LA, Kaweski C, Di Ilio L, Hahn S (eds). La Sociedad Beaudet A, Sly W et al. The metabolic and molecular bases of
Canadiense de Mucopolisacaridosis y Enfermedades Relacio- inherited disease. New York: McGraw Hill, 2001: 3389-3420.
nadas, Inc., Vancouver: Grfica e Imprenta Ticky, 2001. 78. Kishnani PS, Steiner RD, Bali D. Pompe disease diagnosis and
60. Martin R, Beck M, Eng C, Giugliani R, Harmatz P, Muoz V. management guideline. Genet Med. 2006; 8: 267-288.
Recognition and diagnosis of mucopolysaccharidosis II (Hun- 79. Kuo WL, Hirschhorn R, Huie ML, Hirschhorn K. Localization
ter Syndrome). Pediatrics. 2008; 121: 377-386. and ordering of acid alpha-glucosidase (GAA) and thymidine
61. Baehner F, Schmiedeskamp C, Krummenauer F. Cumulative kinase (TK1) by fluorescence in situ hybridization. Hum Ge-
incidence rates of the mucopolysaccharidoses in Germany. J net. 1996; 97: 404-406.
Inherit Metab Dis. 2005; 28: 1011-1017. 80. Becker JA, Vlach J, Raben N. The African origin of the com-
62. Applegarth DA, Toone JR, Lowry RB. Incidence of inborn mon mutation in African American patients with glycogen-
errors of metabolism in British Columbia, 1969-1996. Pe- storage disease type II. Am J Hum Genet. 1998; 62: 991-994.
diatrics. 2000; 105(1). Available at: www.pediatrics.org/cgi/ 81. Ausems MG, Verbiest J, Hermans MP. Frecuency of glycogen
content/full/105/1/e10 storage disease type II in The Netherlands: implications for
63. Bach G, Eisenberg F Jr, Cantz M, Neufeld EF. The defect in diagnosis and genetic counseling. Eur J Hum Genet. 1999; 7:
the Hunter syndrome: deficiency of sulfoiduronate sulfatase. 713-716.
Proc Natl Acad Sci USA. 1973; 70: 2134-2138. 82. Howell RR, Byrne B, Darras B, Kishnani P, Nicolino M, van
64. Young ID, Harper PS. Incidence of Hunters syndrome. Hum der Ploeg A. Diagnostic challengefor Pompe disease: an un-
Genet. 1982; 60: 391-392. der-recognized cause of floppy baby syndrome. Genet Med.
65. Zlotogora J, Schaap T, Zeigler M, Bach G. Hunter syndrome 2006; 8: 289-296.
in Jews in Israel: further evidence for prenatal selection favo- 83. Li Y, Scott CR, Chamoles NA. Direct multiplex assay of lyso-
ring the Hunter allele. Hum Genet. 1991; 86: 531-533. somal enzymes in dried blood spots for newborn screening.
66. Li P, Bellows AB, Thompson JN. Molecular basis of iduronate- Clin Chem. 2004; 50: 1785-1796.
2-sulphatase gene mutations in patients with mucopolysac- 84. Meikle PJ, Grasby DJ, Dean CJ. Newborn screening for ly-
charidosis type II (Hunter syndrome). J Med Genet. 1999; 36: sosomal storage disorders. Mol Genet Metab. 2006; 88: 307-
21-27. 314.
67. Lin SP, Chang JH, Lee-Chen GJ, Lin DS, Lin HY, Chuang CK. 85. Young SP, Stevens RD, An Y, Chen YT, Millington DS. Analysis
Detection of Hunter syndrome (mucopolysaccharidosis type of a glucose tetrasaccharide elevated in Pompe disease by
II) in Taiwanese: biochemical and linkage studies of the iduro- stable isotope dilution-electrospray ionization tandem mass
nate-2-sulfatase gene defects in MPS II patients and carriers. spectrometry. Anal Biochem. 2003; 316: 175-180.
Clin Chim Acta. 2006; 369: 29-34. 86. Baethmann M. Pompe disease. 1st ed. Bremen: UNI-MED:
68. Dean CJ, Bockmann MR, Hopwood JJ, Brooks DA, Meikle PJ. Verlag AG, D 2008; pp. 11-102.
Detection of mucopolysaccharidosis type II by measurement 87. Kishnani PS, Corzo D, Nicolino M, Byrne B, Mandel H, Hwu
of iduronate-2-sulfatase in dried blood spots and plasma WI. Recombinant human acid (alpha)-glucosidase: major cli-
samples. Clin Chem. 2006; 52: 643-649. nical benefits in infantile-onset Pompe disease. Neurology.
69. Muenzer J, Wraith JE, Beck M. A phase II/III clinical study of 2007; 68: 99-109.
enzyme replacement therapy with idursulfase in mucopolysac- 88. Van Capelle CI, Winkel LPF, Hagemans MLC, Shapira SK,
charidosis II (Hunter syndrome) [published correction appears Arts WFM, van Doorn PA. Eight years experience with enzy-
70. www.medigraphic.org.mx
in Genet Med. 2006; 8: 599]. Genet Med. 2006; 8: 465-473.
Decker C, Yu ZF, Giugliani R, Schwartz IV, Guffon N, Teles
me replacement therapy in two children and one adult with
Pompe disease. Neuromuscul Disord, in press. 2008.
EL. Enzyme replacement therapy for mucopolysaccharido- 89. Van der Ploeg AT, Marsden DL. Response to treatment with
sis VI: Growth and pubertal development in patients treated Myozyme in juvenile patients with Pompe disease. Mol Gen
with recombinant human N-acetylgalactosamine 4-sulfatase. Metab. 2007; 90: 227: 65.
J Pediatr Reha Med. 2010; 3: 89-100. 90. Wraith JE, Guffon N, Rohrbach M, Hwu WL, Korenke GC,
71. Giugliani R, Harmatz P, Wraith JE. Management guidelines for Bembi B. Natural history of Niemann-Pick disease type C in
mucopolysaccharidosis VI. Pediatrics. 2007; 120: 405-418. a multicentre observational retrospective cohort study. Mo-
72. Giugliani R, Harmatz P, Wraith J. Directrices para el trata- lecular Genetics and Metabolism. 2009; 98: 250-254.
miento de la Mucopolisacaridosis VI. Pediatrics. 2007; 120: 91. Pineda M, Perez-Poyato MS, OCallaghan M, Vilaseca MA,
1-14. Pocovi M, Domingo R. Clinical experience with miglustat
Rev Mex Pediatr 2014; 81(4); 143-153 152
Carbajal RL y cols. Enfoque simplificado para el diagnstico de enfermedades lisosomales en nios
therapy in pediatric patients with Niemann-Pick disease type disease type C: long-term data from a clinical trial. Mol Genet
C: a case series. Mol Genet Metabol. 2010; 99: 358-366. Metabol. 2010; 99: 351-357.
92. Pineda M, Wraith JE, Mengel E, Sedel F, Hwu L, Rohrbach
M. Miglustat in patients with Niemann-Pick disease Type C
(NPC): A multicenter observational retrospective cohort
study. Mol Genet Metabol. 2009; 98: 243-249. Correspondencia:
93. Patterson MC, Darleen V, Jacklin E, Larry A, Harbajan CB, Dr. Luis Carbajal Rodrguez
Luzy C. Long-Term miglustat therapy in children with Nie- Dpto. de Medicina Interna,
mann-Pick disease type C. J Child Neurol OnlineFirst. 2009; Instituto Nacional de Pediatra,
00: 1-6. Insurgentes Sur Nm. 3700-C,
94. Wraith JE, Darleen V, Jacklin E, Larry A, Harbajan CB, Luzy 04530, Mxico, D.F.
C. Miglustat in adult and juvenile patients with Niemann-Pick Tels: 1084-5518 y 1084-0900, ext. 1357
www.medigraphic.org.mx
153 Rev Mex Pediatr 2014; 81(4); 143-153
Anda mungkin juga menyukai
- Formativa 4 Bioquimica 2Dokumen28 halamanFormativa 4 Bioquimica 2Ever Lozano100% (1)
- Enfermedades Por Depósito LisosomalDokumen3 halamanEnfermedades Por Depósito LisosomalCamila MárquezBelum ada peringkat
- 10 1016@j Med 2016 09 020Dokumen10 halaman10 1016@j Med 2016 09 020Rafael Antonio Rodríguez DíazBelum ada peringkat
- Pedi Atria Yen FR Aras 3Dokumen129 halamanPedi Atria Yen FR Aras 3auroraBelum ada peringkat
- Enfermedades LisososmaDokumen13 halamanEnfermedades LisososmaHebert Jhimsy Pastor GarcíaBelum ada peringkat
- Lisosoma 1Dokumen4 halamanLisosoma 1José Gómez VicenteBelum ada peringkat
- Enfermedades LisosomalesDokumen6 halamanEnfermedades LisosomalesJuan carlos Bogallo CastroBelum ada peringkat
- Guia-Hunter MPS PDFDokumen30 halamanGuia-Hunter MPS PDFBoBelum ada peringkat
- Revista Médica Del Instituto Mexicano Del Seguro Social 0443-5117Dokumen17 halamanRevista Médica Del Instituto Mexicano Del Seguro Social 0443-5117MARÍA KAORY ROJAS LAGOSBelum ada peringkat
- Enfermedad de GaucherDokumen11 halamanEnfermedad de GaucherHeidy Bravo RamosBelum ada peringkat
- Breve Resumen Del Sindrome de MorquioDokumen8 halamanBreve Resumen Del Sindrome de Morquiojorge boquinBelum ada peringkat
- Enfermedades LisosomalesDokumen9 halamanEnfermedades LisosomalesCarlos Yhair GuerronBelum ada peringkat
- Síndrome Zellweger (Cerebro-Hepato-Renal) - Reporte de Un CasoDokumen6 halamanSíndrome Zellweger (Cerebro-Hepato-Renal) - Reporte de Un CasoNathaly LapoBelum ada peringkat
- Mucopolisacaridosis IVDokumen7 halamanMucopolisacaridosis IVMinitoBelum ada peringkat
- Enfermedad de GaucherDokumen7 halamanEnfermedad de GaucherLaura Nieto EstepaBelum ada peringkat
- Tratamiento Enzimatico Sustitutivo en Las Enfermedades LisosomalesDokumen8 halamanTratamiento Enzimatico Sustitutivo en Las Enfermedades Lisosomalesjhossgm8Belum ada peringkat
- Enfermedades Metabólicas Producidas Por Enzimas DefectuosasDokumen14 halamanEnfermedades Metabólicas Producidas Por Enzimas DefectuosasJuancarlos Cruz Muñoz0% (1)
- Galactosemia Revision de La LiteraturaDokumen18 halamanGalactosemia Revision de La LiteraturaJorge Luis Plasencia CubaBelum ada peringkat
- Ultima Entrega Revisiòn - Mucolipidosis Tipos II y IIIDokumen12 halamanUltima Entrega Revisiòn - Mucolipidosis Tipos II y IIIShanon DanielaBelum ada peringkat
- Enfermedades Lisosomales y Mitocondriales TerminadoDokumen28 halamanEnfermedades Lisosomales y Mitocondriales TerminadoMilton Cesar Rodrigo TantaleanBelum ada peringkat
- Sindrome de HurlerDokumen21 halamanSindrome de Hurlerfer.est.lop10Belum ada peringkat
- 81-Texto Del Artículo-485-1-10-20141009 PDFDokumen7 halaman81-Texto Del Artículo-485-1-10-20141009 PDFRolando TorresBelum ada peringkat
- Pdfspediatsp 2007sp071g PDFDokumen8 halamanPdfspediatsp 2007sp071g PDFHelen Jazmin Osorio RodriguezBelum ada peringkat
- ActPed2002 14Dokumen9 halamanActPed2002 14Lizeth PämelaBelum ada peringkat
- Errores Innatos Del MetabolismoDokumen21 halamanErrores Innatos Del MetabolismoDiego Castaño MejiaBelum ada peringkat
- Enfermedades LisosomalesDokumen4 halamanEnfermedades LisosomalesmarianaBelum ada peringkat
- Guía TP 02 - Patología Metabólica y NutricionalDokumen15 halamanGuía TP 02 - Patología Metabólica y NutricionalGiulia LeaoBelum ada peringkat
- Lectura 7Dokumen11 halamanLectura 7AndreBelum ada peringkat
- Guía TP Patología Metabólica y NutricionalDokumen15 halamanGuía TP Patología Metabólica y NutricionalCarolina Emilia PazBelum ada peringkat
- LibroDokumen5 halamanLibroArquimedes Sanchez BravoBelum ada peringkat
- Deficiencia de Glucosa-6-Fosfato Deshidrogenasa: de Lo Clínico A Lo BioquímicoDokumen13 halamanDeficiencia de Glucosa-6-Fosfato Deshidrogenasa: de Lo Clínico A Lo BioquímicoNicolle FelixBelum ada peringkat
- Enfermedad de Gaucher 2010 PDFDokumen7 halamanEnfermedad de Gaucher 2010 PDFMaria HernandezBelum ada peringkat
- HemtologíaDokumen27 halamanHemtologíaPaola Massai MontanerBelum ada peringkat
- TEMA 10: Enfermedades Neurológicas Pediátricas Hereditarias Genética, Diagnóstico Molecular y Medicina de PrecisiónDokumen16 halamanTEMA 10: Enfermedades Neurológicas Pediátricas Hereditarias Genética, Diagnóstico Molecular y Medicina de PrecisiónMark Sal DontoBelum ada peringkat
- Protocolo de Actuación en Las MucopolisacaridosisDokumen13 halamanProtocolo de Actuación en Las MucopolisacaridosisDr. Javier Antonio Toledo LimónBelum ada peringkat
- MucopolisacaridosisDokumen20 halamanMucopolisacaridosisPozole de LavaderoBelum ada peringkat
- Files Mucopolisacaridosis 1493216475Dokumen2 halamanFiles Mucopolisacaridosis 1493216475Alejandra RoaBelum ada peringkat
- Síndrome de Activación Macrofágica PDFDokumen9 halamanSíndrome de Activación Macrofágica PDFJOEGIORDABelum ada peringkat
- Glucogenosis, Lipoidosis y MucopolisacaridosisDokumen81 halamanGlucogenosis, Lipoidosis y Mucopolisacaridosisemely perezBelum ada peringkat
- ADL MetabolismoDokumen18 halamanADL MetabolismoDiana ZavalaBelum ada peringkat
- Portafolio Casoclinico VeraDokumen28 halamanPortafolio Casoclinico VeraJuan Jose VABelum ada peringkat
- MucopolisacaridosisDokumen97 halamanMucopolisacaridosisJessica Lissette CadilloBelum ada peringkat
- Guía de Morfología SanguíneaDokumen39 halamanGuía de Morfología Sanguíneaedson floresBelum ada peringkat
- Sind. Activacion MacrofagicaDokumen7 halamanSind. Activacion MacrofagicaMIGUEL ROJAS SIVILABelum ada peringkat
- 1968 7534 1 PBDokumen17 halaman1968 7534 1 PBJore-LeeBelum ada peringkat
- Enfermedad de WolmanDokumen12 halamanEnfermedad de WolmanDanilo Paul Villa PortillaBelum ada peringkat
- Monografia TAPBDokumen11 halamanMonografia TAPBNIURKA PAMELA AGUILAR CRUZBelum ada peringkat
- Sindrome Chediak-HigashiDokumen6 halamanSindrome Chediak-HigashiAngela RodriguezBelum ada peringkat
- Enfermedades Relacionadas Al Plegamienoto de ProteinasDokumen25 halamanEnfermedades Relacionadas Al Plegamienoto de ProteinasNessa Chacon JBelum ada peringkat
- Inhibidores Metabolicos de Los CarbohidratosDokumen5 halamanInhibidores Metabolicos de Los CarbohidratosMichelleBelum ada peringkat
- Enfermedades LisosomalesDokumen7 halamanEnfermedades Lisosomalesnelly carranza rodriguezBelum ada peringkat
- Diagnóstico Diferencial de Los Síndromes Del Sistema HemolinfopoyeticoDokumen110 halamanDiagnóstico Diferencial de Los Síndromes Del Sistema HemolinfopoyeticoCDBH100% (1)
- Patologia Neurologica en Metabolopatias Hereditarias y Adquiridas PDFDokumen9 halamanPatologia Neurologica en Metabolopatias Hereditarias y Adquiridas PDFAna Carmenza Romero PeñaBelum ada peringkat
- Resumen de Invetsigacion 5Dokumen4 halamanResumen de Invetsigacion 5Roxy L.GBelum ada peringkat
- Guia de Seminario de Enfermedades Por Deposito Lisosomal de LipidosDokumen18 halamanGuia de Seminario de Enfermedades Por Deposito Lisosomal de LipidosRuddy Humberto Sotomayor BrenesBelum ada peringkat
- Tema 35 + Tema 39Dokumen26 halamanTema 35 + Tema 39pablorafaeluBelum ada peringkat
- Alteraciones MetabolicasDokumen15 halamanAlteraciones MetabolicasSari Carhuamaca OréBelum ada peringkat
- Errores innatos en el metabolismo: Un abordaje integral del diagnóstico al tratamientoDari EverandErrores innatos en el metabolismo: Un abordaje integral del diagnóstico al tratamientoBelum ada peringkat
- Autocontrol de la Diabetes Mellitus Tipo 2. Un tratamiento psicológico cognitivo-conductualDari EverandAutocontrol de la Diabetes Mellitus Tipo 2. Un tratamiento psicológico cognitivo-conductualPenilaian: 2 dari 5 bintang2/5 (1)
- Onicomicosis - Diagnóstico y TratamientoDokumen10 halamanOnicomicosis - Diagnóstico y TratamientoOmar SotoBelum ada peringkat
- Déficit de Pantotenato Quinasa PDFDokumen7 halamanDéficit de Pantotenato Quinasa PDFOsiithaa CañaszBelum ada peringkat
- OnicomicosisDokumen14 halamanOnicomicosisthevintage100% (1)
- Glucogenosis PDFDokumen6 halamanGlucogenosis PDFOsiithaa CañaszBelum ada peringkat
- EnzimopatíasDokumen11 halamanEnzimopatíasOsiithaa CañaszBelum ada peringkat
- Cardio Congenitas GPCDokumen7 halamanCardio Congenitas GPCOsiithaa CañaszBelum ada peringkat
- Alteraciones de Las UñasDokumen7 halamanAlteraciones de Las UñasErico Passos De LimaBelum ada peringkat
- Nac AepedDokumen8 halamanNac AepedjoscleonBelum ada peringkat
- 18 EspinaDokumen6 halaman18 EspinafhernandagBelum ada peringkat
- Uña 1Dokumen8 halamanUña 1Osiithaa CañaszBelum ada peringkat
- Tumor de Willms PDFDokumen3 halamanTumor de Willms PDFOsiithaa CañaszBelum ada peringkat
- SalamDokumen6 halamanSalamOmr RrzBelum ada peringkat
- Enfermedad de Von Gierke PDFDokumen5 halamanEnfermedad de Von Gierke PDFOsiithaa CañaszBelum ada peringkat
- PTIDokumen10 halamanPTIOsiithaa CañaszBelum ada peringkat
- Micosis Profundas PDFDokumen14 halamanMicosis Profundas PDFRogerio GobbiBelum ada peringkat
- LesDokumen13 halamanLesFanni GDBelum ada peringkat
- Clasificacion de La HTPDokumen9 halamanClasificacion de La HTPmoshoolindoBelum ada peringkat
- Choque AnafilácticoDokumen6 halamanChoque AnafilácticoOsiithaa CañaszBelum ada peringkat
- HIPOPOTASEMIADokumen5 halamanHIPOPOTASEMIARoy Acosta SobradoBelum ada peringkat
- Obstruccion IntestinalDokumen4 halamanObstruccion Intestinaldeiner83Belum ada peringkat
- Obstruccion IntestinalDokumen4 halamanObstruccion Intestinaldeiner83Belum ada peringkat
- Metabolismo Del Agua y Los ElectrolitosDokumen16 halamanMetabolismo Del Agua y Los Electrolitosbiofisica2011100% (1)
- Funcion EndocrinaDokumen8 halamanFuncion EndocrinaOsiithaa CañaszBelum ada peringkat
- V33n4a06 PDFDokumen9 halamanV33n4a06 PDFOsiithaa CañaszBelum ada peringkat
- Defectos Del Tubo NeuralDokumen7 halamanDefectos Del Tubo NeuralOsiithaa CañaszBelum ada peringkat
- 18 EspinaDokumen6 halaman18 EspinafhernandagBelum ada peringkat
- Metabolismo Del Agua y Los ElectrolitosDokumen16 halamanMetabolismo Del Agua y Los Electrolitosbiofisica2011100% (1)
- Manual de Procedimiento de Pediatria 2010Dokumen60 halamanManual de Procedimiento de Pediatria 2010María Yakeline Rodríguez RodríguezBelum ada peringkat
- Otitis Media Aguda-Guía de Práctica ClínicaDokumen23 halamanOtitis Media Aguda-Guía de Práctica ClínicaEdwin VargasBelum ada peringkat
- Ficha de Inscripción InicialDokumen2 halamanFicha de Inscripción Inicialcoframatachira50% (2)
- La Estimulación Vestibular en El Desarrollo PsicomotrizDokumen180 halamanLa Estimulación Vestibular en El Desarrollo PsicomotrizMaría Constanza Marín JaramilloBelum ada peringkat
- Programa Manejo y Rehabilitacion de Pacientes Traqueostomizados PDF 439 KBDokumen25 halamanPrograma Manejo y Rehabilitacion de Pacientes Traqueostomizados PDF 439 KBVanessa Saavedra VerdejoBelum ada peringkat
- Historia Del Cuidado en SaludDokumen10 halamanHistoria Del Cuidado en SaludHernandez BeniBelum ada peringkat
- Cuadro Medico AsdeslasDokumen150 halamanCuadro Medico AsdeslasAlvaro SuarezBelum ada peringkat
- Formulario SAN 2017Dokumen3 halamanFormulario SAN 2017Fredy TzubBelum ada peringkat
- Noche de Alice MunroDokumen5 halamanNoche de Alice MunroMaria Jose Viera-GalloBelum ada peringkat
- Anexos 1 Sin Vencimientos Documentos Lista de ChequeoDokumen2 halamanAnexos 1 Sin Vencimientos Documentos Lista de ChequeoCarol ForeroBelum ada peringkat
- Estapedectomía Resumen ExposoiconDokumen12 halamanEstapedectomía Resumen Exposoicongama100% (1)
- Protocolo Aplicacion Procedimiento Eutanasia ColombiaDokumen120 halamanProtocolo Aplicacion Procedimiento Eutanasia ColombiaEsteban CausadoBelum ada peringkat
- Educación PrenatalDokumen12 halamanEducación PrenatalPauosi Herrera100% (3)
- 00 Pauta de Cotejo Sobre Sir Drogas ComplejasDokumen1 halaman00 Pauta de Cotejo Sobre Sir Drogas ComplejasConstanza Hernández GrandónBelum ada peringkat
- Bibliografía Elsa MejiaDokumen2 halamanBibliografía Elsa MejiarchisculBelum ada peringkat
- Instr - Categ. I-3Dokumen37 halamanInstr - Categ. I-3Alex Corzo MarticorenaBelum ada peringkat
- Tema 1. La Evaluación y La ValoraciónDokumen11 halamanTema 1. La Evaluación y La ValoraciónZaira Sánchez GonzaloBelum ada peringkat
- Evaluación de Los Pares CranealesDokumen2 halamanEvaluación de Los Pares CranealesLuis Alvarez33% (3)
- ENFERMEDADES PROFEsionales NeumoconiosisDokumen14 halamanENFERMEDADES PROFEsionales NeumoconiosisCarlos GonzalesBelum ada peringkat
- Universidad de Los Andes - Medicina PDFDokumen5 halamanUniversidad de Los Andes - Medicina PDFkarent lombanaBelum ada peringkat
- Historia de La Oncologia PDFDokumen101 halamanHistoria de La Oncologia PDFmarcelo ferrariBelum ada peringkat
- Triple Rule OutDokumen33 halamanTriple Rule OutFigueroa Cristian100% (2)
- Informe Tecnico Hospital General 1a RevisiónDokumen113 halamanInforme Tecnico Hospital General 1a RevisiónRocio Ucan100% (1)
- Situacion de Micronutrientes en GuatemalaDokumen18 halamanSituacion de Micronutrientes en GuatemalaPao MendozaBelum ada peringkat
- 13 15Dokumen3 halaman13 15ABBelum ada peringkat
- La Funcion Reguladora Del Lenguaje en El AddhDokumen31 halamanLa Funcion Reguladora Del Lenguaje en El AddhJonathan Carlos Aguilar BuenoBelum ada peringkat
- Historia de La EpidemiologíaDokumen4 halamanHistoria de La Epidemiología作之助Belum ada peringkat
- Osteocondrosis en Animales MenoresDokumen22 halamanOsteocondrosis en Animales MenoresMaria Gabriela Ramirez CoronelBelum ada peringkat
- SjdeluriganchoDokumen78 halamanSjdeluriganchoDaniel CHBelum ada peringkat
- Nuevos Procedimientos en Anestesia Loca en Odontología: El Sistema Injex ®Dokumen8 halamanNuevos Procedimientos en Anestesia Loca en Odontología: El Sistema Injex ®Virtualdental MexicoBelum ada peringkat