History and Physical Examination: Evaluation
Diunggah oleh
Dore Nan0 penilaian0% menganggap dokumen ini bermanfaat (0 suara)
9 tayangan3 halamanEdema agudo pulmonar
Judul Asli
Edema
Hak Cipta
© © All Rights Reserved
Format Tersedia
DOCX, PDF, TXT atau baca online dari Scribd
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniEdema agudo pulmonar
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai DOCX, PDF, TXT atau baca online dari Scribd
0 penilaian0% menganggap dokumen ini bermanfaat (0 suara)
9 tayangan3 halamanHistory and Physical Examination: Evaluation
Diunggah oleh
Dore NanEdema agudo pulmonar
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai DOCX, PDF, TXT atau baca online dari Scribd
Anda di halaman 1dari 3
The following two fundamentally different types of pulmonary edema occur in
humans: cardiogenic pulmonary edema (also termed hydrostatic or hemodynamic
edema) and noncardiogenic pulmonary edema (also known as increased-permeability
pulmonary edema, acute lung injury, or acute respiratory distress syndrome).
Although they have distinct causes, cardiogenic and noncardiogenic pulmonary edema
may be difficult to distinguish because of their similar clinical manifestations.
Knowledge of the cause of acute pulmonary edema has important implications
for treatment. Patients with cardiogenic pulmonary edema typically are treated with
diuretics and afterload reduction, although the underlying cause may require other
treatment, including coronary revascularization. 1 Patients with noncardiogenic pulmonary
edema who require mechanical ventilation should be ventilated with a low
tidal volume (6 ml per kilogram of predicted body weight) and a plateau airway
pressure less than 30 cm of water. This lung-protective strategy of ventilation reduces
mortality in patients with acute lung injury.2,3 In addition, for patients with
severe sepsis, recombinant activated protein C4 and low-dose hydrocortisone5 should
be considered. Prompt diagnosis of the cause of acute pulmonary edema with the use
of noninvasive methods, supplemented by catheterization of the pulmonary artery
when there is diagnostic uncertainty, facilitates timely and appropriate treatment.
Accurate diagnosis of acute pulmonary edema requires an understanding of microvascular
fluid exchange in the lung (Fig. 1). In the normal lung (Fig. 1A), fluid
and protein leakage is thought to occur primarily through small gaps between capillary
endothelial cells. Fluid and solutes that are filtered from the circulation into
the alveolar interstitial space normally do not enter the alveoli because the alveolar
epithelium is composed of very tight junctions. Rather, once the filtered fluid enters
the alveolar interstitial space, it moves proximally into the peribronchovascular
space. Under normal conditions the lymphatics remove most of this filtered fluid
from the interstitium and return it to the systemic circulation. Movement of larger
plasma proteins is restricted. The hydrostatic force for fluid filtration across the lung
microcirculation is approximately equal to the
hydrostatic pressure in the pulmonary capillaries
(Fig. 1A), which is partially offset by a protein osmotic
pressure gradient.
A rapid increase in hydrostatic pressure in the
pulmonary capillaries leading to increased transvascular
fluid filtration is the hallmark of acute
cardiogenic or volume-overload edema (Fig. 1B).
Increased hydrostatic pressure in the pulmonary
capillaries is usually due to elevated pulmonary
venous pressure from increased left ventricular
end-diastolic pressure and left atrial pressure. Mild
elevations of left atrial pressure (18 to 25 mm Hg)
cause edema in the perimicrovascular and peribronchovascular
interstitial spaces.7 As left atrial
pressure rises further (>25 mm Hg), edema fluid
breaks through the lung epithelium, flooding the
alveoli with protein-poor fluid7 (Fig. 1B).
By contrast, noncardiogenic pulmonary edema
is caused by an increase in the vascular permeability
of the lung, resulting in an increased flux of
fluid and protein into the lung interstitium and
air spaces (Fig. 1C). Noncardiogenic pulmonary
edema has a high protein content because the vascular
membrane is more permeable to the outward
movement of plasma proteins. The net quantity of
accumulated pulmonary edema is determined by
the balance between the rate at which fluid is
filtered into the lung7 and the rate at which fluid
is removed from the air spaces and lung interstitium.
evaluation
History and Physical Examination
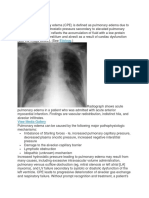
The presenting features of acute cardiogenic and
noncardiogenic pulmonary edema are similar. Interstitial
edema causes dyspnea and tachypnea. Alveolar
flooding leads to arterial hypoxemia and
may be associated with cough and expectoration
of frothy edema fluid. The history should focus
on determining the underlying clinical disorder
that has led to pulmonary edema.8 Common
causes of cardiogenic pulmonary edema include
ischemia with or without myocardial infarction,
exacerbation of chronic systolic or diastolic heart
failure, and dysfunction of the mitral or aortic valve.
Volume overload should also be considered. A typical
history of paroxysmal nocturnal dyspnea or
orthopnea suggests cardiogenic pulmonary edema.
However, a silent myocardial infarction or occult diastolic dysfunction may also manifest as
acute
pulmonary edema, with few clues provided by the
history.9
In contrast, noncardiogenic pulmonary edema
is associated primarily with other clinical disorders,
including pneumonia, sepsis, aspiration of
gastric contents, and major trauma associated with
the administration of multiple blood-product transfusions.
3 The history should focus on signs and
symptoms of infection, a decrease in the level of
consciousness associated with vomiting, trauma,
and details of medications and ingestions. Unfortunately,
the history is not always reliable in distinguishing
cardiogenic from noncardiogenic pulmonary
edema. For example, an acute myocardial
infarction (suggesting cardiogenic edema) may be
complicated by syncope or cardiac arrest with aspiration
of gastric contents and noncardiogenic
edema. Conversely, in patients with severe trauma
or infection (suggesting noncardiogenic edema),
fluid resuscitation may lead to volume overload
and pulmonary edema from an increase in lung
vascular hydrostatic pressure.
Patients with cardiogenic pulmonary edema
often have an abnormal cardiac examination. Auscultation
of an S3 gallop is relatively specific for
elevated left ventricular end-diastolic pressure and
left ventricular dysfunction10-13 and suggests cardiogenic
pulmonary edema.10-12 The specificity of
this finding is high (90 to 97 percent), but its sensitivity
is low (9 to 51 percent).10-13 The wide range
of sensitivity probably reflects the difficulty in
clearly identifying an S3 gallop on physical examination,
a particular challenge in a critically ill
patient in whom intrathoracic sounds created by
mechanical ventilation interfere with auscultation.
Data are lacking on the sensitivity and specificity
of other findings on examination for cardiogenic
edema. A murmur consistent with valvular
stenosis or regurgitation should raise suspicion
for the diagnosis of cardiogenic edema. Elevated
neck veins, an enlarged and tender liver, and peripheral
edema suggest elevated central venous
pressure. However, assessment of central venous
pressure by physical examination in a critically ill
patient can be difficult. Also, peripheral edema is
not specific for left heart failure and may be associated
with hepatic or renal insufficiency, right
heart failure, or systemic infection. The lung examination
is not helpful, since alveolar flooding
from any cause will manifest as inspiratory crackles
and often rhonchi. The abdominal, pelvic, and
rectal examinations are important. An intraabdominal
crisis such as perforation of a viscus
can cause acute lung injury with noncardiogenic
edema, and patients who are mechanically ventilated
may be unable to provide a history of abdominal
symptoms. Patients with noncardiogenic
edema often have warm extremities, even in the
absence of sepsis, whereas patients with cardiogenic
edema and poor cardiac output usually have
cool extremities.
Laboratory Testing
Electrocardiographic findings may suggest myocardial
ischemia or infarction. Elevated troponin
levels may indicate damage to myocytes. However,
elevated troponin levels can occur in patients with
severe sepsis in the absence of evidence for an
acute coronary syndrome.14-17 In a patient who is
obtunded and has pulmonary edema of an unknown
cause, measurement of electrolytes, the serum
osmolarity, and a toxicology screen may lead
to the diagnosis of an unsuspected ingestion. Elevated
levels of serum amylase and lipase suggest
acute pancreatitis.
Plasma levels of brain natriuretic peptide (BNP)
are often used in the evaluation of pulmonary
edema. BNP is secreted predominantly by the cardiac
ventricles in response to wall stretch or increased
intracardiac pressures. In patients with
congestive heart failure, plasma BNP levels correlate
with left ventricular end-diastolic pressure18-21
and pulmonary-artery occlusion pressure.22 According
to a consensus panel, a BNP level below
100 pg per milliliter indicates that heart failure
is unlikely (negative predictive value, >90 percent),
whereas a BNP level greater than 500 pg per milliliter
indicates that heart failure is likely (positive
predictive value, >90 percent).23 However, BNP
levels between 100 and 500 pg per milliliter provide
inadequate diagnostic discrimination.
BNP levels must be interpreted with caution in
critically ill patients, since the predictive value of
BNP levels is uncertain in this group. Some reports
indicate that BNP levels can be elevated in critically
ill patients even in the absence of heart failure.
24,25 Levels between 100 and 500 pg per milliliter
are common in these patients.26 In one report,
all eight patients with sepsis with normal left
ventricular function had BNP levels above 500 pg
per milliliter.27 Thus, measuring BNP is most useful
in critically ill patients if the level is below
100 pg per milliliter. BNP levels are also higher in
Anda mungkin juga menyukai
- Medicine in Brief: Name the Disease in Haiku, Tanka and ArtDari EverandMedicine in Brief: Name the Disease in Haiku, Tanka and ArtPenilaian: 5 dari 5 bintang5/5 (1)
- Acute Pulmonary Edema - NEJMDokumen4 halamanAcute Pulmonary Edema - NEJMSuzika Dewi0% (1)
- Pathophysiology of Cardiogenic Pulmonary Edema - UpToDateDokumen14 halamanPathophysiology of Cardiogenic Pulmonary Edema - UpToDateStefani AtlleBelum ada peringkat
- Pathophysiology of Cardiogenic Pulmonary Edema - UpToDateDokumen9 halamanPathophysiology of Cardiogenic Pulmonary Edema - UpToDateRoberto López MataBelum ada peringkat
- Pathophysiology of Cardiogenic Pulmonary EdemaDokumen13 halamanPathophysiology of Cardiogenic Pulmonary EdemaIrina DuceacBelum ada peringkat
- Definition of Noncardiogenic Pulmonary Edemanoncardiogenic Pulmonary Edema Is IdentifiedDokumen35 halamanDefinition of Noncardiogenic Pulmonary Edemanoncardiogenic Pulmonary Edema Is IdentifiedAya EyadBelum ada peringkat
- Noncardiogenic Pulmonary EdemaDokumen19 halamanNoncardiogenic Pulmonary EdemaCláudio CalixtoBelum ada peringkat
- Pulmonary Edema - StatPearls - NCBI BookshelfDokumen7 halamanPulmonary Edema - StatPearls - NCBI BookshelfriyanasirBelum ada peringkat
- Cor PulmonaleDokumen15 halamanCor PulmonaleRizky Zulfa Afrida100% (1)
- 3487 VJXHXVDokumen13 halaman3487 VJXHXVمحمد عقيليBelum ada peringkat
- Cor PulmonaleDokumen62 halamanCor PulmonaleAlbert NixonBelum ada peringkat
- Sclerodactyly, and Telangiectasis) Syndrome Accompany-: Cor PulmonaleDokumen2 halamanSclerodactyly, and Telangiectasis) Syndrome Accompany-: Cor PulmonaledivinaBelum ada peringkat
- CorpulmonalefinalDokumen37 halamanCorpulmonalefinalRay ReyesBelum ada peringkat
- Cor Pulmonale by Prof. Dr. Rasheed Abd El Khalek M. DDokumen45 halamanCor Pulmonale by Prof. Dr. Rasheed Abd El Khalek M. DvaishnaviBelum ada peringkat
- Noncardiogenic Pulmonary EdemaDokumen12 halamanNoncardiogenic Pulmonary EdemaIrina DuceacBelum ada peringkat
- Cor Pulmonale EmedicineDokumen16 halamanCor Pulmonale EmedicineHengki Permana PutraBelum ada peringkat
- SDL 19 Cor Pulmonale - BMS16091064Dokumen6 halamanSDL 19 Cor Pulmonale - BMS16091064Jonathan YeohBelum ada peringkat
- Pathophysiology of Cardiogenic Pulmonary EdemaDokumen8 halamanPathophysiology of Cardiogenic Pulmonary EdemaLili Fiorela CRBelum ada peringkat
- Adult Respiratory Distress SyndromeDokumen5 halamanAdult Respiratory Distress SyndromeMeh AdroidBelum ada peringkat
- Acute Pulmonary Edema: NEJM December 2005 Presentation: R2 黃志宇Dokumen25 halamanAcute Pulmonary Edema: NEJM December 2005 Presentation: R2 黃志宇Rinda Putri AnggrainiBelum ada peringkat
- Non-Cardiogenic Pulmonary EdemaDokumen7 halamanNon-Cardiogenic Pulmonary EdemadarmariantoBelum ada peringkat
- Cor PulmonaleDokumen14 halamanCor PulmonaleEvangelin MelvinBelum ada peringkat
- Pulmonary HypertensionDokumen15 halamanPulmonary HypertensionChinju CyrilBelum ada peringkat
- Copd and Cor PulmonalDokumen14 halamanCopd and Cor PulmonalAldi RafaelBelum ada peringkat
- Pulmonary ThromboembolismDokumen4 halamanPulmonary ThromboembolismValerrie NgenoBelum ada peringkat
- Acute Pumonary EdemaDokumen7 halamanAcute Pumonary EdemaLucas AresBelum ada peringkat
- Uptodate Cor Pulmonale PDFDokumen13 halamanUptodate Cor Pulmonale PDFcristianamihailaBelum ada peringkat
- Cor PulmonaleDokumen27 halamanCor PulmonaleumapathisivanBelum ada peringkat
- Management of Pulmonary EdemaDokumen42 halamanManagement of Pulmonary Edemaademato4real576Belum ada peringkat
- Pathogenesis and PathophysiologyDokumen3 halamanPathogenesis and PathophysiologyEduardoFerreiraBelum ada peringkat
- Cor Pulmonale PresentationDokumen17 halamanCor Pulmonale Presentationandi reskifaisBelum ada peringkat
- Pulmonary HemorrhageDokumen6 halamanPulmonary HemorrhageSubas SharmaBelum ada peringkat
- Ali A Sovari, MD: Author:, Staff Physician, Department of Internal MedicineDokumen9 halamanAli A Sovari, MD: Author:, Staff Physician, Department of Internal MedicineMaríaDelPilarAguilarBelum ada peringkat
- Acute Pulmonary OdemaDokumen9 halamanAcute Pulmonary OdemaAnonymous ysrxggk21cBelum ada peringkat
- 1 FullDokumen4 halaman1 FullMuli YamanBelum ada peringkat
- Objectives:: Access Free Multiple Choice Questions On This Topic. Go ToDokumen6 halamanObjectives:: Access Free Multiple Choice Questions On This Topic. Go ToClarisse Cloie LamberteBelum ada peringkat
- Pulmonary Edema Facts What Causes Pulmonary Edema?Dokumen23 halamanPulmonary Edema Facts What Causes Pulmonary Edema?Louella RamosBelum ada peringkat
- Portopulmonary Hypertension and Hepatopulmonary Syndrome: ReviewDokumen8 halamanPortopulmonary Hypertension and Hepatopulmonary Syndrome: ReviewAdel HamadaBelum ada peringkat
- Kuliah 4 Oedem Paru PulmonaryedemaDokumen43 halamanKuliah 4 Oedem Paru PulmonaryedematrianaamaliaBelum ada peringkat
- Cor Pulmonale - StatPearls - NCBI BookshelfDokumen4 halamanCor Pulmonale - StatPearls - NCBI BookshelfAldi RafaelBelum ada peringkat
- Cardiac TamponadeDokumen6 halamanCardiac TamponadeVicky XieBelum ada peringkat
- Eisenmenger SyndromeDokumen9 halamanEisenmenger SyndromesyifandikaBelum ada peringkat
- Fluid Management in Pre-EclampsiaDokumen13 halamanFluid Management in Pre-Eclampsiakiki pransiskaBelum ada peringkat
- Respiratory Distress - Tintanalli's Emergency MedicineDokumen31 halamanRespiratory Distress - Tintanalli's Emergency MedicineRon KBelum ada peringkat
- Anglisht 2Dokumen8 halamanAnglisht 2Ejona SuloBelum ada peringkat
- Pulmonary Edema: René P. Michel, Peter GoldbergDokumen34 halamanPulmonary Edema: René P. Michel, Peter GoldbergNadya NovianiBelum ada peringkat
- Background: EtiologyDokumen5 halamanBackground: Etiologylia lykimBelum ada peringkat
- Pulmonary Embolism (PE) - Pulmonary Disorders - MSD Manual Professional EditionDokumen25 halamanPulmonary Embolism (PE) - Pulmonary Disorders - MSD Manual Professional Editionpeterpavel112Belum ada peringkat
- Edema ParuDokumen4 halamanEdema ParuSri BayaniBelum ada peringkat
- Cor Pulmonale ReDokumen35 halamanCor Pulmonale ReFabb NelsonBelum ada peringkat
- 3.3 Pathophysiology of Plasma LeakDokumen1 halaman3.3 Pathophysiology of Plasma LeakM Adil AliBelum ada peringkat
- Pulmonary Embolism Is A Common and Potentially Lethal ConditionDokumen13 halamanPulmonary Embolism Is A Common and Potentially Lethal ConditionJasleen KaurBelum ada peringkat
- Cardiac AsthmaDokumen12 halamanCardiac AsthmaNeupane KsabBelum ada peringkat
- Acute Respiratory Distress SyndromeDokumen36 halamanAcute Respiratory Distress Syndromedr9348345000Belum ada peringkat
- Congestive Heart FailureDokumen19 halamanCongestive Heart FailureIlavenil PanduranganBelum ada peringkat
- Bookshelf NBK360Dokumen3 halamanBookshelf NBK360Atikah PurnamasariBelum ada peringkat
- KP 2.5.5.3 Cor PulmonaleDokumen17 halamanKP 2.5.5.3 Cor Pulmonalenurul ramadhiniBelum ada peringkat
- Cardiogenic Shock: Physical FindingsDokumen9 halamanCardiogenic Shock: Physical FindingsEduardo Perez GonzalezBelum ada peringkat
- Pulmonary EdemaDokumen6 halamanPulmonary EdemaJelly BeanBelum ada peringkat
- Abordaje Nodulo PulmonarDokumen11 halamanAbordaje Nodulo PulmonarDore NanBelum ada peringkat
- SATI Ventilacion MecanicaDokumen389 halamanSATI Ventilacion MecanicaDore NanBelum ada peringkat
- 2021 - TorresDokumen28 halaman2021 - TorresCarlosBelum ada peringkat
- BTS Guidelines For The Investigation and Management of Pulmonary NodulesDokumen64 halamanBTS Guidelines For The Investigation and Management of Pulmonary NodulesHARIHARAN NARENDRANBelum ada peringkat
- DysrhythmiaDokumen30 halamanDysrhythmiaJJ JirapathBelum ada peringkat
- 9 Heart MuscleDokumen31 halaman9 Heart Muscleshoeb4uBelum ada peringkat
- 07 Blood Pressure Abnormality 1-8-2021 配布用Dokumen22 halaman07 Blood Pressure Abnormality 1-8-2021 配布用Lan NguyenBelum ada peringkat
- LWW BATES 10 Cardiovascular System Transcript FINALDokumen11 halamanLWW BATES 10 Cardiovascular System Transcript FINALShivamBelum ada peringkat
- Orbital AVMDokumen7 halamanOrbital AVMDedeh KurniasihBelum ada peringkat
- Nadi Pareeksha (Pulse Diagnosis) - An Authentic Scientific ApproachDokumen44 halamanNadi Pareeksha (Pulse Diagnosis) - An Authentic Scientific ApproachNaveen Kumar100% (1)
- Heart Failure Pelatihan Dokter KeluargaDokumen55 halamanHeart Failure Pelatihan Dokter KeluargaDelia NingrumBelum ada peringkat
- Central Venous PressureDokumen10 halamanCentral Venous PressureDenise EspinosaBelum ada peringkat
- Electrical System of The HeartDokumen4 halamanElectrical System of The HeartEldhaBelum ada peringkat
- Acute Coronary Syndrome in Clinical Practice: Firman B. LeksmonoDokumen48 halamanAcute Coronary Syndrome in Clinical Practice: Firman B. Leksmonojames klemens phieter phieBelum ada peringkat
- MidcabDokumen7 halamanMidcabNs PadiludinBelum ada peringkat
- K - 7 Atrial Flutter (IKA)Dokumen8 halamanK - 7 Atrial Flutter (IKA)thomasfelixBelum ada peringkat
- Circle of VillisDokumen17 halamanCircle of VillisAradhana SamuelBelum ada peringkat
- Jurnal JantungDokumen5 halamanJurnal JantungFileo AdisonBelum ada peringkat
- Slide Materi Dr. Kevin Triangto, SPKFR - Heart Failure - PMR4GP 2022Dokumen27 halamanSlide Materi Dr. Kevin Triangto, SPKFR - Heart Failure - PMR4GP 2022Maulia Wisda Era ChresiaBelum ada peringkat
- Sudden Death SyndromeDokumen72 halamanSudden Death SyndromeStella CooKeyBelum ada peringkat
- Transportation in Animals and PlantsDokumen2 halamanTransportation in Animals and PlantsHimanshi JainBelum ada peringkat
- Informe DEMO Del Holter de Arritmia Contec TLC9803Dokumen14 halamanInforme DEMO Del Holter de Arritmia Contec TLC9803Edward MoralesBelum ada peringkat
- Blood and Circulation PDFDokumen47 halamanBlood and Circulation PDFeric sivaneshBelum ada peringkat
- Neuro-Ultrasonography 2020Dokumen15 halamanNeuro-Ultrasonography 2020Santiago PovedaBelum ada peringkat
- Introduction To The CvsDokumen43 halamanIntroduction To The CvsParmesh PandeyBelum ada peringkat
- Circle of WillisDokumen3 halamanCircle of WillisRisah Mae SarinoBelum ada peringkat
- Thrombosis and EmbolismDokumen39 halamanThrombosis and Embolismilva100% (1)
- Ginekologija Sa AkusertsvomDokumen2 halamanGinekologija Sa AkusertsvomZaklina RistovicBelum ada peringkat
- Angiography and ArteriographyDokumen64 halamanAngiography and ArteriographyRyBone95100% (1)
- Naskah Publikasi Agiliaaa-1Dokumen5 halamanNaskah Publikasi Agiliaaa-1Indra AlazharBelum ada peringkat
- The Heart: Test I: Read and Understand The Question Circle The Best AnswerDokumen2 halamanThe Heart: Test I: Read and Understand The Question Circle The Best AnswerHolly May MontejoBelum ada peringkat
- Medical Form Vehicle Licensing CharnwoodDokumen12 halamanMedical Form Vehicle Licensing CharnwoodKalkin43Belum ada peringkat
- Cardiovascular Agents PDFDokumen118 halamanCardiovascular Agents PDFgherlethrBelum ada peringkat
- Approach To Diagnosis of Congenital Heart DiseasesDokumen85 halamanApproach To Diagnosis of Congenital Heart DiseasesNirav CHOVATIYABelum ada peringkat