Ivabradine PDF
Diunggah oleh
lilisJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Ivabradine PDF
Diunggah oleh
lilisHak Cipta:
Format Tersedia
Anticancer Section / Short Communication
Chemotherapy 2018;63:315–320 Received: August 24, 2018
Accepted: November 19, 2018
DOI: 10.1159/000495576 Published online: March 6, 2019
Ivabradine in Cancer Treatment-Related
Left Ventricular Dysfunction
Matteo Sarocchi a, b Eleonora Arboscello c Giorgio Ghigliotti a, b
Roberto Murialdo d Claudia Bighin e Francesca Gualandi f Vera Sicbaldi d
Manrico Balbi a, b Claudio Brunelli a, b Paolo Spallarossa a
a Cardiovascular Disease Unit, San Martino Policlinic Hospital, Genoa, Italy; b Department of Internal Medicine,
University of Genoa, Genoa, Italy; c Emergency Medicine Unit, San Martino Policlinic Hospital, Genoa, Italy; d Internal
Medicine Unit, San Martino Policlinic Hospital, Genoa, Italy; e Medical Oncology Unit, San Martino Policlinic Hospital,
Genoa, Italy; f Haematology Unit, San Martino Policlinic Hospital, Genoa, Italy
Keywords carefully titrated. Hypotension (70%) and fatigue (77%)
Ivabradine · Cardiotoxicity · Cardio-oncology · Heart failure · were the main causes limiting the treatment with ACEi/ARB
Cancer · Adverse drug reaction · Hypotension and BB. After a mean follow-up of 6.5 months, LVEF in-
creased from 45.1% (SD = 6.4) to 53.2% (SD = 3.9; p < 0.001).
When patients were analyzed according to the type of can-
Abstract cer therapy, no difference in LVEF changes across the
Background: Patients developing cancer treatment-relat- groups was found. NYHA class ameliorated in 11 patients,
ed left ventricular dysfunction (CTrLVD) require a prompt while fatigue improved in 8 patients. No serious cardiovas-
therapy. Hypotension, dizziness, and fatigue often limit the cular side effects were reported. Conclusions: The ability to
use of angiotensin-converting enzyme inhibitors (ACEi), an- improve symptoms and LVEF in unfit cancer patients makes
giotensin receptor blockers (ARB), and β-blockers (BB) in ivabradine a reasonable pharmacological tool for treating
cancer patients who may already be afflicted by these CTrLVD. © 2019 S. Karger AG, Basel
symptoms. Ivabradine is a heart rate-lowering drug that
does not cause hypotension and may be used in heart fail-
ure with reduced left ventricular ejection fraction (LVEF).
Objective: The aim of this paper was to investigate the role Introduction
of ivabradine to treat CTrLVD. Methods: A retrospective
analysis in a cohort of 30 patients with CTrLVD (LVEF <50%) Left-ventricular dysfunction (LVD) related to can-
receiving ivabradine on top of the maximal tolerated dose cer treatment (CTrLVD) is a threatening cardiovascu-
of ACEi/ARB and BB was performed. We evaluated cardio- lar complication of many chemotherapeutic agents
vascular treatment, oncologic treatment, LVEF, functional and molecular-targeted therapies [1, 2] that should be
class (New York Heart Association [NYHA]), and fatigue dur- treated as soon as possible [3]. Patients who develop
ing the study period. Results: Ivabradine was initially start- this complication are likely to benefit from angioten-
ed at the dose of 2.5 mg/b.i.d. in most patients and then sin-converting enzyme inhibitors (ACEi), angiotensin
East Carolina University - Laupus Library
150.216.68.200 - 3/7/2019 11:51:44 AM
© 2019 S. Karger AG, Basel Paolo Spallarossa, MD
Cardiovascular Disease Unit, Policlinic Hospital San Martino
Largo Rosanna Benzi 10
E-Mail karger@karger.com
IT–16132 Genoa (Italy)
www.karger.com/che
Downloaded by:
E-Mail paolo.spallarossa @ unige.it
Functional class HR SBP
20
140 180
**
15 p < 0.01 120 160
Patients, n
100 140
10
mm Hg
bpm
80 120
5
60 100
0
40 80
NYHA 1 NYHA 2 NYHA 3
LVEF
60 ** ** ** * ** ** ** **
40
LVEF, %
20
0
Continued Discontinued ANT TRZ HSCT TnI− TnI+
Overall Cancer treatment Cancer treatment Troponin
discontinuation type alteration
■ Before ivabradine ■ After ivabradine
Fig. 1. New York Heart Association (NYHA) class, heart rate (HR), systolic blood pressure (SBP), and left ven-
tricular ejection fraction (LVEF) before and after ivabradine. Patients were grouped according to the type of an-
ticancer treatment (ANT, anthracyclines; TRZ, trastuzumab; HSCT, allogeneic stem cell transplantation), main-
tenance or interruption of treatment, and occurrence or absence of troponin abnormalities (TnI– or TnI+).
** p < 0.001, * p < 0.01.
receptor blockers (ARB), and β-blocker (BB) treatment influence patients’ compliance and might drive the
to a similar extent as in the general population [1]. oncologist to discontinue the best anticancer therapy
There are, however, conflicting reports in this regard [2, 7].
[4], as well as concerns about the side effects of these Ivabradine is a heart rate-lowering medication that in-
drugs in patients undergoing active cancer treatments hibits the If channel in the sinoatrial node without effects
[5, 6]. Cancer and cancer treatment may induce varia- on inotropy, systemic blood pressure, and vascular resis-
tions in volume load, heart rate, and blood pressure. tance [8, 9]. In patients with heart failure and reduced left
Accordingly, it is not surprising that the use of vasoac- ventricular ejection fraction (LVEF), ivabradine im-
tive medications in oncologic patients may exacerbate proves functional class and LVEF, reduces hospitaliza-
dizziness, hypotension, and fatigue, which are likely to tion for heart failure, and cardiovascular death [10]. Sig-
East Carolina University - Laupus Library
150.216.68.200 - 3/7/2019 11:51:44 AM
316 Chemotherapy 2018;63:315–320 Sarocchi et al.
DOI: 10.1159/000495576
Downloaded by:
Table 1. Echocardiographic parameters during the study
Echocardiography Baseline visit Follow-up visit p
mean SD mean SD
LV EDD, mm 50.2 5.4 50.5 4.1 0.64
LV ESD, mm 35.4 5.2 32.2 4.8 <0.001
LV EF, % 45.1 6.4 53.2 3.9 <0.001
WMSI 1.22 0.21 1.12 0.14 <0.001
Left atrial diameter, mm 37.5 7.8 38.0 7.0 0.39
PASP estimated, mm Hg 31.4 7.8 32.4 10.4 0.61
Mitral regurgitation Patients % Patients % p
Mild 21 70 20 67 0.04
Moderate 6 20 5 17
Severe 1 3 0 0
LV mass calculated by the Deauville formula. EDD, end-diastolic diameter; EF, ejection fraction; ESD, end-
systolic diameter; PASP, pulmonary artery systolic pressure; WMSI, wall motion score index.
nificant hypotension, fatigue, and severe bradycardia are Results
uncommon, due to its peculiar mechanism of action [11].
For all these reasons, ivabradine could be well tolerated in Of 37 CTrLVD patients treated with ivabradine, 7
cancer-treated patients. were excluded because they did not have clinical and
The aim of our study was to confirm the efficacy and echocardiographic evaluations after ivabradine introduc-
handling of ivabradine treatment in cancer patients with tion owing to missing follow-up visits (n = 3), noncardiac
CTrLVD. death (n = 3), or early ivabradine discontinuation (n = 1)
decided by the general practitioner without providing
any reason.
Patients and Methods The study cohort included 30 patients (23 females, 7
males) with a median age of 54 years (interquartile range:
A retrospective observational analysis of patients with CTrLVD
44–66). Eleven patients were smokers, while patients with
treated with ivabradine was performed at the Cardio-Oncology
Outpatient Clinic of the San Martino Policlinic Hospital in Genoa hypertension, type 2 diabetes, and dyslipidemia were 9, 2,
from 2014 to 2017. and 14, respectively. Six patients had stable ischemic or
We included patients with a CTrLVD defined as (1) a reduction nonischemic cardiac disease before CTrLVD. Breast can-
of LVEF below 50% if previously normal (≥55%), or (2) >10 per- cer and hematological diseases accounted for 40 and 57%
centage point loss if previously subnormal, in the absence of any
of the diagnoses of malignancy, respectively. CTrLVD
other explanation. Since anticancer treatments may be withheld,
stopped, or changed when a cardiovascular complication occurs, was attributed to anthracyclines (n = 6), trastuzumab
we considered a “change of treatment” any unscheduled delay or (n = 11), or allogeneic stem cell transplant (n = 10) in
variation lasting for more than two cycles of the cancer therapy. most cases. The mean duration of follow-up was 6.5
Continuous variables are presented as mean and standard de- months (median 5.6, interquartile range: 4.2–7.2).
viation, unless otherwise specified. Hypothesis testing was per-
The main clinical and echocardiographic results are
formed using Student’s t test for paired data (continuous variables)
and the Wilcoxon signed-rank test (ordinal variables), and a two- represented in Figure 1 and Table 1. NYHA class amelio-
tailed p < 0.05 was considered significant. The duration of the ob- rated in 11 patients, while 1 patient referred functional
servation period was normalized by log-transformation. Correla- class worsening, but further testing revealed an opportu-
tions between LVEF change and other continuous variables were nistic pneumonia. Eight patients reported resolution or
assessed by Pearson’s correlation test, while Kendall’s test was used
improvement of fatigue. Mean LVEF increased from
for New York Heart Association (NYHA) class. Associations in-
volving LVEF change were also examined in a univariate and mul- 45.1% (SD = 6.4) to 53.2% (SD = 3.9; p < 0.001). The ex-
tivariate linear regression model with heart rate change and the tent of LVEF improvement was slightly correlated to a
observation period as covariates. longer observation period (r2 = 0.18; p = 0.02). Neverthe-
East Carolina University - Laupus Library
150.216.68.200 - 3/7/2019 11:51:44 AM
Ivabradine in Cardiotoxicity Chemotherapy 2018;63:315–320 317
DOI: 10.1159/000495576
Downloaded by:
Table 2. Ivabradine and other cardiovascular drugs in the study
25
Cardiovascular treatment Baseline Follow-up p
visit visit
20
r2 = 0.22
patients % patients %
15
Ivabradine
LVEF change, %
2.5 mg/b.i.d. 18 60 13 43 0.06
10 5 mg/b.i.d. 12 40 15 50
7.5 mg/b.i.d. 0 0 2 7
5 BB
<25% target dose 4 13 3 10 0.78
25 to <50% target dose 6 20 6 20
0
≥50% target dose* 2 7 3 10
ACEi or ARB 13 43 14 47 1.00
−5 Loop diuretics 9 30 13 43 0.07
−60 −40 −20 0 20 MRA 4 13 9 30 0.03
HR change, bpm
ACEi, angiotensin-converting enzyme inhibitor; ARB,
angiotensin receptor blocker; BB, β-blockers; MRA, mineral-
corticoid receptor blocker.
Fig. 2. Heart rate (HR) and left ventricular ejection fraction (LVEF)
* Target dose: bisoprolol 10 mg o.d.; carvedilol 25 mg b.i.d.;
changes from baseline after ivabradine. Univariate model (r2 =
nebivolol 10 mg o.d.; metoprolol succinate 200 mg o.d. (as defined
0.22, β = –0.47, p < 0.01). In a multivariate model including the
in the 2016 guidelines for the diagnosis and treatment of acute and
log-transformed observation period (OP), the HR relation to
chronic heart failure by the European Society of Cardiology).
LVEF remained significant (HR: β = –0.45, p < 0.01; OP: β = 0.40,
p = 0.01; r2 = 0.38, p < 0.01).
less, LVEF improvement was significant in the subgroup respectively) in patients who did not require diuretic,
of 15 patients observed in an early time window before ACEi/ARB, and BB dose increase.
the median value of the study follow-up (from 47.0% In 8 patients, the oncologist changed the anticancer
SD = 2.9–53.6 SD = 3.2, p < 0.001). No relation between treatments to prevent a more severe scenario of cardiac
NYHA class changes and the observation period was impairment. They had a lower LVEF and a more severe
found (tau = 0.04, p = 0.78). NYHA class, but the improvement was not greater in
Of note, a significant correlation (r2 = 0.22; p < 0.01) these patients when compared to those who continued
between heart rate reduction and LVEF improvement treatment. Serum troponin I was minimally altered in 12
was found. The strength of the association with heart rate patients (TnI+) in at least one determination during an-
did not change when the observation period was added in ticancer treatment, without clinical or electrocardio-
a multivariate model (Fig. 2). graphic signs of myocardial ischemia. LVEF improve-
Ivabradine was chosen because blood pressure val- ment was similar in TnI+ and TnI– patients. Likewise, the
ues, fatigue, dizziness, and drug intolerance cautioned LVEF increase was consistent across cancer treatment
against the introduction or the increase of BB and an groups. Notably, all anthracycline treated patients ame-
ACEi/ARB. Hypotension (77%) and fatigue (70%) were liorated, 4 out of them showing more than 10 percentage
the most common causes limiting the treatment with point gain of LVEF.
these drugs. Anemia did not have a significant effect in the study.
Concomitant cardiovascular medications are shown The hemoglobin value did not change significantly dur-
in Table 2. BB and ACEi/ARB doses did not change in ing follow-up, and only 5 patients had a hemoglobin val-
most patients and were slightly increased in few patients. ue below 10.0 g/dL, but still >9.0 g/dL.
The dose of loop diuretic and mineralocorticoid receptor Ivabradine was well tolerated in all patients. No seri-
antagonist was increased in 7 patients, but NYHA class ous symptomatic bradycardia was reported. Two patients
did not ameliorate in 6 of them. NYHA class and LVEF reported mild phosphine-type visual disturbances soon
improvements were significant (p < 0.05 and p < 0.001, after drug initiation, which are a well-known, usually re-
East Carolina University - Laupus Library
150.216.68.200 - 3/7/2019 11:51:44 AM
318 Chemotherapy 2018;63:315–320 Sarocchi et al.
DOI: 10.1159/000495576
Downloaded by:
versible side effect. Accordingly, a brief, temporary sus- To the best of our knowledge, no previous clinical
pension of ivabradine was enough to resolve the symp- study investigated the role of ivabradine in the treatment
toms completely. of CTrLVD. De Gregorio et al. [19] reported a case of se-
vere cardiotoxicity responsive to a lisinopril-ivabradine-
multivitamin treatment, while Colak et al. [20] showed
Discussion that ivabradine pretreatment mitigates doxorubicin-in-
duced hemodynamic and biomarker alteration in rats.
Our study documents that the addition of ivabradine Noteworthy, patients with anthracycline cardiotoxic-
to the best medical therapy improves LVEF and symp- ity – widely considered not reversible – showed a relevant
toms in patients with CTrLVD. The LVEF improvement LVEF improvement in our study. This result may depend
did not come at the expense of worsening symptoms, as on how quickly we initiated the cardiovascular treatment
may happen when BB or ACEi/ARB are uptitrated but, [3]. In our cardio-oncology outpatient clinic, we evaluate
on the contrary, joined a remarkable improvement of fa- asymptomatic patients at risk of cardiotoxicity to identify
tigue, palpitations, and effort tolerance. This point is of CTrLVD as soon as possible and to start treatment with-
paramount importance for cancer patients. In fact, the out delay [21]. The small number of withdrawal of anti-
above-quoted symptoms not only impair the quality of cancer treatment indicates that such practice is clinically
life but also limit the compliance to cancer therapies, effective. This course of action is consistent with the mod-
worsening the clinical outcome. ern concept of “actionable cardiotoxicity” that suggests
Ivabradine was additional, not alternative, to standard not to deprive patients of highly effective anticancer drugs
treatment. Following the introduction of ivabradine, because of cardiotoxicity but to adopt all the diagnostic
there were minor therapy adjustments, including the in- and therapeutic tools, such as ivabradine, to mitigate the
troduction of ACEi/ARB in 1 patient and a small uptitra- toxicity [7].
tion of BB in few patients. This more effective use of ACEi This is a retrospective study with major limitations.
and BB that was eased by better clinical conditions – pos- Ivabradine in the cardio-oncologic setting was pre-
sibly achieved by the addition of ivabradine – can in turn scribed only in selected patients. As a result, our study
have contributed to improve symptoms and LVEF. Con- has a small sample size and lacks a control group, which
versely, the addition of loop diuretic and/or mineralocor- could not be matched by the propensity score. In addi-
ticoid receptor antagonist appears not to be associated tion, patients were collected during a long-time span,
with a better outcome. and the different periods of observation may have influ-
Notably, the significant correlation that was found be- enced the study findings. LVEF improved more in pa-
tween heart rate reduction and LVEF improvements sup- tients with a longer observation period, while changes in
ports the pivotal role of heart rate in the pathophysiology the NYHA class were not affected by the length of ob-
of LVD and heart failure and provides an explanation of servation time.
the mechanism through which ivabradine exerts its ben- Prospective investigations are needed to overcome
eficial effects in CTrLVD. these limitations and to support our results that confirm
We should not forget that an association between the efficacy and safety in cancer patients with CTrLVD
heart rate and cancer mortality had already been docu- previously seen in a large heart failure trial [10].
mented [12]. Moreover, a recent prospective study
showed that heart rate is associated with mortality in
patients with different types of cancer, irrespective of Conclusions
other comorbidities, particularly infections and ane-
mia [13]. Heart rate therefore has been seen as a poten- This is the first structured study that looked at the use
tial modifiable risk factor [14] that, based on these of ivabradine in cardio-oncology. Data derived by our ob-
findings, can be treated pharmacologically. Unlike BB, servations support the claim that ivabradine might im-
ivabradine reduces the heart rate without causing prove LVEF, NYHA class, and symptoms in patients with
hypotension and improves diastolic function, which CTrLVD. When frailty limits other cardiovascular thera-
seems to precede CTrLVD [15]. Moreover, anticancer pies in cancer patients, ivabradine might allow a prompt
treatments cause substantial endothelial dysfunction treatment of cardiotoxicity to avoid irreversible cardiac
[16, 17] that may be reduced through heart rate reduc- damage and to preserve anticancer treatment opportuni-
tion with ivabradine [18]. ties.
East Carolina University - Laupus Library
150.216.68.200 - 3/7/2019 11:51:44 AM
Ivabradine in Cardiotoxicity Chemotherapy 2018;63:315–320 319
DOI: 10.1159/000495576
Downloaded by:
Statement of Ethics Funding Sources
The study was approved by the local institutional review board. This study received no external funding.
All procedures in the study were in accordance with national stan-
dards and the Helsinki declaration.
Author Contributions
Disclosure Statement Study design: M.S., E.A., G.G., C. Brunelli, P.S. Manuscript ed-
iting: M.S., G.G., P.S. Data collection and analysis: M.S., R.M., C.
Dr. Paolo Spallarossa received speaker honoraria from Servier. Bighin, F.G., V.S., M.B., P.S. Manuscript revision and approval: all
The other authors have no conflicts of interest to disclose. authors.
References
1 Zamorano JL, Lancellotti P, Rodriguez Mu- 7 Ewer M, Gianni L, Pane F, Sandri MT, Steiner 15 Calabrese V, Menna P, Annibali O, Armento
ñoz D, Aboyans V, Asteggiano R, Galderisi M, RK, Wojnowski L, et al. Report on the in G, Carpino A, Cerchiara E, et al.; Collabora-
et al.; ESC Scientific Document Group. 2016 ternational colloquium on cardio-oncology tors. Early Diastolic Dysfunction after Cancer
ESC Position Paper on cancer treatments and (Rome, 12–14 March 2014). Ecancermedi- Chemotherapy: Primary Endpoint Results of
cardiovascular toxicity developed under the calscience. 2014 May;8:433. a Multicenter Cardio-Oncology Study. Che-
auspices of the ESC Committee for Practice 8 Rosa GM, Ferrero S, Ghione P, Valbusa A, motherapy. 2018;63(2):55–63.
Guidelines: the Task Force for cancer treat- Brunelli C. An evaluation of the pharma 16 Wolf MB, Baynes JW: The anti-cancer drug,
ments and cardiovascular toxicity of the Eu- cokinetics and pharmacodynamics of iv- doxorubicin, causes oxidant stress-induced
ropean Society of Cardiology (ESC). Eur abradine for the treatment of heart failure. endothelial dysfunction. Biochim Biophys
Heart J. 2016 Sep;37(36):2768–801. Expert Opin Drug Metab Toxicol. 2014 Feb; Acta. 2006 Feb;1760(2):267–271.
2 Minotti G, Salvatorelli E, Menna P. Pharma- 10(2): 279–91. 17 Sandoo A, Kitas GD, Carmichael AR. Endo-
cological foundations of cardio-oncology. J 9 Koruth JS, Lala A, Pinney S, Reddy VY, thelial dysfunction as a determinant of trastu-
Pharmacol Exp Ther. 2010 Jul;334(1):2–8. Dukkipati SR. The Clinical Use of Iv- zumab-mediated cardiotoxicity in patients
3 Cardinale D, Colombo A, Lamantia G, Co- abradine. J Am Coll Cardiol. 2017 Oct; with breast cancer. Anticancer Res. 2014 Mar;
lombo N, Civelli M, De Giacomi G, et al. An- 70(14): 1777–84. 34(3):1147–51.
thracycline-induced cardiomyopathy: clinical 10 Swedberg K, Komajda M, Böhm M, Borer JS, 18 Mangiacapra F, Colaiori I, Ricottini E, Balduc-
relevance and response to pharmacologic Ford I, Dubost-Brama A, et al.; SHIFT Inves- ci F, Creta A, Demartini C, et al. Heart Rate
therapy. J Am Coll Cardiol. 2010 Jan; 55(3): tigators. Ivabradine and outcomes in chronic reduction by IVabradine for improvement of
213–20. heart failure (SHIFT): a randomised placebo- ENDothELial function in patients with coro-
4 Gulati G, Heck SL, Ree AH, Hoffmann P, controlled study. Lancet. 2010 Sep;376(9744): nary artery disease: the RIVENDEL study. Clin
Schulz-Menger J, Fagerland MW, et al. Pre- 875–85. Res Cardiol. 2017 Jan;106(1):69–75.
vention of cardiac dysfunction during adju- 11 Savelieva I, Camm AJ. I f Inhibition with iv- 19 de Gregorio C, Potenza G, Ferraro G. Effec-
vant breast cancer therapy (PRADA): a 2 × 2 abradine: electrophysiological effects and tiveness of the combination therapy with
factorial, randomized, placebo-controlled, safety. Drug Saf. 2008;31(2):95–107. lisinopril, ivabradine and multivitamin sup-
double-blind clinical trial of candesartan and 12 Jouven X, Escolano S, Celermajer D, Empana plementation in anthracycline-induced se-
metoprolol. Eur Heart J. 2016 Jun; 37(21): JP, Bingham A, Hermine O, et al. Heart rate vere cardiotoxicity. Int J Cardiol. 2014 Oct;
1671–80. and risk of cancer death in healthy men. PLoS 176(3):1374–6.
5 Plana JC, Galderisi M, Barac A, Ewer MS, Ky One. 2011;6(8):e21310. 20 Colak MC, Parlakpinar H, Tasdemir S, Sam-
B, Scherrer-Crosbie M, et al. Expert consen- 13 Anker MS, Ebner N, Hildebrandt B, Springer danci E, Kose E, Polat A, et al. Therapeutic
sus for multimodality imaging evaluation of J, Sinn M, Riess H, et al. Resting heart rate is effects of ivabradine on hemodynamic pa-
adult patients during and after cancer thera- an independent predictor of death in patients rameters and cardiotoxicity induced by doxo-
py: a report from the American Society of with colorectal, pancreatic, and non-small cell rubicin treatment in rat. Hum Exp Toxicol.
Echocardiography and the European Associ- lung cancer: results of a prospective cardio- 2012 Sep;31(9):945–54.
ation of Cardiovascular Imaging. J Am Soc vascular long-term study. Eur J Heart Fail. 21 Spallarossa P, Maurea N, Cadeddu C, Madon-
Echocardiogr. 2014 Sep;27(9):911–39. 2016 Dec;18(12):1524–34. na R, Mele D, Monte I, et al. A recommended
6 Spallarossa P, Guerrini M, Arboscello E, 14 Nikolovska Vukadinović A, Vukadinović D, practical approach to the management of an-
Sicbaldi V. Enalapril and carvedilol for pre- Borer J, Cowie M, Komajda M, Lainscak M, et thracycline-based chemotherapy cardiotoxici-
venting chemotherapy-induced left ventricu- al. Heart rate and its reduction in chronic ty: an opinion paper of the working group on
lar systolic dysfunction. J Am Coll Cardiol. heart failure and beyond. Eur J Heart Fail. drug cardiotoxicity and cardioprotection, Ital-
2013 Dec;62(25):2451–2. 2017 Oct;19(10):1230–41. ian Society of Cardiology. J Cardiovasc Med
(Hagerstown). 2016 May;17 Suppl 1:S84–92.
East Carolina University - Laupus Library
150.216.68.200 - 3/7/2019 11:51:44 AM
320 Chemotherapy 2018;63:315–320 Sarocchi et al.
DOI: 10.1159/000495576
Downloaded by:
Anda mungkin juga menyukai
- Grit: The Power of Passion and PerseveranceDari EverandGrit: The Power of Passion and PerseverancePenilaian: 4 dari 5 bintang4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)Dari EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Penilaian: 4 dari 5 bintang4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDari EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifePenilaian: 4 dari 5 bintang4/5 (5795)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDari EverandNever Split the Difference: Negotiating As If Your Life Depended On ItPenilaian: 4.5 dari 5 bintang4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDari EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RacePenilaian: 4 dari 5 bintang4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDari EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersPenilaian: 4.5 dari 5 bintang4.5/5 (345)
- Shoe Dog: A Memoir by the Creator of NikeDari EverandShoe Dog: A Memoir by the Creator of NikePenilaian: 4.5 dari 5 bintang4.5/5 (537)
- The Little Book of Hygge: Danish Secrets to Happy LivingDari EverandThe Little Book of Hygge: Danish Secrets to Happy LivingPenilaian: 3.5 dari 5 bintang3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDari EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FuturePenilaian: 4.5 dari 5 bintang4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDari EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryPenilaian: 3.5 dari 5 bintang3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealDari EverandOn Fire: The (Burning) Case for a Green New DealPenilaian: 4 dari 5 bintang4/5 (74)
- The Emperor of All Maladies: A Biography of CancerDari EverandThe Emperor of All Maladies: A Biography of CancerPenilaian: 4.5 dari 5 bintang4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDari EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaPenilaian: 4.5 dari 5 bintang4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaDari EverandThe Unwinding: An Inner History of the New AmericaPenilaian: 4 dari 5 bintang4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnDari EverandTeam of Rivals: The Political Genius of Abraham LincolnPenilaian: 4.5 dari 5 bintang4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDari EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyPenilaian: 3.5 dari 5 bintang3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDari EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You ArePenilaian: 4 dari 5 bintang4/5 (1091)
- Rise of ISIS: A Threat We Can't IgnoreDari EverandRise of ISIS: A Threat We Can't IgnorePenilaian: 3.5 dari 5 bintang3.5/5 (137)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Dari EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Penilaian: 4.5 dari 5 bintang4.5/5 (121)
- Her Body and Other Parties: StoriesDari EverandHer Body and Other Parties: StoriesPenilaian: 4 dari 5 bintang4/5 (821)
- Green Energy BricksDokumen8 halamanGreen Energy BricksSriram NandipatiBelum ada peringkat
- Sentinel Node Biopsy For CA BreastDokumen7 halamanSentinel Node Biopsy For CA Breastwasima1956Belum ada peringkat
- Olper's Brand AuditDokumen60 halamanOlper's Brand AuditUsama NaseemBelum ada peringkat
- Final Exam Review AnswerDokumen6 halamanFinal Exam Review AnswerJosh ClickBelum ada peringkat
- Neuro Reviewer 2Dokumen33 halamanNeuro Reviewer 2joanneBelum ada peringkat
- Nissan RPX80 Vs Crown PE4500Dokumen13 halamanNissan RPX80 Vs Crown PE4500Forklift Systems IncorporatedBelum ada peringkat
- People in OrganisationsDokumen8 halamanPeople in OrganisationsBritney valladares100% (1)
- Biphasic Liquid Dosage FromDokumen8 halamanBiphasic Liquid Dosage FromSwaroopSinghJakhar100% (1)
- Fuji FCR 5000Dokumen435 halamanFuji FCR 5000Lion Micheal OtitolaiyeBelum ada peringkat
- Reliance Individual Mediclaim Policy ScheduleDokumen1 halamanReliance Individual Mediclaim Policy ScheduleKoushik Dutta38% (13)
- Outlet Temperature HighDokumen5 halamanOutlet Temperature HighOzan Gürcan100% (3)
- Literature Review Devin KilburnDokumen4 halamanLiterature Review Devin Kilburnapi-549230129Belum ada peringkat
- Hcs CatalogueDokumen16 halamanHcs CatalogueG MuratiBelum ada peringkat
- Ayoade Kalejaiye: Learning EffectivenessDokumen4 halamanAyoade Kalejaiye: Learning EffectivenessOlabooye AyodejiBelum ada peringkat
- SLL VS NLRCDokumen11 halamanSLL VS NLRCJepski ScopeBelum ada peringkat
- Ayurvedic and Naturopathic Way of Life by Manthena Sathyanarayana RajuDokumen7 halamanAyurvedic and Naturopathic Way of Life by Manthena Sathyanarayana RajuvishnuprakashBelum ada peringkat
- 1HDH 118 041 en - Rev. IDokumen53 halaman1HDH 118 041 en - Rev. IKhắc Đồng Trần100% (1)
- 11 - Chapter 4 Muslim Law PDFDokumen87 halaman11 - Chapter 4 Muslim Law PDFAnfal BarbhuiyaBelum ada peringkat
- Mind Body Relationship in PsychotherapyDokumen8 halamanMind Body Relationship in Psychotherapymoyosore100% (1)
- COVID 19 Pandemic Lifestyle Students MenDokumen173 halamanCOVID 19 Pandemic Lifestyle Students MenwalkensiocsBelum ada peringkat
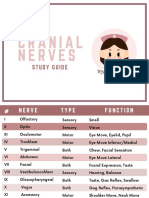
- Cranial NervesDokumen6 halamanCranial Nervesvienny kayeBelum ada peringkat
- Fs Chapter I Vi ApprovalDokumen119 halamanFs Chapter I Vi ApprovalPauline OrtegaBelum ada peringkat
- Section A: Jawab Semua SoalanDokumen3 halamanSection A: Jawab Semua SoalanAzreen IzetBelum ada peringkat
- 2018 Bfa NatWest Franchise SurveyDokumen28 halaman2018 Bfa NatWest Franchise SurveyPapa Johny100% (1)
- TOXICITY of FLOURIDESDokumen45 halamanTOXICITY of FLOURIDESNavneet KaurBelum ada peringkat
- Steca TR 0201 Instruction enDokumen40 halamanSteca TR 0201 Instruction endaviko313Belum ada peringkat
- Auto Collision 1Dokumen2 halamanAuto Collision 1api-252290060Belum ada peringkat
- O Feci : (BL (ElDokumen107 halamanO Feci : (BL (ElKaren Elsy Gonzales CamachoBelum ada peringkat
- SafetyMAP Measuring ManagementDokumen4 halamanSafetyMAP Measuring Managementindri anggrainiBelum ada peringkat
- Epithelial TissueDokumen10 halamanEpithelial Tissuememe bolongonBelum ada peringkat