Homework: Advanced Chemical Reaction Engineering II
Diunggah oleh
Jelor GallegoDeskripsi Asli:
Judul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Homework: Advanced Chemical Reaction Engineering II
Diunggah oleh
Jelor GallegoHak Cipta:
Format Tersedia
Department of Chemical Engineering
College of Engineering
University of the Philippines
Diliman, Quezon City
Second Semester AY2017-2018
ChE 126 Advanced Chemical Reaction Engineering II
HOMEWORK
Due 20 April, 8:30 am, before the start of the lecture
Show your complete solution. State all assumptions.
PROBLEM
A second-order heterogeneous irreversible reaction 2A → B is taking place within a cylindrical catalyst
pellet which is plated with platinum throughout the pellet. The reactant concentration halfway between
the external surface and the center of the pellet is a tenth of the concentration at the external surface. The
concentration at the external surface is 0.001 M, the diameter is 210-3 cm and length 110-2 cm, and the
diffusion coefficient is 0.05 cm2/s.
a. What is the concentration of reactant at a distance of 610-4 cm from the external pellet surface?
b. What is the concentration of reactant at the center of the catalyst pellet?
c. What is the effectiveness factor?
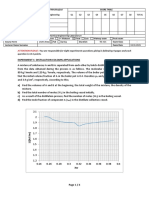
d. How does the effectiveness factor vary with the Thiele modulus? Show graphically.
e. To what diameter should the pellet be reduced if the effectiveness factor is to be 0.75?
Anda mungkin juga menyukai
- Redox Reaction: Discover How Batteries Work: Experiment No. 4Dokumen8 halamanRedox Reaction: Discover How Batteries Work: Experiment No. 4theressaBelum ada peringkat
- ChE 140 1.01 IntroductionDokumen30 halamanChE 140 1.01 IntroductionJelor GallegoBelum ada peringkat
- C H O + A O + B NH C C H NO + D H O+eCO: InstructionsDokumen4 halamanC H O + A O + B NH C C H NO + D H O+eCO: InstructionsJohn Paul Jandayan33% (3)
- Role of Metal Ions in Dopamine Oxidation (1) (Autorecovered)Dokumen19 halamanRole of Metal Ions in Dopamine Oxidation (1) (Autorecovered)intresting videoBelum ada peringkat
- Angelidaki 2009Dokumen8 halamanAngelidaki 2009Nixon AlfonsoBelum ada peringkat
- Experiment 2Dokumen4 halamanExperiment 2Ynah CasiaBelum ada peringkat
- Bulsu-Cs Not For Sale: Unit Title: Review of Mathematical Concepts and Fundamentals of Chemistry Title of The LessonDokumen38 halamanBulsu-Cs Not For Sale: Unit Title: Review of Mathematical Concepts and Fundamentals of Chemistry Title of The Lessonprettyrose0% (1)
- CEE 523 MIDTERM EXAM 2nd Sem 2019 2020Dokumen1 halamanCEE 523 MIDTERM EXAM 2nd Sem 2019 2020Jat BacsafraBelum ada peringkat
- Syllabus OuDokumen26 halamanSyllabus OuTharun KumarBelum ada peringkat
- Treenate 2017 IOP Conf. Ser. Mater. Sci. Eng. 222 012010Dokumen7 halamanTreenate 2017 IOP Conf. Ser. Mater. Sci. Eng. 222 012010Kanishka PriyankaraBelum ada peringkat
- PPT II Plastics Materials and Its Applications II 20 221Dokumen1 halamanPPT II Plastics Materials and Its Applications II 20 221Raj ThakurBelum ada peringkat
- PPT II Plastics Materials and Its Applications II 20 221Dokumen1 halamanPPT II Plastics Materials and Its Applications II 20 221Raj ThakurBelum ada peringkat
- Chemical Reaction EngineeringDokumen11 halamanChemical Reaction Engineeringkushaal narothamBelum ada peringkat
- 15CHE111 - Spring 2019 ProjectDokumen5 halaman15CHE111 - Spring 2019 ProjectPunith KumarBelum ada peringkat
- FinalDokumen4 halamanFinalSimge DemirBelum ada peringkat
- Identify Whether The Following Processes Are Endothermic or Exothermic. Justify Your Answer On The Space ProvidedDokumen5 halamanIdentify Whether The Following Processes Are Endothermic or Exothermic. Justify Your Answer On The Space ProvidedAngelique BarquillaBelum ada peringkat
- Final Theory Exam-307 Dec11-FinalDokumen14 halamanFinal Theory Exam-307 Dec11-FinalJagadeesh EllilBelum ada peringkat
- Peter 2021Dokumen14 halamanPeter 2021Radouane El-AmriBelum ada peringkat
- Internship ReportDokumen10 halamanInternship Reportdeepan kumarBelum ada peringkat
- Engineering Chemistry PDFDokumen293 halamanEngineering Chemistry PDFAnonymous tdtTl8KypBelum ada peringkat
- Atom EkonomiDokumen16 halamanAtom Ekonomisman24 kabtangerangBelum ada peringkat
- 30 05 16 UAS Kimia FisikaDokumen3 halaman30 05 16 UAS Kimia FisikaNIABelum ada peringkat
- Ipca ReportDokumen47 halamanIpca Reportevil kingBelum ada peringkat
- Attempt Only 3 Questions: Final Examination / 1 Attempt / First SemesterDokumen1 halamanAttempt Only 3 Questions: Final Examination / 1 Attempt / First Semesterali alkassemBelum ada peringkat
- Angew Chem Int Ed - 2014 - Maturi - Enantioselective Catalysis of The Intermolecular 2 2 Photocycloaddition BetweenDokumen4 halamanAngew Chem Int Ed - 2014 - Maturi - Enantioselective Catalysis of The Intermolecular 2 2 Photocycloaddition BetweenDicky Tak Hin WongBelum ada peringkat
- Synthesis and Characterization of Red, Green and Blue Pigments For High Temperature ResistantDokumen6 halamanSynthesis and Characterization of Red, Green and Blue Pigments For High Temperature Resistantsunny butterBelum ada peringkat
- Final Theory Exam-307 June2012Dokumen13 halamanFinal Theory Exam-307 June2012Jagadeesh EllilBelum ada peringkat
- Vibration Damping Mechanism of CuAlMnPolymerCarbon NanomaterialsDokumen13 halamanVibration Damping Mechanism of CuAlMnPolymerCarbon Nanomaterialsrafael.ramalhoBelum ada peringkat
- Masturi JPCS 2018Dokumen6 halamanMasturi JPCS 2018masturiBelum ada peringkat
- 2017 Noor JMES SurfaceandthermodynamicstudyofmicellizationofnonionicsurfactantDokumen8 halaman2017 Noor JMES SurfaceandthermodynamicstudyofmicellizationofnonionicsurfactantArian VelayatiBelum ada peringkat
- Essay 01Dokumen10 halamanEssay 01maedeh.216.froBelum ada peringkat
- Instructions:: Michelle C. Almendrala, PHD 4Q 2016-2017 School of Chemical Engineering and Chemistry. Mapua UniversityDokumen2 halamanInstructions:: Michelle C. Almendrala, PHD 4Q 2016-2017 School of Chemical Engineering and Chemistry. Mapua UniversityNikki EbañezBelum ada peringkat
- Bit Campus, Anna University Tiruchirappalli - 620 024 First Internal Assessment Year & Semester: FINAL & VII SemesterDokumen1 halamanBit Campus, Anna University Tiruchirappalli - 620 024 First Internal Assessment Year & Semester: FINAL & VII SemesterSuki SudharsanBelum ada peringkat
- Of Method': Study C.hemica1 Engineering by Unit-OperationDokumen4 halamanOf Method': Study C.hemica1 Engineering by Unit-OperationRudrajyoti MishraBelum ada peringkat
- Template2 1 PDFDokumen2 halamanTemplate2 1 PDFJack BadanBelum ada peringkat
- ChE 401 CRD Assignment-4Dokumen3 halamanChE 401 CRD Assignment-4Husnain GujjarBelum ada peringkat
- Appendix Chemical Engineering 34Dokumen154 halamanAppendix Chemical Engineering 34JAGATHA NAGESWARA RAOBelum ada peringkat
- Enchem Activity 2 SubstanceDokumen4 halamanEnchem Activity 2 SubstanceNala, Vangie A.Belum ada peringkat
- Gas Diffusion in Polymer Nanocomposites Role of Defects and Caves in FillersDokumen13 halamanGas Diffusion in Polymer Nanocomposites Role of Defects and Caves in Fillerswan jianfengBelum ada peringkat
- File 2023021766377Dokumen68 halamanFile 2023021766377ApurvaBelum ada peringkat
- Board Class XII Worksheet KineticsDokumen3 halamanBoard Class XII Worksheet KineticsShazia FarheenBelum ada peringkat
- SyllabusDokumen4 halamanSyllabusemhufafBelum ada peringkat
- Alumina Particle size-KPMDokumen7 halamanAlumina Particle size-KPMrajeshayrBelum ada peringkat
- Thesis On CatalystDokumen36 halamanThesis On CatalystMahfuzur Rahman SiddikyBelum ada peringkat
- Experiment 1A: Continuous Stirred Tank Reactor: Department of Chemical EngineeringDokumen1 halamanExperiment 1A: Continuous Stirred Tank Reactor: Department of Chemical EngineeringKelly Sheine SisonBelum ada peringkat
- JHKJHDokumen2 halamanJHKJHDonPedrewBelum ada peringkat
- J Cjche 2018 08 020Dokumen35 halamanJ Cjche 2018 08 020DEV RAJBelum ada peringkat
- A Fast and Room-Temperature Self-Healing Thermal Conductive Polymer CompositeDokumen9 halamanA Fast and Room-Temperature Self-Healing Thermal Conductive Polymer CompositeMd. Abdur RashidBelum ada peringkat
- B.E. (AI & ML) SyllabusDokumen26 halamanB.E. (AI & ML) SyllabusJeevan SaiBelum ada peringkat
- Summative Test in ScienceDokumen2 halamanSummative Test in ScienceprecillaugartehalagoBelum ada peringkat
- Summative Test in ScienceDokumen2 halamanSummative Test in ScienceprecillaugartehalagoBelum ada peringkat
- 10 1016@j Compscitech 2017 06 008Dokumen30 halaman10 1016@j Compscitech 2017 06 008ramesh tBelum ada peringkat
- Introduction To BioengineeringDokumen1 halamanIntroduction To Bioengineeringhuni matataBelum ada peringkat
- ProfileDokumen3 halamanProfileAiman AmirullahBelum ada peringkat
- General Chemistry (Chem101) : September 2017 Department of Chemistry BuetDokumen2 halamanGeneral Chemistry (Chem101) : September 2017 Department of Chemistry BuetA. F. M. Mahfuzul KabirBelum ada peringkat
- Soham LolageDokumen1 halamanSoham LolagepranavBelum ada peringkat
- Final - 1nd Semesterالدور الثانيDokumen12 halamanFinal - 1nd Semesterالدور الثانيAli aliraqiBelum ada peringkat
- Li 2020Dokumen11 halamanLi 2020shubh shahBelum ada peringkat
- Engineering Chemistry 1ST YEAR LMDokumen111 halamanEngineering Chemistry 1ST YEAR LMSANJEEV100% (1)
- Civil PDFDokumen117 halamanCivil PDFinam ul haqBelum ada peringkat
- ChE 131 PS-MassDokumen1 halamanChE 131 PS-MassJelor GallegoBelum ada peringkat
- Che 131 Ps-Heat1Dokumen1 halamanChe 131 Ps-Heat1Jelor GallegoBelum ada peringkat
- Che 131 Ps-Heat2Dokumen1 halamanChe 131 Ps-Heat2Jelor GallegoBelum ada peringkat
- 131.8 Introduction To Heat TransferDokumen41 halaman131.8 Introduction To Heat TransferJelor GallegoBelum ada peringkat
- 131.11c Unsteady-State Heat TransferDokumen25 halaman131.11c Unsteady-State Heat TransferJelor GallegoBelum ada peringkat
- 131.10a Shell Energy BalanceDokumen28 halaman131.10a Shell Energy BalanceJelor GallegoBelum ada peringkat
- 131.11b Heat Equations of Change Part 2Dokumen20 halaman131.11b Heat Equations of Change Part 2Jelor GallegoBelum ada peringkat
- ChE 134 Attrition Mill - GuanlaoDokumen1 halamanChE 134 Attrition Mill - GuanlaoJelor Gallego100% (1)
- Che 125: Chemical Reaction Engineering IDokumen2 halamanChe 125: Chemical Reaction Engineering IJelor GallegoBelum ada peringkat
- 131.15 Equations of ContinuityDokumen25 halaman131.15 Equations of ContinuityJelor GallegoBelum ada peringkat
- Lecture 3 - Independent and Dependent ReactionsDokumen19 halamanLecture 3 - Independent and Dependent ReactionsJelor GallegoBelum ada peringkat
- Mibk MSDS PDFDokumen5 halamanMibk MSDS PDFJelor GallegoBelum ada peringkat
- Eq Uitf Bpi Gefof Nov 2017Dokumen4 halamanEq Uitf Bpi Gefof Nov 2017Jelor GallegoBelum ada peringkat
- Greening The Roadmap of The Philippine Pulp and Paper IndustryDokumen14 halamanGreening The Roadmap of The Philippine Pulp and Paper IndustryJelor Gallego100% (1)
- World Voyager Fund - March 2016Dokumen41 halamanWorld Voyager Fund - March 2016Jelor GallegoBelum ada peringkat
- 1 s2.0 S0263876299718186 Main PDFDokumen7 halaman1 s2.0 S0263876299718186 Main PDFLeydi PatiñoBelum ada peringkat
- BPI US Equity Feeder Fund Latest DisclosureDokumen3 halamanBPI US Equity Feeder Fund Latest DisclosureJelor GallegoBelum ada peringkat
- Che 197 SWX Lecture 14 - Mmda - MSWDokumen19 halamanChe 197 SWX Lecture 14 - Mmda - MSWJelor GallegoBelum ada peringkat
- Anies. Cleofe. Estacio. NogalesDokumen44 halamanAnies. Cleofe. Estacio. NogalesJelor GallegoBelum ada peringkat
- 08 Double Integrals in Polar Coordinates - HandoutDokumen12 halaman08 Double Integrals in Polar Coordinates - HandoutJelor GallegoBelum ada peringkat
- Mickley MethodDokumen1 halamanMickley MethodJelor GallegoBelum ada peringkat
- Compliance Monitoring ReportDokumen6 halamanCompliance Monitoring ReportJelor Gallego100% (1)