Acetone Kinetics
Diunggah oleh
Eddie ChuiDeskripsi Asli:
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Acetone Kinetics
Diunggah oleh
Eddie ChuiHak Cipta:
Format Tersedia
Chemistry 1B
The Kinetics of the Iodination of Acetone
This experiment involves two parts: the first is to determine the reaction orders for acetone and hydrogen ion using the method of initial rates. The second involves using the Arrhenius equation to find the activation energy and frequency factor for the reaction. You will be assigned either a reaction mixture from Part 1 or a specific temperature from Part 2 to investigate. Class data will be used for complete analysis.
Part 1 Determination of Reaction Orders
Theory of Reaction Rates
The rate of a chemical reaction depends upon many factors, such as concentration of reactants, temperature, and the presence of a catalyst. The rate of a reaction is usually dependent on the concentration of reactants. The rate decreases as concentration decreases. We can write a rate law equation to show this relationship. The rate law equation relates the rate of a reaction to the concentration of each reactant. Consider the general reaction below: a A + b B c C and d D + The small letters represent the stoichiometric coefficients. We can express the general rate of reaction in terms of the disappearance of the reactants or appearance of the products. rate of reaction = rate = ! (1)
"[A] "[B] ![C] ![D] or or ! or "t "t !t !t
(2)
Here [A] is the change in the molar concentration of reactant A over time, t. The reaction rate will have units of M/time. By convention, the minus sign for reactants ensures a positive value for the rate since a change in concentration of a reactant with time, ([A]/t), is a negative value. An empirical formula (rate law equation or rate equation) has been found from experiment that relates rate to the concentration of each reactant at any time in the reaction. The rate law equation has the form rate = k[A] m[B]n (3)
In the rate law equation, [A] and [B] are the molarities of each reactant in the balanced equation at some time t. Here k is the rate constant (depends only on temperature), and m and n are the orders of the reaction with respect to A and B. Important: m and n are not related to the stoichiometric coefficients in the balanced equation! Reaction orders must be determined by experiment! Reaction orders are usually integers of value 0, 1, or 2. The rate constant, k, is also determined by experiment.
Determining Reaction Orders
One method of determining the reaction orders (and rate constant) for a chemical reaction is through the method of initial rates. In this method an experiment is performed where the rate of reaction is measured at the beginning of the reaction under a known set of initial concentrations for each reactant. A second experiment is then performed where the concentration of only one reactant is changed (usually doubled or halved) and the rate of reaction is again measured. By comparing how the rate of reaction changes as the concentration of one reactant is changed the value of the reaction order can be determined for that reactant. For example consider the following simple rate law equation: rate = k[A]n (4)
The value of n will determine how the reaction rate changes as the concentration of A changes. If n = 0, a zero order reaction, then changing the concentration of A will have no effect on the rate since [A]n will always equal 1, therefore rate = k at all times. As we change the initial concentration of A in an experiment we will see no change in the reaction rate for a zero order reactant. Now, if n = 1, a first order reaction, then rate = k[A]. If [A] is doubled in an experiment we should see the reaction rate double as well. What will happen to the reaction rate if n = 2, a second order reaction, if [A] is doubled? You should convince yourself that the reaction rate increases by a factor of 22 = 4.
Acetone Kinetics.doc
February 28, 2011
Chemistry 1B
Determining the Rate Constant
Once the reaction orders have been determined the rate constant can be calculated from the measured rate and the initial reactant concentrations. Consider an experiment where the rate depends on reactants A and B with reaction orders of n and m as in equation 3. For a set of initial concentrations, [A]0, and [B]0, the initial reaction rate is measured. Rearranging equation 3 we have
k=
rate [A]n[ B]m 0 0
(5)
Where [A]0 and [B]0 are the initial concentrations of A and B and n and m are found from previous experiments.
Application of the Theory: The Iodination of Acetone
In this lab we will study the iodination of acetone. This is a simple reaction a single hydrogen on acetone is replaced by an iodine atom: CH3COCH3(aq)+ I2(aq) + H +(aq) > CH3COCH2I(aq)+ I(aq) + 2 H+(aq) (6) The reaction requires the presence of hydrogen ions (acidic conditions) to take place. The hydrogen ions act as a catalyst and influence the reaction rate. Without the hydrogen ions the reaction rate is exceedingly slow. The rate law for this reaction is rate = k[Ac]m[H+]n[I2]p where Ac = acetone From previous studies it has been determined that the rate of reaction is zero order with respect to iodine, p = 0. Thus, equation 7 can be rewritten as rate = k[Ac]m[H+]n (8) (7)
We will use the method of initial rates to determine the reaction order for both acetone and the hydrogen ion. Once they are known we will be able to find the value of the rate constant, k.
The Method of Initial Rates - Revisited
Figure 1. Reaction Rate Comparison Unfortunately, determining the initial rate of reaction can be 0.0110 very difficult experimentally since the reaction rate decreases 0.0100 immediately after the reaction begins. We would like to 0.00900 measure the reaction rate at t = 0, the instant the reaction begins since we know the reactant concentrations at that time. Any 0.00800 Constant Reaction Rate = k' time after t = 0 the reaction rate has changed since the reactant 0.00700 Reaction Rate Decreses with Time concentrations are decreasing. Thus, measuring the initial 0.00600 reaction rate can be experimentally challenging. How do we 0.00500 overcome this experimental limitation? Well for the iodination 0.00400 of acetone we are fortunate to have a system where one reactant has a reaction order of zero, the iodine. The rate of 0.00300 reaction at any time does not depend upon the concentration of 0.00200 iodine (recall equation 8) as long as some iodine is present. 0.00100 With this in mind we can experimentally set up initial 0.00 concentrations for acetone, hydrogen ions, and iodine where 0.00 40.0 80.0 120 160 200 240 280 320 Time (s) the rate of reaction becomes independent of changes in the acetone and hydrogen ion concentration over time. That is, the rate of reaction will not change as the reaction proceeds towards equilibrium. The reaction rate is constant over time. Since the reaction rate is not changing over time we can measure the rate any time that is experimentally convenient and assign this value to the initial rate of reaction. Usually this is over the course of the entire reaction as the limiting reactant concentration goes to zero.
Key Experimental Design Concept Pay Attention!
To make the reaction rate independent of time for hydrogen and acetone we set the iodine as the limiting reactant and provide a large excess of acetone and hydrogen ions. Recall from equation 8 that the reaction rate will change as the concentration of acetone and hydrogen ion change. If the initial concentrations of acetone and hydrogen ion are much greater than
Acetone Kinetics.doc
Reaction Rate (M/s)
February 28, 2011
Chemistry 1B
iodine, ([Ac]0 and [H+]0 >> [I2]0), then as the reaction proceeds the concentrations of acetone and hydrogen ion will not change appreciably during the reaction. From equation 8 we then have the following rate = k[Ac]0m[H+]0n = k (9)
The constant k is called a pseudo (false) rate constant since it is the product of the true rate constant k and the initial concentrations of reactants that do not change over time, acetone to the m power, [Ac]0m, and hydrogen ion to the n power, [H+]0n. This does not imply the reaction rate no longer depends upon acetone and hydrogen ion concentration: what we have done is removed the time dependence of the reaction rate with respect to acetone and hydrogen ion. See figure 1. We are now free to measure the reaction rate over any time period we chose. From equation 9 we see that the rate should change (but remain constant over time) if the initial concentrations of acetone and/or hydrogen ion are changed since the rate depends upon the initial concentrations of hydrogen and acetone. We will use the method of initial reaction rates to determine the reaction orders of acetone and hydrogen ion. It is expected that you understand this concept of constant reaction rate.
Part 1 Experimental Procedure Reaction Orders
To find the reaction orders for acetone and hydrogen ion we will measure the rate of reaction using three different sets of initial concentrations. Table 1 gives the volumes of each reactant. As you can see in mixture 2 we have doubled the volume of acetone compared to mixture 1 and in mixture 3 we have doubled the volume of hydrogen ion (compared to 1). Table 1. Initial Reactant Volumes COPY TO YOUR NOTEBOOK! 4.00 M Acetone (mL) 1.00 M Hydrochloric acid (mL) 5.0x10-3 M Iodine (mL) Deionized Water (mL) 1.0 1.0 1.0 2.0 2.0 1.0 1.0 1.0 1.0 2.0 1.0 1.0
Mixture 1 2 3
To measure the rate of reaction we take advantage of the fact that iodine, I2, is yellow in solution while acetone and hydrogen ion are colorless. The products of the iodination reaction (refer to equation 7) are also colorless. As the reaction proceeds the mixture will change from yellow to colorless. To measure the reaction rate we time how long it takes for the solution to become colorless after all the reactants have been mixed. This time corresponds to the complete reaction (100%) of the limiting reactant iodine. From equation 2 the reaction rate can now be written as rate = !
[I 2 ] final - [I 2 ]inital "[I 2 ] = "t "t
= -
0 - [I 2 ] 0 [I 2 ]0 = "t "t
(10)
Thus, we can measure the reaction rate by measuring the time required to consume all the iodine initially present in each reaction mixture.
Detailed Procedure You should be assigned one mixture from Table 1 by your instructor. Repeat the procedure below at least three times for your mixture. 1. Wear safety glasses 2. Prepare a water bath as close to 25 C as possible using a half-full a 600-mL beaker. Obtain two large test tubes. 3. Clean and dry the test tubes. Label them A, and B. 4. In test tube A pipette the required volumes of acetone and hydrochloric acid for your assigned mixture. Use Table 1 as a guide. In test tube B pipette the required volumes of iodine and water. Place the test tubes in the 25 C water bath and allow to thermally equilibrate (about 5 minutes). 5. Remove test tubes A and B from the water bath. While watching the clock (or using a timer) pour the iodine (B) into the acetone/HCl mixture (A). Record the time if mixing or start a timer. To insure all the iodine is transferred, pour the entire mixture back-and-forth between the two test tubes several times. Place the test tube with the reaction mixture back in the water bath. 6. Insert a clean and dry stirring rod into the reaction mixture and stir gently while you observe the yellow color fade. Watching carefully record the time when the mixture becomes colorless. 7. Repeat steps 3 through 6 as time permits for your assigned reaction mixture.
Acetone Kinetics.doc
February 28, 2011
Chemistry 1B
Part 2: Determination of the Activation Energy
Theory of Reaction Energetics
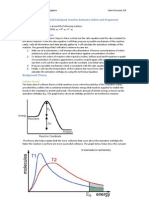
Figure 2. Reaction Coordinate Diagram for a Simple Reaction Activation Energy Molecules require some minimum energy to react. This energy barrier to initiate a reaction is called the activation energy, Ea. Figure 2 shows the activation energy profile for the reaction of NO2 and CO (to give NO and CO2). If the activation energy is small, then the kinetic energy needed to overcome the barrier is low. At room temperature a high proportion of the molecules will have sufficient energy to react and the reaction rate is fast. Conversely, if the activation energy is large then at room temperature few molecules will have sufficient kinetic energy to overcome the barrier and the reaction rate is slow. The maximum in the reaction coordinate diagram (energy curve) is called the transition state. Enough energy has been added to initiate the bond breaking process while the bond forming process has also begun. As the new bonds form energy is released until the product state is formed. The structure of the system at the transition state is of interest to chemists, however analysis of this short-lived state is difficult experimentally. Figure 3. Boltzman Distribution of Kinetic Energies Temperature Effects on Reaction Rates Temperature has a great effect on reaction rates. As temperature increases the fraction of collisions with enough kinetic energy to overcome the activation energy increases. As shown in figure 3 as temperature is raised more molecules have higher kinetic energies. Thus more collisions have the minimum energy to overcome the activation energy, hence a greater reaction rate. As a rule-of-thumb, for every 10 C increase in temperature the reaction rate will double. Effect of Molecular Orientation on Reaction Rates Not only must molecules have a minimum kinetic energy to react they also must come together in the correct orientation. The orientation during the collision of the molecules will determine if the correct bonds will begin to break. If the orientation of the molecules is not correct, the molecules will simple bounce off each other and no reaction will take place. A shown in figure 2 one of the O atoms of NO2 must collide with the carbon atom of CO. This orientation dependence is called the steric factor for the reaction.
The Arrhenius Equation The activation energy, steric factor and temperature dependence of the reaction rate are summarized by the Arrhenius equation:
k = Ae !E a RT
(11)
Where k is the rate constant for the reaction, A is the frequency factor, Ea is the activation energy, and T is the temperature. The frequency factor, A, is related to the number of collisions with the correct orientation or steric-factor to react. R is the molar mass gas constant, 8.3145 J/(mol-K). The frequency factor has the same units of the rate constant and can be
Acetone Kinetics.doc 4 February 28, 2011
Chemistry 1B
interpreted as the number of collisions per unit time per unit molarity of reactants. This is typically a large number, 108 or !E RT greater. The quantity e a is interpreted as the fraction of collisions having at least the minimum energy for reaction. This value is always significantly less than one, 1012 or even smaller. The Arrhenius equation is valuable since we can use it to determine A, and Ea for a reaction by measuring the rate constant as a function of temperature. Using the properties of logarithms we have
E !1$ ln(k) = a # & + ln(A ) R "T %
This is an equation for a straight line.
(12)
Part 2 Experimental Procedure Activation Energy
For the temperature dependence study all students will use mixture 2 from Table 1. Suggested temperatures are: 30, 20, 15 and 10 C. Your instructor will assign you a single temperature. Note: the 15 and 10 C reaction rates are slow. More than one group should be assigned these temperatures. Repeat the procedure below at least three times (or as time allows) for your temperature. 1. Wear Safety Glasses 2. Prepare a water bath as close to your assigned temperature as possible using a half-full a 600-mL beaker. Obtain two large test tubes, 3. Clean and dry the test tubes. Label them A, and B. 4. In test tube A pipette the required volumes of acetone and hydrochloric acid for mixture 2. Use Table 1 as a guide. In test tube B pipette the required volumes of iodine and water. Place the test tubes in the water bath and allow to thermally equilibrate (about 5 minutes). 5. Remove test tubes A and B from the water bath. While watching the clock (or using a timer) pour the iodine (B) into the acetone/HCl mixture (A). Record the time of mixing or start a timer. To insure all the iodine is transferred, pour the entire mixture back-and-forth between the two test tubes several times. Place the test tube with the reaction mixture back in the water bath! 6. Insert a clean and dry stirring rod into the reaction mixture and stir gently while you observe the yellow color fade. Watching carefully record the time when the mixture becomes colorless. 7. Repeat steps 3 through 6 as time permits for your assigned temperature.
Processing the Data Follow these general guidelines.
Part 1: Determination of Reaction Orders 1. 2. 3. The rate of reaction is calculated using equation 10 and the average reaction time and initial concentration of I2. By comparing the rates of reaction between the three mixtures you should be able to determine the reaction orders for acetone and hydrogen ion. Use the method of initial rates. Show your work on the report sheet. Using equation 5 calculate the rate constant for each mixture from the rate of reaction. What are the units of k? Enter the results of the calculation in the table. Is the rate constant the same between the three trials?
Part 2: The Temperature Dependence of the Rate Constant - Use Graphical Analysis for the following calculations. 1. From the class data for Part 2 calculate a rate of reaction for each temperature-time data pair. Remember all the data is from reaction mixture 2. You should have as many data pairs covering the temperature range from 30 C down to about 10C as possible. DO NOT AVERAGE TIMES at the same temperature. Dont forget to include the data for mixture 2 taken in Part 1. Calculate the rate of reaction for each time. Using the orders determined from Part 1 calculate the rate constant as a function of temperature. Make a graph of rate constant (y-axis) as a function of Kelvin temperature.(x-axis). Use T for the short name for the x-axis in Graphical Analysis. Finally, fit the rate constant (y-axis) to the nonlinear function A*exp(E/(8.3145*T)) where A and E are the fitting constants, T is the x-axis short name, and 8.3145 is the value of the gas constant R with units of J/molK. From the fit A is the frequency factor and E is the activation energy.
5 February 28, 2011
2. 3. 4.
Acetone Kinetics.doc
Name Locker # Partner
Report Sheet: Acetone Kinetics
Part 1
Enter the following from the class experimental results and analysis. Include any units missing in the column headings! Mixture 1 [Acetone]0 (M) [H+]0 (M) [I2]0 (M) Avg. time Reaction Rate Rate constant, k Avg. temp
Average k
Reaction orders from your analysis of Part 1 Data
Show your work for Acetone and H+
Acetone
H+
I2
Questions on Part 1
1. If the concentration of iodine is doubled in mixture 1, with the acetone and hydrogen ion remaining the same: a) Determine the reaction rate.
b) How long would it take of the mixture to become colorless?
Acetone Kinetics.doc
February 28, 2011
Name Locker # Partner
Part 2: Include a copy of your Arrhenius Graph, k versus T.
1. From your Arrhenius graph, report your activation energy and frequency factor values to the proper significant figures and with correct units. activation energy 2. frequency factor
Using your activation energy and frequency factor values, a) calculate the rate constant, k, at 0 C.
b) calculate the temperature (in C) the system would have to be taken to increase the rate constant by 10 times the value found at 0 C.
c)
calculate the fraction of collisions that have enough energy to overcome the activation energy barrier at 25 C.
3.
Two reactions have identical values for Ea. Does this ensure that they will have the same rate constant if run at the same temperature? Explain in terms of the Arrhenius equation.
Acetone Kinetics.doc
February 28, 2011
Name Locker # Partner
4.
Based on the provided reaction profile diagram, a) how many intermediates are formed in the reaction A>D?
b) how many transition states are there?
c)
which step (A > B, B > C, C > D) is the fastest?
d) Draw a line on the graph showing the magnitude of the overall activation energy for A > D.
e)
Is the reaction A> D exothermic or endothermic?
5.
The reaction between ethyl iodide and hydroxide ion in ethanol (C2H5OH) solution C2H5I(alc) + OH(alc) > C2H5OH(l) + I(alc) has an activation energy of 86.8 kJ/mol and a frequency factor of 2.10 1011 M1 s1. a) Calculate the rate constant for the reaction at 35C.
b) Equal volumes of a 0.0239 M KOH in ethanol solution and a 0.0373 M C2H5I in ethanol solution are mixed. Assuming the reaction is first order in each reactant, what is the initial rate of reaction at 35C? Hint: Do your dilutions first!
Acetone Kinetics.doc
February 28, 2011
Anda mungkin juga menyukai
- Activation Energy of Iodination of AcetoneDokumen26 halamanActivation Energy of Iodination of AcetonejoebidenyyzBelum ada peringkat
- Plano Electrico PC130-8Dokumen9 halamanPlano Electrico PC130-8jon fredyBelum ada peringkat
- Airflow Measuring With Piezometer RingDokumen4 halamanAirflow Measuring With Piezometer RingcassindromeBelum ada peringkat
- Investigation - Ideas - Files - The Kinetics of The Iodination of Acetone PDFDokumen9 halamanInvestigation - Ideas - Files - The Kinetics of The Iodination of Acetone PDFkgothatso maleteBelum ada peringkat
- Experiment FourDokumen10 halamanExperiment FourYana SyafriyanaBelum ada peringkat
- Iodination of Acetone KineticsDokumen4 halamanIodination of Acetone Kineticsinstrutech0% (2)
- Liquid Phase ReactorDokumen22 halamanLiquid Phase Reactorkrishy19s100% (2)
- Lab3 KineticsiiDokumen9 halamanLab3 KineticsiiDHANABOINA SAIRAMBelum ada peringkat
- 162 Lab IodineClockReaction 20140227GF1Dokumen11 halaman162 Lab IodineClockReaction 20140227GF1Rza AbdullayevBelum ada peringkat
- The Kinetic Study of The IodinationDokumen6 halamanThe Kinetic Study of The IodinationsamBelum ada peringkat
- Physical CHemistry:Iodinization of AcetoneDokumen4 halamanPhysical CHemistry:Iodinization of AcetoneLevy Medina TrayaBelum ada peringkat
- Lab 5 - Iodine - Clock ReactionDokumen13 halamanLab 5 - Iodine - Clock Reactionsunho leeBelum ada peringkat
- Rate of Iodation of AcetoneDokumen9 halamanRate of Iodation of AcetoneAbdur Rakib M Sarwar0% (1)
- Experiment: KinieticsDokumen9 halamanExperiment: KinieticsJoey PabalinasBelum ada peringkat
- 114 Exp 2 F13Dokumen14 halaman114 Exp 2 F13Amy WuBelum ada peringkat
- Rate of ReactionDokumen13 halamanRate of ReactionFrank LaporteBelum ada peringkat
- 1.1 How FastDokumen14 halaman1.1 How FastG M Ali KawsarBelum ada peringkat
- Reaction Rates Chemistry)Dokumen14 halamanReaction Rates Chemistry)Nasya TehBelum ada peringkat
- Kinetika KimiaDokumen29 halamanKinetika KimiaOkta DefaBelum ada peringkat
- Rate and KineticsDokumen30 halamanRate and KineticsAbdulAhadBelum ada peringkat
- Chemical KineticsDokumen3 halamanChemical KineticsYEEHSHIN JILL GAYOBelum ada peringkat
- Reaction Rate: A+B ABDokumen5 halamanReaction Rate: A+B ABFaisal Mohad Al SakhenBelum ada peringkat
- I. Title of Experiment: II. Date of Experiment: Iii. The End of Experiment: IV. Purpose of Experiment: V. Basic Theories A. Reaction RateDokumen14 halamanI. Title of Experiment: II. Date of Experiment: Iii. The End of Experiment: IV. Purpose of Experiment: V. Basic Theories A. Reaction RateputriBelum ada peringkat
- Lab ReportpdfDokumen7 halamanLab ReportpdfStefano FochesattoBelum ada peringkat
- Pengaruh Suhu Terhadap Laju ReaksiDokumen22 halamanPengaruh Suhu Terhadap Laju Reaksiayrajiu alexanderBelum ada peringkat
- Determining Reaction Order Initial Rates and The Method of IsolationDokumen4 halamanDetermining Reaction Order Initial Rates and The Method of IsolationDeepak PandeyBelum ada peringkat
- CHM 112 Kinetics Practice Problems Answers - Reader ViewDokumen19 halamanCHM 112 Kinetics Practice Problems Answers - Reader ViewSyasya FaqihahBelum ada peringkat
- 08 Kinetics ExperimentsDokumen13 halaman08 Kinetics ExperimentsEduardo AndresBelum ada peringkat
- Experiment 4: Kinetics of An Iodine Clock ReactionDokumen16 halamanExperiment 4: Kinetics of An Iodine Clock ReactionAgustian ChuagestuBelum ada peringkat
- Boon PinDokumen13 halamanBoon PinjayaprinaBelum ada peringkat
- A2 Chemistry Unit 4 NotesDokumen27 halamanA2 Chemistry Unit 4 NotesRebecca78% (9)
- Chemical KineticsDokumen4 halamanChemical KineticsLorenzo TorresBelum ada peringkat
- Chemical Kinetics PDFDokumen4 halamanChemical Kinetics PDFAcademicBMBelum ada peringkat
- 401 Expt8Dokumen7 halaman401 Expt8Agustian ChuagestuBelum ada peringkat
- Kinetics of The Acid Catalysed Reaction Between Iodine and Propanone Final LolDokumen23 halamanKinetics of The Acid Catalysed Reaction Between Iodine and Propanone Final LolYann Perusset90% (10)
- Chemical KineticsDokumen62 halamanChemical KineticsIbnu Abbas Al BasthomiBelum ada peringkat
- Sample Exercise 14.1: SolutionDokumen32 halamanSample Exercise 14.1: SolutionCik ApaiBelum ada peringkat
- AP Lab Manual 18 - Kinetics of A ReactionDokumen13 halamanAP Lab Manual 18 - Kinetics of A ReactionRodrigo GarciaBelum ada peringkat
- Questions/ ExercisesDokumen11 halamanQuestions/ ExercisesjahmanBelum ada peringkat
- Reactor Design CH 8Dokumen8 halamanReactor Design CH 8Sami WhiteBelum ada peringkat
- Experiment 5: Chemical Kinetics - The Iodine Clock ReactionDokumen10 halamanExperiment 5: Chemical Kinetics - The Iodine Clock ReactionLero LeroBelum ada peringkat
- Reaction KineticsDokumen7 halamanReaction Kineticsjathan160% (1)
- Chemistry Unit 5.4Dokumen8 halamanChemistry Unit 5.4Sonal PereraBelum ada peringkat
- Experiment 2: Determination of Rate LawsDokumen11 halamanExperiment 2: Determination of Rate LawsgrcmllssBelum ada peringkat
- Chemical KineticsDokumen4 halamanChemical KineticsSheila Camille OrdoñaBelum ada peringkat
- Topic 4.1 Kinetics Rate Equations Determining Orders of Reaction Explaining Orders of Reaction Effect of Changing Conditions On The Rate ConstantDokumen10 halamanTopic 4.1 Kinetics Rate Equations Determining Orders of Reaction Explaining Orders of Reaction Effect of Changing Conditions On The Rate ConstantNur Kintan ApriliaBelum ada peringkat
- Chemical EquilibriaDokumen5 halamanChemical Equilibriaroku shinBelum ada peringkat
- Topic 4.1 Kinetics Rate Equations Determining Orders of Reaction Explaining Orders of Reaction Effect of Changing Conditions On The Rate ConstantDokumen9 halamanTopic 4.1 Kinetics Rate Equations Determining Orders of Reaction Explaining Orders of Reaction Effect of Changing Conditions On The Rate ConstantxcomBelum ada peringkat
- Unit Chemical Kinetics: StructureDokumen48 halamanUnit Chemical Kinetics: Structureradha911Belum ada peringkat
- Chem IA - Luca MehlDokumen18 halamanChem IA - Luca MehlYukiyo TouwBelum ada peringkat
- Rates of Reaction A Level ChemistryDokumen9 halamanRates of Reaction A Level ChemistrytabithaBelum ada peringkat
- Reaction KineticsDokumen37 halamanReaction KineticsDaisyBelum ada peringkat
- Experiment 6: Chemical KineticsDokumen28 halamanExperiment 6: Chemical KineticsBalqees HasanBelum ada peringkat
- Module in 2: General ChemistryDokumen5 halamanModule in 2: General Chemistryriza amoresBelum ada peringkat
- For Student - General Chemistry I - Module 5 - Phan Tai HuanDokumen36 halamanFor Student - General Chemistry I - Module 5 - Phan Tai HuanEsat GoceriBelum ada peringkat
- Thermometric Titrimetry: International Series of Monographs in Analytical ChemistryDari EverandThermometric Titrimetry: International Series of Monographs in Analytical ChemistryBelum ada peringkat
- Ion Association in Proton Transfer Reactions: Use of ESR for the Quantitative Determination of Gas Phase Atom and Radical ConcentrationsDari EverandIon Association in Proton Transfer Reactions: Use of ESR for the Quantitative Determination of Gas Phase Atom and Radical ConcentrationsBelum ada peringkat
- Selected Constants: Oxidation–Reduction Potentials of Inorganic Substances in Aqueous SolutionDari EverandSelected Constants: Oxidation–Reduction Potentials of Inorganic Substances in Aqueous SolutionBelum ada peringkat
- Physico-Chemistry of Solid-Gas Interfaces: Concepts and Methodology for Gas Sensor DevelopmentDari EverandPhysico-Chemistry of Solid-Gas Interfaces: Concepts and Methodology for Gas Sensor DevelopmentBelum ada peringkat
- High Performance Computing in Science and Engineering 16Dokumen665 halamanHigh Performance Computing in Science and Engineering 16Kamchatka TacalaskaBelum ada peringkat
- MP4004 Advanced Manufacturing and Nanotechnology: ContentDokumen26 halamanMP4004 Advanced Manufacturing and Nanotechnology: ContentVincent KohBelum ada peringkat
- Steel Design 1Dokumen26 halamanSteel Design 1Gracielle NebresBelum ada peringkat
- CB307 - 11 Sep - CondensersDokumen18 halamanCB307 - 11 Sep - CondensersKiran KumarBelum ada peringkat
- Itp MelchoritaDokumen7 halamanItp MelchoritadietersimaBelum ada peringkat
- SAP NonwovenDokumen6 halamanSAP NonwovenPARAN THAMANBelum ada peringkat
- Fracture Toughness and Carbide Precipitation Behavior of A737 GraDokumen98 halamanFracture Toughness and Carbide Precipitation Behavior of A737 GraMatias PachuBelum ada peringkat
- Analysis of Yield Stress Ratio and OverconsolidatiDokumen10 halamanAnalysis of Yield Stress Ratio and OverconsolidatiZakwan GusnadiBelum ada peringkat
- CTVPRC 007 enDokumen56 halamanCTVPRC 007 enrommel duranBelum ada peringkat
- 53 TOP Structural Analysis - Civil Engineering Multiple Choice Questions and Answers PDF - MCQs Preparation For Engineering Competitive ExamsDokumen7 halaman53 TOP Structural Analysis - Civil Engineering Multiple Choice Questions and Answers PDF - MCQs Preparation For Engineering Competitive Examseid ibrahimBelum ada peringkat
- A Pipe Is Gradually Tapering in SizeDokumen2 halamanA Pipe Is Gradually Tapering in SizeRez AdolfoBelum ada peringkat
- PhysicsDokumen12 halamanPhysicsDhaval MistryBelum ada peringkat
- Chapter 6 - Phase 1-Clouds & Precipitation Lecture NotesDokumen7 halamanChapter 6 - Phase 1-Clouds & Precipitation Lecture NotesGf MilesBelum ada peringkat
- 16Dokumen2 halaman16Muhammad Tayyab MadniBelum ada peringkat
- Well StimulationDokumen58 halamanWell StimulationRizwan FaridBelum ada peringkat
- Technical Data: 65x50 FS2HA 5 7.5Dokumen5 halamanTechnical Data: 65x50 FS2HA 5 7.5DavidThânBelum ada peringkat
- IntroductionDokumen24 halamanIntroductionHabibi1451Belum ada peringkat
- Project'S Ideas I. Idea Title: Heron'S Fountain Ii. Reasons To Choose An IdeasDokumen2 halamanProject'S Ideas I. Idea Title: Heron'S Fountain Ii. Reasons To Choose An IdeasTiomeldaBelum ada peringkat
- Evaporator and Its TypesDokumen22 halamanEvaporator and Its TypesIrfan ShafiqBelum ada peringkat
- C4 Inlet Bellmouth Casing (Top & Bottom Half) Coating ThicknessDokumen19 halamanC4 Inlet Bellmouth Casing (Top & Bottom Half) Coating ThicknessDave CheungBelum ada peringkat
- Moon Body Resonance: Mensur OmerbashichDokumen13 halamanMoon Body Resonance: Mensur OmerbashichgeomerBelum ada peringkat
- IS 4991 (1968) - Criteria For Blast Resistant Design of Structures For Explosions Above Ground-TableDokumen1 halamanIS 4991 (1968) - Criteria For Blast Resistant Design of Structures For Explosions Above Ground-TableRenieBelum ada peringkat
- Electronic Distance Measurement (EDM)Dokumen29 halamanElectronic Distance Measurement (EDM)Kavi MaranBelum ada peringkat
- Ex Barometric LevelingDokumen4 halamanEx Barometric LevelingStephen Ross RoledaBelum ada peringkat
- Renewable Energy: S.S. Chandel, M. Nagaraju Naik, Vikrant Sharma, Rahul ChandelDokumen10 halamanRenewable Energy: S.S. Chandel, M. Nagaraju Naik, Vikrant Sharma, Rahul ChandelCleiber NichidaBelum ada peringkat
- Blue Moon Approach To Rare Events: Molecular Simulation September 2004Dokumen9 halamanBlue Moon Approach To Rare Events: Molecular Simulation September 2004Della KhoirunnisaBelum ada peringkat
- Fundamentals of Temperature MeasurementDokumen18 halamanFundamentals of Temperature MeasurementHeather KramerBelum ada peringkat
- Materials For Engineering 4 PDFDokumen2 halamanMaterials For Engineering 4 PDFHimanshu MishraBelum ada peringkat