Reactions of Alcohols
Diunggah oleh
Neen NaazHak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Reactions of Alcohols
Diunggah oleh
Neen NaazHak Cipta:
Format Tersedia
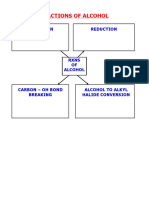
Reactions of alcohols
With dehydrating agents
Alcohols reacts with concentrated sulphuric acid, heat under reflux at 170oC and eliminate a water molecule. Concentrated H3PO4 at 70oC and anhydrous Al2O3 at 600K can also be used as dehydrating agents. Alcohols can be converted to alkenes by a single step. This is elimination.
Reactions with halogenating agents
Reaction with PCL5(s)
C2H5OH(l) + PCl5(s) room temp dry condition C2H5Cl(g) + POCl3(l) + HCl(g)
1-chloroethane fumes
Reaction with PX3 (l) (X=Cl. Br, I)
Alcohols react with PCL3 or PBr3 or PI3 and form haloalkane. PCL (l)/rtp
3
PBr3 (l)/heat under reflux
PI3 (l)/heat under reflux
Moist red phosporus and liquid bromine
+ moist red P/Br2(l)
CH3CH2Br
CH3CHOHCH3
+ moist red P/Br2(l)
CH3CHBrCH3
Alcohols react with moist red phosphorus and liquid bromine, heating under reflux and produce bromoalkanes. In the reaction, moist red phosphorus react with Br2 (l) and form phosphorus tri bromide (PBr3), which reacts with alcohol. 3Br2 +2P (s) 2PBr3(l) PBr3 (l) + 3CH3CH2OH 3CH3CH2Br +H3PO3
Reaction with alkali iodine
Primary and secondary alcohols which contain CH3-C(OH)H- react with alkali iodine under warming and results yellow ppt of triiodomethane (CHI3). Aldehydes and ketone with CH3-CO- group can also give positive results for this reactions. Alkali iodine acts as an oxidizing agent. Tertiary alcohols which contain CH3-C(OH)group cannot be oxidized by alkali iodine
Reagent: aq iodine and aq sodium hydroixde Condition: warm Type of reaction: oxidation Primary alcohol CH3-CH2-OH + 4I2(aq) + 6NaOH(aq) CHI3 (S) + HCOO-Na+ + 5NaI +5H2O Ethanol warm yellow ppt secondary alcohol CH3-CH(OH)-CH2CH3 + 4I2(aq) + 6NaOH(aq) Butan-2-ol
CHI3 (S) + HCOO-Na+ + 5NaI +5H2O
Tertiary alcohol (CH3)COH + I2(aq) + NaOH(aq) no reaction 2-methylpropan-2-ol warm Though CH3-C(OH) H group is present in some tertiary alcohols they cannot be oxidized by using I2 in NaOH or H2SO4 or dil H2SO4 & KMNO4. this is because C-H bond is absent on the carbinol carbon of tertiary alcohol. In tertairy alcohols carbinol carbon has three C-C bonds, which are stronger than C-H bond in primary and secondary alcohols. The oxidizing agent mentioned above are not strong enough to oxidize the C-C bond, but they can oxidize C-H bond easily in primary and secondary alcohols.
Esterification
Catalyst: concentrated H2SO4 (dehydrating agent - it removes water causing the equilibrium to move to the right and increases the yield Conditions: reflux
Uses of esters
Esters are fairly unreactive but that doesnt make them useless Used as flavourings
Naming esters
Named from the alcohol and carboxylic acid which made them... CH3OH + CH3COOH
from ethanoic acid
CH3COOCH3 + H2O
from methanol
CH3COOCH3
METHYL ETHANOATE
Anda mungkin juga menyukai
- Practice Makes Perfect in Chemistry: Acids, Bases, and SaltsDari EverandPractice Makes Perfect in Chemistry: Acids, Bases, and SaltsBelum ada peringkat
- TEMA Alcoholes 2 PARTEDokumen34 halamanTEMA Alcoholes 2 PARTEMaria Jose AramayoBelum ada peringkat
- 2080 New Course XII HaloalkaneDokumen57 halaman2080 New Course XII HaloalkaneSangam PaudelBelum ada peringkat
- Advanced Pharmaceutical analysisDari EverandAdvanced Pharmaceutical analysisPenilaian: 4.5 dari 5 bintang4.5/5 (2)
- Alcohols ChemistryDokumen24 halamanAlcohols ChemistryBritney PattersonBelum ada peringkat
- Organic ReactionsDokumen4 halamanOrganic ReactionsRobbing_HoodBelum ada peringkat
- Chem Factsheet 17Dokumen3 halamanChem Factsheet 17drdre12100% (1)
- ALCOHOLSDokumen10 halamanALCOHOLSsaraBelum ada peringkat
- AlcoholsDokumen91 halamanAlcoholsWAN NUR AISYAH WAN AZIZANBelum ada peringkat
- CHEM 215 F12 Chapter 13 Notes UMICHDokumen13 halamanCHEM 215 F12 Chapter 13 Notes UMICHRoxanne IlaganBelum ada peringkat
- Alcohols, Phenols and Ethers NotesDokumen55 halamanAlcohols, Phenols and Ethers Notessamay gujratiBelum ada peringkat
- Alcohol, Phenols, Thiols, and EthersDokumen17 halamanAlcohol, Phenols, Thiols, and EthersJohn Paul CuBelum ada peringkat
- Xii CH-7 Synopsis Chemistry CCWSDokumen10 halamanXii CH-7 Synopsis Chemistry CCWSTanmay RawatBelum ada peringkat
- Alkyl HalidesDokumen15 halamanAlkyl Halidesutkarsh jain100% (4)
- Live Sahab20 - Aldehydes, Ketones & Carboxylic AcidsDokumen74 halamanLive Sahab20 - Aldehydes, Ketones & Carboxylic Acidssamay gujratiBelum ada peringkat
- Alcohols 2Dokumen15 halamanAlcohols 2Junaid KhanBelum ada peringkat
- Carbonyl Compounds: Carboxylic Acids & EsterDokumen28 halamanCarbonyl Compounds: Carboxylic Acids & Esterrustam effendyBelum ada peringkat
- Kuliah - Alcohols, Ethers, and ThiolsDokumen33 halamanKuliah - Alcohols, Ethers, and ThiolsDian Eka FajriyantoBelum ada peringkat
- Reactions of Alcohol: Oxidation ReductionDokumen18 halamanReactions of Alcohol: Oxidation ReductioncikguhafidzuddinBelum ada peringkat
- Alcohols and EthersDokumen51 halamanAlcohols and EthersnanaBelum ada peringkat
- Alcohols and Ethers CHM457Dokumen51 halamanAlcohols and Ethers CHM457AIMAN IMAN SHAIFUDDINBelum ada peringkat
- Aldehydes and KetonesDokumen45 halamanAldehydes and Ketonesroku_soraBelum ada peringkat
- Hydroxy Compounds: (Alcohols)Dokumen71 halamanHydroxy Compounds: (Alcohols)NorsyazaEdmiraBelum ada peringkat
- Organic Reactions Summary SheetDokumen2 halamanOrganic Reactions Summary Sheetthacheee64% (11)
- Anic Chemistry Alkyl HalidesDokumen15 halamanAnic Chemistry Alkyl Halideseamcetmaterials100% (6)
- Alcohol, Esters and Carboxylic AcidsDokumen55 halamanAlcohol, Esters and Carboxylic AcidsHuiru ZhaoBelum ada peringkat
- Organic Chemistry - II: Alkyl Halides and ChloroformDokumen15 halamanOrganic Chemistry - II: Alkyl Halides and ChloroformAtul VermaBelum ada peringkat
- Alkyl HalidesDokumen15 halamanAlkyl HalidesDjdj DjdjBelum ada peringkat
- Chem 121 Carbonyl CompsDokumen36 halamanChem 121 Carbonyl CompsAbubakarBelum ada peringkat
- Organic Compounds Containing OxygenDokumen44 halamanOrganic Compounds Containing OxygenKeishaBelum ada peringkat
- ALCOHOLS, ETHERS AND RELATED COMPOUNDSDokumen26 halamanALCOHOLS, ETHERS AND RELATED COMPOUNDSꓰꓡꓡꓰꓠ-ꓓꓰꓖꓰꓠꓰꓣꓰꓢꓢ.Belum ada peringkat
- Alcohols: Which of The Structures Is/are Classified As Phenols?Dokumen7 halamanAlcohols: Which of The Structures Is/are Classified As Phenols?Kaviraj SinghBelum ada peringkat
- Lecture9 Alkenes2010Dokumen79 halamanLecture9 Alkenes2010Inoxcent MoonBelum ada peringkat
- LEARN ABOUT ALCOHOLSDokumen42 halamanLEARN ABOUT ALCOHOLSThe Mini KitchenBelum ada peringkat
- Hydroxy CompoundsDokumen7 halamanHydroxy CompoundsahumanbeinginearthBelum ada peringkat
- CH 10Dokumen11 halamanCH 10Vishal KushwahBelum ada peringkat
- Carbonyl Compounds Aldehydes and KetonesDokumen62 halamanCarbonyl Compounds Aldehydes and KetonesSubhabrata MabhaiBelum ada peringkat
- Chapter 6, 7 Halohydrocarbon, Alcohol, PhenolDokumen89 halamanChapter 6, 7 Halohydrocarbon, Alcohol, PhenolGan Suk Ling100% (1)
- Chapter 12 Study Guide PDFDokumen44 halamanChapter 12 Study Guide PDFkBelum ada peringkat
- Hasan Sayginel: Edexcel A Level Organic ChemistryDokumen41 halamanHasan Sayginel: Edexcel A Level Organic ChemistryDEEBANBelum ada peringkat
- Alcohol Ether EpoxideDokumen31 halamanAlcohol Ether EpoxideANISTHESIAHYUNI BINTI DURMAN FKJBelum ada peringkat
- Alcohols ClassDokumen29 halamanAlcohols ClassRyan JamesBelum ada peringkat
- ALKANOLSDokumen25 halamanALKANOLSKoki KingBelum ada peringkat
- Alcoholpresentation 111029054727 Phpapp01Dokumen27 halamanAlcoholpresentation 111029054727 Phpapp01Namra SarvaiyaBelum ada peringkat
- PPT Alcohol JadiDokumen60 halamanPPT Alcohol JadiBudi AbutBelum ada peringkat
- Chapter 1 ALCOHOLDokumen65 halamanChapter 1 ALCOHOLNURUL AINUN MUHAMMAD NOR100% (1)
- Class - 12/chemistry-2 - Reactions of AlcoholsDokumen60 halamanClass - 12/chemistry-2 - Reactions of AlcoholsAISHA AHAMMEDBelum ada peringkat
- RADHA SWAMI ACADEMY: Aldehydes, Ketones and Carboxylic AcidsDokumen10 halamanRADHA SWAMI ACADEMY: Aldehydes, Ketones and Carboxylic AcidsVishal KushwahBelum ada peringkat
- Synthesis of Drug - 2Dokumen44 halamanSynthesis of Drug - 2'Nurirjawati ElRuri KawangBelum ada peringkat
- Alcohol, Phenol & EtherDokumen23 halamanAlcohol, Phenol & EtherRahul PrajapatiBelum ada peringkat
- Carboxylic AcidDokumen37 halamanCarboxylic Acidaman2611994_1920111580% (5)
- Aldehids and KetonesDokumen47 halamanAldehids and Ketonesistri kyungsoBelum ada peringkat
- Water An An Ether: AlcoholDokumen35 halamanWater An An Ether: AlcoholEshita SharmaBelum ada peringkat
- IAL Chemistry Unit 4 Reactions SummaryDokumen12 halamanIAL Chemistry Unit 4 Reactions SummarySilmaSubahHoqueBelum ada peringkat
- Chemistry Form 6 Sem 3 10Dokumen29 halamanChemistry Form 6 Sem 3 10Anonymous WAnr0jvBelum ada peringkat
- Org Chem Alcohol Mechanisms From JasperseDokumen22 halamanOrg Chem Alcohol Mechanisms From JasperseChloe JazminesBelum ada peringkat
- Halogeno AlkanesDokumen12 halamanHalogeno AlkanessaraBelum ada peringkat
- Summary of Organic Chemistry2 - Week19Dokumen105 halamanSummary of Organic Chemistry2 - Week19Dollar'sCornerBelum ada peringkat
- Chemistry AlcoholsDokumen44 halamanChemistry AlcoholsSayan Kumar KhanBelum ada peringkat
- C 341 F 2011 Chapter 5 StereochemistryDokumen23 halamanC 341 F 2011 Chapter 5 StereochemistryNeen NaazBelum ada peringkat
- Addu High School Hithadhoo/Addu C TOPIC: ENTROPY-Enthalpy of Solution Worksheet 2Dokumen8 halamanAddu High School Hithadhoo/Addu C TOPIC: ENTROPY-Enthalpy of Solution Worksheet 2Neen NaazBelum ada peringkat
- 10 1 1 564 6139Dokumen8 halaman10 1 1 564 6139Neen NaazBelum ada peringkat
- NMR Analysis of Raspberry EsterDokumen9 halamanNMR Analysis of Raspberry EsterNeen NaazBelum ada peringkat
- Periodic QuestionsDokumen22 halamanPeriodic QuestionsDrRemyaranjith MenonBelum ada peringkat
- Unit 4.3 How Fast-RatesDokumen99 halamanUnit 4.3 How Fast-RatesNeen NaazBelum ada peringkat
- 4.4 Entropy PDFDokumen49 halaman4.4 Entropy PDFLis ViegasBelum ada peringkat
- Chemistry Revision Pack - 2012Dokumen20 halamanChemistry Revision Pack - 2012api-21735041071% (7)
- Alkali MetalsDokumen31 halamanAlkali Metalsromiifree100% (1)
- Analysis of red wine compounds by GC-MSDokumen16 halamanAnalysis of red wine compounds by GC-MSNeen NaazBelum ada peringkat
- Lecture26 3 IntroNMR Spectro!Dokumen31 halamanLecture26 3 IntroNMR Spectro!AninditaTrikusumaBelum ada peringkat
- Task 1: Group 1 - The Alkali MetalsDokumen4 halamanTask 1: Group 1 - The Alkali MetalsNeen NaazBelum ada peringkat
- Ess Chem Unit 1 Answers PDFDokumen2 halamanEss Chem Unit 1 Answers PDFSeong Hun LeeBelum ada peringkat
- Cambridge IGCSE Chemistry Practice Book Answer - Bryan Earl, Doug WilfordDokumen44 halamanCambridge IGCSE Chemistry Practice Book Answer - Bryan Earl, Doug WilfordOswald Chan17% (6)
- 11 - Topic 20 Organic Chemistry AnswersDokumen60 halaman11 - Topic 20 Organic Chemistry AnswersNeen NaazBelum ada peringkat
- Carbonyl Compounds, Carboxylic Acids, Esters & Polyesters 1 MSDokumen17 halamanCarbonyl Compounds, Carboxylic Acids, Esters & Polyesters 1 MSNeen NaazBelum ada peringkat
- WCH04 01 MSC 20160302 PDFDokumen25 halamanWCH04 01 MSC 20160302 PDFNeen NaazBelum ada peringkat
- A2as Chem Revised Support 18964Dokumen12 halamanA2as Chem Revised Support 18964Neen NaazBelum ada peringkat
- Chemistry 162 Workbook 3Dokumen51 halamanChemistry 162 Workbook 3Neen NaazBelum ada peringkat
- F324: Analysis of Painkillers, Detergents and EstersDokumen9 halamanF324: Analysis of Painkillers, Detergents and EstersNeen NaazBelum ada peringkat
- EdExcel A Level Chemistry Unit 4 Mark Scheme Results Paper 1 Jan 2006Dokumen11 halamanEdExcel A Level Chemistry Unit 4 Mark Scheme Results Paper 1 Jan 2006Neen NaazBelum ada peringkat
- Chemistry: Edexcel GCEDokumen24 halamanChemistry: Edexcel GCEaquu174015100% (1)
- C4e 3 'Group 7 (The Halogens) ' HW SheetDokumen3 halamanC4e 3 'Group 7 (The Halogens) ' HW SheetNeen NaazBelum ada peringkat
- 1.3 FormulaeDokumen64 halaman1.3 FormulaeNeen NaazBelum ada peringkat
- Introduction To The Periodic Table: Atomic Number Symbol Atomic Weight Element Compound MixtureDokumen20 halamanIntroduction To The Periodic Table: Atomic Number Symbol Atomic Weight Element Compound Mixturepreeti.2405Belum ada peringkat
- 1.3 FormulaeDokumen64 halaman1.3 FormulaeNeen NaazBelum ada peringkat
- Balancing Chemical EquationsDokumen13 halamanBalancing Chemical EquationsNeen NaazBelum ada peringkat
- Ionic Bonds: Chemistry Mrs. CoyleDokumen35 halamanIonic Bonds: Chemistry Mrs. CoyleNeen NaazBelum ada peringkat
- Basic Probability and Reliability Concepts: Roy Billinton Power System Research Group University of Saskatchewan CanadaDokumen248 halamanBasic Probability and Reliability Concepts: Roy Billinton Power System Research Group University of Saskatchewan CanadaSimon van BentenBelum ada peringkat
- 2.design and Analysis of Escalator FrameDokumen7 halaman2.design and Analysis of Escalator FrameSyamil Dzulfida100% (1)
- Typescript Cheat Sheet: by ViaDokumen2 halamanTypescript Cheat Sheet: by ViapbecicBelum ada peringkat
- Electrical Circuit Analysis ProblemsDokumen14 halamanElectrical Circuit Analysis ProblemsMd Rafiqul IslamBelum ada peringkat
- Areas Related To CircleDokumen32 halamanAreas Related To CircleGiorno GiovannaBelum ada peringkat
- Week 11 - Fundamentals of Distribution SystemsDokumen39 halamanWeek 11 - Fundamentals of Distribution SystemsEstika Vriscilla GintingBelum ada peringkat
- Introduction To Probability: Business Statistics: Communicating With Numbers, 4eDokumen52 halamanIntroduction To Probability: Business Statistics: Communicating With Numbers, 4eRetno Ajeng Anissa WidiatriBelum ada peringkat
- Structural Soils Engineer's Quick Reference GuideDokumen64 halamanStructural Soils Engineer's Quick Reference GuideGreg McNamaraBelum ada peringkat
- Technology: ControlsDokumen32 halamanTechnology: ControlsAli Hossain AdnanBelum ada peringkat
- Liquefaction AnalysisDokumen16 halamanLiquefaction AnalysisKristi Engineering Services Pvt. Ltd.Belum ada peringkat
- Valuation and Risk ModelsDokumen226 halamanValuation and Risk Models1690295017qq.comBelum ada peringkat
- LCD Interfacing PDFDokumen37 halamanLCD Interfacing PDFPeeyush Kp100% (1)
- Scalable Analog/Addressable Fire Alarm Control System: IFP-2100ECS / IFP-2100ECSBDokumen2 halamanScalable Analog/Addressable Fire Alarm Control System: IFP-2100ECS / IFP-2100ECSBMurali DaranBelum ada peringkat
- Application of Nitrous Oxide in AutomobilesDokumen26 halamanApplication of Nitrous Oxide in AutomobilesMohammed NuhmanBelum ada peringkat
- Dell Emc Data Domain Dd3300 Faq: Frequently Asked QuestionsDokumen14 halamanDell Emc Data Domain Dd3300 Faq: Frequently Asked QuestionseriquewBelum ada peringkat
- SAT-101 User Manual: Document No: MAN-0013 Issue No: 4 Dated: 24 Aug 2004Dokumen24 halamanSAT-101 User Manual: Document No: MAN-0013 Issue No: 4 Dated: 24 Aug 2004VM ServicesBelum ada peringkat
- Charger Energic Plus (Catalog)Dokumen24 halamanCharger Energic Plus (Catalog)sugar44Belum ada peringkat
- RPGS Complete Lecture Note1 Inpdf Compressed1Dokumen203 halamanRPGS Complete Lecture Note1 Inpdf Compressed1Purusottam PandeyBelum ada peringkat
- Midterm - ICT1 ExamDokumen4 halamanMidterm - ICT1 ExamHelen PerlasBelum ada peringkat
- Mechanical Technology Concepts: Applied ThermodynamicsDokumen45 halamanMechanical Technology Concepts: Applied ThermodynamicsLe0 GamingBelum ada peringkat
- Wilo Fire Fighting BrochureDokumen20 halamanWilo Fire Fighting BrochureAkhmad Darmaji DjamhuriBelum ada peringkat
- Fontargen AJ PDFDokumen282 halamanFontargen AJ PDFantonyBelum ada peringkat
- KN Yb 1000Dokumen13 halamanKN Yb 1000taharBelum ada peringkat
- PDCT - Hydraulic Torque Wrench - Hangzhou Penad Machinery Co.,LtdDokumen4 halamanPDCT - Hydraulic Torque Wrench - Hangzhou Penad Machinery Co.,LtdQS BMDSBelum ada peringkat
- Graph Theory 6Dokumen30 halamanGraph Theory 6Zeeshan AhmedBelum ada peringkat
- Speed Control of DC Shunt MotorDokumen7 halamanSpeed Control of DC Shunt MotorAakash0% (1)
- May June 2019 P02Dokumen40 halamanMay June 2019 P02Rongrui ZhouBelum ada peringkat
- Classification of The Concha Type Microtia and Their New Suitable Tratment Strategies Without Autogenous Costal Cartilage GraftingDokumen7 halamanClassification of The Concha Type Microtia and Their New Suitable Tratment Strategies Without Autogenous Costal Cartilage Graftingromina paz morales camposBelum ada peringkat
- Definition, Scope and Nature of EconomicsDokumen29 halamanDefinition, Scope and Nature of EconomicsShyam Sunder BudhwarBelum ada peringkat
- ScanKromsator ManualDokumen9 halamanScanKromsator ManualcensoredchapterBelum ada peringkat