Corrective and Preventive Actions: "A Five Step Approach"
Diunggah oleh
Arun KumarJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Corrective and Preventive Actions: "A Five Step Approach"
Diunggah oleh
Arun KumarHak Cipta:
Format Tersedia
Corrective and
Preventive Actions
A Five Step Approach
Topics to Be Covered
What is CAPA?
Governing authority
Five steps to a good CAPA
process
Where companies have
difficulty
Example citations
Recap
What Is CAPA?
Corrective Action Preventive Action
Eliminate Prevent
detected nonconformity
nonconformity occurrence
CAPA Process Map
Deviations/OOS/Failure Problem
Occurs
Determine Root Cause
Determine Corrective Action
Initiate CAR
CAR Respondent(s) and
Approver(s) Determined
CAPA Process Map
Respondent(s) Provides Corrective Responses, Root Cause
Verification, and Implement Due Dates
Response(s) Summarized
Corrective Action
Implementation begins
Respondent(s) review similar systems for Preventive Action
Opportunities
Effectiveness review date set
CAPA Process Map
Respondent(s) sign-off when
implementation is complete
Effectiveness is reviewed
and signed-off
CAR Closed
Records and Reports
Any unexplained discrepancy shall be thoroughly investigated.
The investigation shall extend to other batches that may have
been associated with the specific failure or discrepancy.
A written record of the investigation shall be made and shall
include conclusions and follow up
Written procedures should be established for investigating
critical deviations or failures of intermediate or final batches to
meet specifications.
Investigations should extend to other batches.
Corrective and Preventive Action
Manufacturer shall establish procedures for implementing
corrective and preventive action.
The procedures shall include requirements for:
1) Analyzing processes, work operations, concessions,
quality audit reports, quality records, service records,
complaints, returned product.
2) identify existing and potential causes of
nonconforming product, or other quality problems
Statistical methodology shall be employed to detect
recurring quality problems
3) Investigating the cause of nonconformities relating to
product, processes, and the quality system.
Corrective and Preventive Action
4) Identify action(s) needed to correct and prevent
recurrence of nonconforming product and other quality
problems;
5) Verifying or validating the corrective and preventive
action to ensure actions are effective;
6) Implement and record changes in methods and
procedures needed to correct and prevent identified
quality problems;
7) Submitting relevant information on corrective and
preventive actions, for management review.
1. Failure to implement corrective/preventive
action or conduct a thorough investigation
a) Repeated test failures not investigated
b) Inadequate investigation of failed
particulate inspection
Failure to document corrective action
regarding instrument calibration check
which did not meet specification
Failure to establish and maintain adequate procedures
for implementing corrective and preventive action, as
required.
For-example:
(a) The procedure titled corrective Action Handling
[redacted] was not approved and implemented to
address corrective and preventive action and no
established procedure was found to have been in
place.
Failure to adequately establish and maintain
procedures for implementing corrective and preventive
actions as required
Example : a. Corrective Action Request # & # were not
closed out by the QC Supervisor as required by (SOP)
Corrective and Preventive Action, Rev 1.
Failure to adequately establish and maintain procedures for
implementing corrective and preventive actions as required
Example :(b) (SOP) Corrective and Preventive Action, identifies
repair reports as a source for identification of potential CAPA
activities; however, repair reports are not being trended or
reviewed for CAPA
RECAP
Anda mungkin juga menyukai
- Capa ToolsDokumen44 halamanCapa ToolsChandan ShahBelum ada peringkat
- Good Distribution Practices A Complete Guide - 2021 EditionDari EverandGood Distribution Practices A Complete Guide - 2021 EditionBelum ada peringkat
- GMP Audit Checklist For ManufacturersDokumen90 halamanGMP Audit Checklist For ManufacturersJagtar Singh Chandel100% (2)
- Iqoqpq RMG PDFDokumen11 halamanIqoqpq RMG PDFsrinivas rBelum ada peringkat
- GMP Supplier Assessment QuestionnaireDokumen2 halamanGMP Supplier Assessment Questionnairedrs_mdu48Belum ada peringkat
- CAPA ExampleDokumen5 halamanCAPA Examplechemist_tmaBelum ada peringkat
- Asean GMP Training ModuleDokumen50 halamanAsean GMP Training ModuleMarinelle AngelesBelum ada peringkat
- OQ FormatDokumen13 halamanOQ FormatAliqahwash100% (1)
- Eu GMPDokumen16 halamanEu GMPamirin_kingBelum ada peringkat
- Validation VialWasher OQ NIHDokumen30 halamanValidation VialWasher OQ NIHcongacon3aBelum ada peringkat
- VMP For EVFDokumen56 halamanVMP For EVFPrashansa ShresthaBelum ada peringkat
- GMP Audit Inspections and Audit PreparationsDokumen19 halamanGMP Audit Inspections and Audit PreparationsabhijitBelum ada peringkat
- Analytical Method TransferDokumen36 halamanAnalytical Method Transferimawan_wast100% (1)
- PICS Inspection QC LabDokumen18 halamanPICS Inspection QC LabMax HuangBelum ada peringkat
- Training in PharmaDokumen4 halamanTraining in PharmaPrince MoniBelum ada peringkat
- VMPDokumen45 halamanVMPAshok Lenka100% (1)
- Requirements For Good Documentation Practice (GDP) - PharmaceutDokumen3 halamanRequirements For Good Documentation Practice (GDP) - PharmaceutRizki AmeliaBelum ada peringkat
- Quality Assurance (QA) Management Procedures: By: Pharma Tips - Views: 14415 - Date: 06-May-2012Dokumen6 halamanQuality Assurance (QA) Management Procedures: By: Pharma Tips - Views: 14415 - Date: 06-May-2012SrinivasBelum ada peringkat
- Writing SOP For CAPADokumen17 halamanWriting SOP For CAPACrystalBelum ada peringkat
- Annual Products ReviewDokumen5 halamanAnnual Products Reviewmainart50% (2)
- GMP Slide ShareDokumen77 halamanGMP Slide SharePenmetsa Satyanarayana RajuBelum ada peringkat
- Enkap Quality Manual PDFDokumen19 halamanEnkap Quality Manual PDFpiyusharora1964Belum ada peringkat
- Validation PolicyDokumen3 halamanValidation PolicyneppoanandBelum ada peringkat
- Capa SopDokumen23 halamanCapa SopHaroon Rasheed100% (3)
- F Deviation Investigation ReportDokumen4 halamanF Deviation Investigation ReportBilal AbbasBelum ada peringkat
- RTP 10i Tablet Press IQ/OQ: We Don't Just Sell Machines - We Provide ServiceDokumen34 halamanRTP 10i Tablet Press IQ/OQ: We Don't Just Sell Machines - We Provide ServiceAndreas StathatosBelum ada peringkat
- Access Control To GMP AreaDokumen4 halamanAccess Control To GMP AreaNishit SuvaBelum ada peringkat
- Dietary Supplement Documentation Self-AssessmentDokumen2 halamanDietary Supplement Documentation Self-AssessmentJ.w. MorganBelum ada peringkat
- Audit Report With GMP Questionnaire (Template)Dokumen106 halamanAudit Report With GMP Questionnaire (Template)Anonymous FH6OddBelum ada peringkat
- 9 C Validation Protocol TABLETDokumen20 halaman9 C Validation Protocol TABLETMohammed ZubairBelum ada peringkat
- Lifecycle Approach To Cleaning ValidationDokumen60 halamanLifecycle Approach To Cleaning Validational68130Belum ada peringkat
- Product Quality Review FlowDokumen1 halamanProduct Quality Review Flownasreen anjumBelum ada peringkat
- SMF Update SOLITAIRE PDFDokumen35 halamanSMF Update SOLITAIRE PDFmaheshBelum ada peringkat
- SOP On Handling of DeviationsDokumen10 halamanSOP On Handling of Deviationschemist_tma100% (1)
- QRM SOP Issue# 01 ApprovedDokumen9 halamanQRM SOP Issue# 01 ApprovedibrahimgomaaBelum ada peringkat
- GMP ComparisonDokumen84 halamanGMP ComparisonRoberto FernandezBelum ada peringkat
- International Research Journal of Pharmacy: A Review On Concept of Cleaning Validation in Pharmaceutical IndustryDokumen3 halamanInternational Research Journal of Pharmacy: A Review On Concept of Cleaning Validation in Pharmaceutical IndustryMohamed KamalBelum ada peringkat
- CGMP Training ToolDokumen21 halamanCGMP Training Toolbabusure99Belum ada peringkat
- Alarm Verification SOPDokumen7 halamanAlarm Verification SOPBALU LTD BALU PHARMACEUTICALBelum ada peringkat
- Good Documentation Practices - IVT - JVTDokumen7 halamanGood Documentation Practices - IVT - JVTdcharliesBelum ada peringkat
- Annual Products ReviewDokumen12 halamanAnnual Products ReviewMubeen Khan100% (1)
- Sop (Capa)Dokumen7 halamanSop (Capa)Sudhanshu ShekharBelum ada peringkat
- Site Master FileDokumen16 halamanSite Master Fileshrikantmsd0% (1)
- Annual Product Review Developing An SOPDokumen26 halamanAnnual Product Review Developing An SOPanants2567% (3)
- PROCESS VALIDATION PROTOCOL ExampleDokumen14 halamanPROCESS VALIDATION PROTOCOL ExampleNazia KabirBelum ada peringkat
- Equipment Logbook 2 2Dokumen7 halamanEquipment Logbook 2 2Belazouz BoualemBelum ada peringkat
- Laboratory Quality Agreement TamplateDokumen10 halamanLaboratory Quality Agreement TamplateMina Maher MikhailBelum ada peringkat
- SOP For Reduce Testing For Raw MaterialDokumen3 halamanSOP For Reduce Testing For Raw MaterialMubarak Patel100% (1)
- FDA QSIT GuidelineDokumen108 halamanFDA QSIT GuidelineTim Sandle100% (2)
- Current Good Manufacturing Practices in Food IndustryDokumen3 halamanCurrent Good Manufacturing Practices in Food IndustryCarlBelum ada peringkat
- Product Quality Risk AssessmentDokumen8 halamanProduct Quality Risk AssessmentApoloTrevino100% (1)
- Guideline For Drug Master FilesDokumen33 halamanGuideline For Drug Master FilesRambabu komati - QA100% (3)
- GMP For Dietary SupplementsDokumen6 halamanGMP For Dietary Supplementsmelimaulani-1Belum ada peringkat
- Cleaning Validation Lifecycle ApproachDokumen50 halamanCleaning Validation Lifecycle ApproachlmqasemBelum ada peringkat
- Pharmaceutical Master Validation Plan The Syed Imtiaz 57743816Dokumen3 halamanPharmaceutical Master Validation Plan The Syed Imtiaz 57743816Dandung RuskarBelum ada peringkat
- Om Megashree Pharmaceutical Pvt. LTD: Jugedi, ChitwanDokumen16 halamanOm Megashree Pharmaceutical Pvt. LTD: Jugedi, ChitwanbpharmbaBelum ada peringkat
- 01.collection, Storage and Control of Reserve Samples For Drug ProductsDokumen4 halaman01.collection, Storage and Control of Reserve Samples For Drug ProductsBejoy KarimBelum ada peringkat
- Natural Gas and LPG DataDokumen2 halamanNatural Gas and LPG DataArun KumarBelum ada peringkat
- JokesDokumen27 halamanJokesArun KumarBelum ada peringkat
- 13 Parking ControlDokumen62 halaman13 Parking ControlArun KumarBelum ada peringkat
- PoetryDokumen2 halamanPoetryArun KumarBelum ada peringkat
- Load Cell CalculationsDokumen6 halamanLoad Cell CalculationsArun KumarBelum ada peringkat
- Candidate Appraisal FormDokumen2 halamanCandidate Appraisal FormArun KumarBelum ada peringkat
- Ehandbook Process Safety 1309 PDFDokumen15 halamanEhandbook Process Safety 1309 PDFArun KumarBelum ada peringkat
- Veg DishesDokumen45 halamanVeg DishesArun KumarBelum ada peringkat
- Seko Dosing DiaDokumen12 halamanSeko Dosing DiaSeptriani LaoliBelum ada peringkat
- Load Cell CalculationsDokumen6 halamanLoad Cell CalculationsArun KumarBelum ada peringkat
- Cutting Plate 2Dokumen1 halamanCutting Plate 2Arun KumarBelum ada peringkat
- Unit Conversion: LengthDokumen35 halamanUnit Conversion: LengthAnonymous 8aj9gk7GCLBelum ada peringkat
- Melting Points of MetalsDokumen1 halamanMelting Points of MetalsArun KumarBelum ada peringkat
- Non Veg DishesDokumen99 halamanNon Veg DishesArun KumarBelum ada peringkat
- Nyc-Asme 2013 PDFDokumen45 halamanNyc-Asme 2013 PDFEvriMert RüzgArdaBelum ada peringkat
- Melting Points of MetalsDokumen1 halamanMelting Points of MetalsArun KumarBelum ada peringkat
- Veg DishesDokumen45 halamanVeg DishesArun KumarBelum ada peringkat
- Windows 10 TipsDokumen7 halamanWindows 10 TipswofebaBelum ada peringkat
- Industrial Pipe Marking GuideDokumen2 halamanIndustrial Pipe Marking GuideArun KumarBelum ada peringkat
- Risk Ranking TemplateDokumen4 halamanRisk Ranking TemplateArun KumarBelum ada peringkat
- QMS For Shah Promotors & Developers PDFDokumen52 halamanQMS For Shah Promotors & Developers PDFAhmed El Shafie100% (3)
- Pug Mills / Paddle Mixers: Design & Principles of OperationDokumen1 halamanPug Mills / Paddle Mixers: Design & Principles of OperationArun KumarBelum ada peringkat
- Safety Is Money in The BankDokumen3 halamanSafety Is Money in The BankArun KumarBelum ada peringkat
- Ims Iso 9001 Iso 14001 Ohsas 18001 Documentation PackageDokumen9 halamanIms Iso 9001 Iso 14001 Ohsas 18001 Documentation PackageArun KumarBelum ada peringkat
- Safety Management SystemDokumen17 halamanSafety Management SystemPradeep NegiBelum ada peringkat
- Earthing RulesDokumen9 halamanEarthing RulesArun KumarBelum ada peringkat
- Geometry Formula SheetDokumen1 halamanGeometry Formula SheetArun KumarBelum ada peringkat
- Glass Bead Making F'urnaceDokumen3 halamanGlass Bead Making F'urnaceArun KumarBelum ada peringkat
- Glass GlossaryDokumen3 halamanGlass GlossaryArun Kumar100% (1)
- SKF BRGDokumen22 halamanSKF BRGVijeth99Belum ada peringkat
- Beda Antara Verifikasi & ValidasiDokumen18 halamanBeda Antara Verifikasi & ValidasiAgustine SetiawanBelum ada peringkat
- Eco Green Park BatuDokumen10 halamanEco Green Park BatukireinafadhillaBelum ada peringkat
- APQP4 BDokumen95 halamanAPQP4 BLise RobertBelum ada peringkat
- Difference Between QA and QC: Quality Assurance (QA) and Quality Control (QC) in A Bit Different Way. AlsoDokumen3 halamanDifference Between QA and QC: Quality Assurance (QA) and Quality Control (QC) in A Bit Different Way. Alsocamasa2011Belum ada peringkat
- Duties of Piping InspectorDokumen4 halamanDuties of Piping Inspectorروشان فاطمة روشان100% (1)
- Rapid Survey - TAPDK 2019Dokumen18 halamanRapid Survey - TAPDK 2019Ahmad Rizal FauziBelum ada peringkat
- Fda Ich 8,9,10Dokumen30 halamanFda Ich 8,9,10Sa'ed Abu Yahia100% (1)
- CAPA Change ManagementDokumen22 halamanCAPA Change ManagementMuhammadAmdadulHoque100% (1)
- O J T Check SheetDokumen1 halamanO J T Check SheetabhishekBelum ada peringkat
- Aplicatie Practica Catapulta TUVDokumen26 halamanAplicatie Practica Catapulta TUVwalaBelum ada peringkat
- EuroQoL Quality of Life Scale (EQ-5D) PDFDokumen2 halamanEuroQoL Quality of Life Scale (EQ-5D) PDFCicci HoffBelum ada peringkat
- Blok A Gas Development Project Request For Inspection (RFI) / Inspection ResultDokumen5 halamanBlok A Gas Development Project Request For Inspection (RFI) / Inspection ResultIjoel PutraBelum ada peringkat
- Formulae and Data SheetDokumen3 halamanFormulae and Data SheetHemantBelum ada peringkat
- Quality Assurance - Control PlanDokumen104 halamanQuality Assurance - Control PlanAhmed SamyBelum ada peringkat
- Statistical Process ControlDokumen40 halamanStatistical Process ControlBrixter GervacioBelum ada peringkat
- Effci GMP Guide - 2008Dokumen48 halamanEffci GMP Guide - 2008Juan Pablo MedinaBelum ada peringkat
- Supplier Quality ManualDokumen29 halamanSupplier Quality Manualppanambo100% (2)
- Tuboscope Mexico - ISO 90012015 - Expires 022023Dokumen6 halamanTuboscope Mexico - ISO 90012015 - Expires 022023Ali KianersiBelum ada peringkat
- Jurnal PDCADokumen6 halamanJurnal PDCAJuwita Verawati SiahaanBelum ada peringkat
- Top 100 Lean BooksDokumen6 halamanTop 100 Lean Bookssribalaji22Belum ada peringkat
- Poison Cake Test: Standard Operating ProcedureDokumen1 halamanPoison Cake Test: Standard Operating ProcedureGirdhar Sahu100% (3)
- Reliability Test Using Cronbach Alpha: Formula SolutionDokumen1 halamanReliability Test Using Cronbach Alpha: Formula SolutionJA BerzabalBelum ada peringkat
- Hafifah - AK1018016Dokumen12 halamanHafifah - AK1018016hapipahBelum ada peringkat
- Prsentation On 5S KaizenDokumen21 halamanPrsentation On 5S Kaizenhandsomekash0% (1)
- Chapter 10: Principles of Six SigmaDokumen25 halamanChapter 10: Principles of Six SigmaGrant Marco MarianoBelum ada peringkat
- Vital Connections Organizational Chart: VCM Governing Board & Policy CouncilDokumen1 halamanVital Connections Organizational Chart: VCM Governing Board & Policy Councilrohan dixitBelum ada peringkat
- Christian N. Madu PH.D (Auth.) - Handbook of Total Quality Management-Springer US (1998) PDFDokumen829 halamanChristian N. Madu PH.D (Auth.) - Handbook of Total Quality Management-Springer US (1998) PDFAL KURNIA SANDIBelum ada peringkat
- Managing Business Individual Assignment APUDokumen12 halamanManaging Business Individual Assignment APUHussein Al-wahaibiBelum ada peringkat
- GE2022 TQM Question BankDokumen21 halamanGE2022 TQM Question BankVadivel AeroBelum ada peringkat
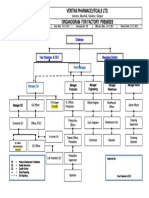
- Veritas Pharmaceuticals Ltd. Organogram For Factory PremisesDokumen1 halamanVeritas Pharmaceuticals Ltd. Organogram For Factory PremisesdidarBelum ada peringkat