Histopatholoy Techniques
Diunggah oleh
Vickypatho100%(1)100% menganggap dokumen ini bermanfaat (1 suara)
571 tayangan27 halamanThis document discusses the key steps in histopathological tissue processing techniques, which include fixation, dehydration, clearing, embedding, section cutting, and staining of tissue samples. The most common fixative used is 10% buffered formaldehyde solution, as it adequately fixes tissues up to 5mm in thickness within 24-48 hours. After fixation, tissues undergo dehydration using increasing concentrations of ethanol or acetone to remove water. Clearing agents like benzene or cedar wood oil are then used to remove dehydrating agents prior to embedding tissues in paraffin wax for section cutting on a microtome.
Deskripsi Asli:
Histopathological techniques
Hak Cipta
© © All Rights Reserved
Format Tersedia
PPT, PDF, TXT atau baca online dari Scribd
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniThis document discusses the key steps in histopathological tissue processing techniques, which include fixation, dehydration, clearing, embedding, section cutting, and staining of tissue samples. The most common fixative used is 10% buffered formaldehyde solution, as it adequately fixes tissues up to 5mm in thickness within 24-48 hours. After fixation, tissues undergo dehydration using increasing concentrations of ethanol or acetone to remove water. Clearing agents like benzene or cedar wood oil are then used to remove dehydrating agents prior to embedding tissues in paraffin wax for section cutting on a microtome.
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PPT, PDF, TXT atau baca online dari Scribd
100%(1)100% menganggap dokumen ini bermanfaat (1 suara)
571 tayangan27 halamanHistopatholoy Techniques
Diunggah oleh
VickypathoThis document discusses the key steps in histopathological tissue processing techniques, which include fixation, dehydration, clearing, embedding, section cutting, and staining of tissue samples. The most common fixative used is 10% buffered formaldehyde solution, as it adequately fixes tissues up to 5mm in thickness within 24-48 hours. After fixation, tissues undergo dehydration using increasing concentrations of ethanol or acetone to remove water. Clearing agents like benzene or cedar wood oil are then used to remove dehydrating agents prior to embedding tissues in paraffin wax for section cutting on a microtome.
Hak Cipta:
© All Rights Reserved
Format Tersedia
Unduh sebagai PPT, PDF, TXT atau baca online dari Scribd
Anda di halaman 1dari 27
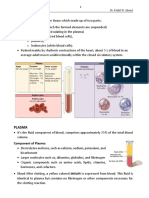
HISTOPATHOLOGICAL TECHNIQUES
Concerned with the demonstration of minute tissue
structures in disease. Before such structures can be
demonstrated, the tissue must be prepared in such a
manner that it is sufficiently thin (one or two cells thick)
to be examined microscopically and the many and
complex structures which go to make up tissue may be
differentiated.
This involves collection of morbid tissues from biopsy or
necropsy, fixation, washing, dehydration, clearing,
embedding, section cutting, staining and microscopical
examination.
Fixation
Rapid killing of tissue elements and their hardening so that
they are preserved in as near as possible the same relations they had
in living body.
The foundation of all good histopathological preparations
is adequate and complete fixation.
Fixation is required to:-
Prevent post-mortem changes such as putrefaction and
autolysis.
Preserve various cell constituents in as life-like manner as
possible.
Protect by hardening of naturally soft tissues thereby
allowing easy manipulations during subsequent processing.
Convert the normal semi-fluid consistency of cell to an
irreversible semisolid consistency.
Aid in visual differentiation (alteration in refractive indices)
of structures by application of biological dyes and chemicals.
To accomplish the objectives of the fixation of
tissues:-
Should be placed in the fixative immediately upon removal
from the body or as soon as possible after death.
Tissue blocks should be cut thin enough (3-5 mm thick) so
that the fixative readily penetrates throughout in a
reasonably short time.
The volume of fixing fluid should be 15 to 20 times that of
the tissue to be fixed.
The tissues which tend to float (lungs and bone marrow)
should be covered with a piece of gauze or absorbent cotton.
Several reagents are used for fixing tissues.
The choice of a fixative will be governed by the type of
investigation required, both immediately and in future.
The most common fixative used is the formaldehyde
(formalin). It is commonly used as 10 per cent solution i.e. 1
volume 40 per cent formaldehyde solution (formalin) with 9
volumes of water (simple 10% formalin) or with normal saline
(formol saline).
It is desirable to render the formalin solution neutral by
adding 4.0 gm sodium phosphate monobasic and 65 gm.
anhydrous disodium phosphate per litre.
This constitutes the buffered neutral formalin which is
considered the best overall fixative and is therefore strongly
recommended for routine use.
Tissues up to 5 mm thickness are adequately fixed in
24-48 hours depending upon the temperature.
Factors affecting the fixation:
- pH.
- Temperature.
- Penetration of fixative.
- Volume of tissue.
According to previous factors we can determine the
concentration of fixative and fixation time.
Types of fixative:
Acetic acid, Formaldehyde, Ethanol, Glutaraldehyde,
Methanol and Picric acid.
Dehydration
Dehydration is removal of all extractable water
by a dehydrant diffusing through the tissues and in
the process diluting itself 2-4%.
Some of the dehydrants used are ethyl alcohol
(ethanol), acetone, dioxane, tetrahydro furan,
isopropyl alcohol, rectified spirit.
Most common are ethyl alcohol or acetone.
Ethyl alcohol :
It is the most commonly used dehydrant and ascending
grades of alcohol are used for this purpose.
The concentration of alcohol in the first bath depends on
the fixation, size and type of tissue to be dehydrated.
After fixation in aqueous solutions, delicate tissues need to
be dehydrated slowly starting in 50% ethyl alcohol whereas
most tissue specimens may be poured into 70% alcohol.
Tissues immersed in too great a concentration of alcohol
after an aqueous fixative will usually show a high degree of
shrinkage due to the too rapid removal of the water.
Tissues from alcoholic fixatives such as carnoys fluid may
be placed in higher grade alcohols or even in absolute alcohol
but it should be remembered in these instances that several
changes are needed to remove acids.
The minimum duration of treatment in
graded alcohols will depend on the size and
type of tissue.
For routine purposes, tissues are dehydrated
in 70%, 80%, 90% and absolute alcohol for 2-4
hours in each dilution.
The volume of reagent (alcohol) should be 50
times the bulk of specimen.
To speed the process of dehydration, the
tissue may be agitated either mechanically in
an automatic tissue processor or by shaking the
container periodically.
Clearing
Since the dehydrating agents are not miscible with
commonly employed embedding materials
(paraffin), these must be removed by clearing
agents.
Removal of the dehydrant makes the tissue some
what translucent (clear) except in chloroform.
The most common clearing agents in use are
chloroform, carbon tetra chloride, xylene or xylol,
benzene, toluene, cedar wood oil.
Most commonly used clearing agent is benzene or
cedar wood oil.
Clearing
Replacing the dehydrating fluid with a fluid that is totally
miscible with both the dehydrating fluid and the embedding
medium.
Choice of a clearing agent depends upon the
following:
- The type of tissues to be processed, and the type of
processing to be undertaken
- The processor system to be used
- Intended processing conditions such as temperature,
vacuum and pressure
- Safety factors
- Cost and convenience
- Speedy removal of dehydrating agent
- Ease of removal by molten paraffin wax
- Minimal tissue damage
Benzene:-
Benzene is fairly rapid in action and small
pieces of tissues are cleared in to 1 hour.
Possesses the advantage that the tissue
becomes clearer as the alcohol is replaced.
possible to determine the end point with
some accuracy.
Over exposure of tissues to it may result
in hardening of tissues.
Cedar wood oil:-
Best reagent for research and treatment of delicate tissues since
it has the least hardening effect.
Tissues may be left in this clearing agent for long periods, even
for months without damage.
In some institutions it is used as a clearing agent for hard
tissues such as skin and dense fibrous tissue. Even small pieces
of tissue need to be left overnight in cedar wood oil.
Following clearing in this reagent, several extra changes of
paraffin wax will be required to remove the oil.
In order to avoid extra changes of paraffin wax, tissues after
clearing in cedar wood oil may be placed for few minutes in
xylene. This treatment will remove excess of cedar wood oil from
the tissues.
Impregnation with paraffin or
Embedding
After clearing, the tissues are impregnated with
paraffin wax in an electric oven heated to 54-62
o
C,
the temperature depending on the melting point of
the wax in use.
The temperature of the oven should be slightly (1-
2
o
C) above the melting point of paraffin wax. The
most popular melting point for routine work under
Indian conditions is 60-62
o
C which gives support for
hard tissues and yet allows easy ribboning of
sections.
After blotting lightly with filter paper, tissue is
transferred from the clearing agent to molten paraffin
wax in open glass containers or copper pots kept in
embedding oven.
The volume of wax should be about 25-50 times the
volume of the tissue.
The length of time and the number of changes
required for thorough impregnation of tissue
with wax largely depends on the size and type
of tissue, the clearing agent employed and
the use of a vacuum embedding oven.
As a routine, 3-4 changes of 1 hour each are
sufficient. By the use of vacuum embedding
oven this time may be halved.
Blocking
Two L-shaped moulds are
arranged in the form of a
rectangle over a glass plate.
Melted paraffin is poured into
the mould and the tissue from
the final wax bath is so oriented
that the surface to be cut rests on
the base of mould.
Block is then allowed to cool.
When the wax has set, the L
pieces are easily removed and
are ready for next block.
Section Cutting
The cutting of good sections depends on
thorough knowledge of the equipment used and
the practical experience.
Trim the block with a sharp knife until 1/8
inch wax remains on all sides of tissue and the
edges of block are parallel.
Fix the block to metal block holder by
heating the latter and pressing it firmly against
the back of paraffin block.
The block holder together with block is
dipped in cold water.
Fix the block holder with
block on the microtome in
such a position that it will be
clear of the knife when this is
in position.
Turn backs the feed
mechanism on the
microtome almost as far as it
will go.
Insert the appropriate
knife in the knife holder and
screw it tightly in position.
Check that the tilt of the knife is set
at the correct angle.
Move the block holder forward or
upward or adjust the feed mechanism
until the wax block is almost touching
the knife.
Ensure that leading edge of block is
parallel to the edge of the knife.
To trim the surface of block, set the section
thickness gauge to about 15 microns and
operate the microtome until complete sections
of the tissue are being cut.
Set the thickness gauge to 4-5 u and operate
the microtome gently until complete sections
are cut.
With practice an easy rhythm is achieved
with the right hand operating the microtome
and the left hand holding the sections away
from the knife either with a needle or forceps.
Fixing the sections to slides
During cutting, paraffin wax embedded sections
become slightly compressed and creased.
Before being attached to slides these creases must
be removed and the sections flattened.
This is achieved by floating the ribbon on warm
water in thermostatically controlled baths known as
tissue flotation water bath.
These baths are commercially available with the
inside coloured black and are maintained at a
temperature 5-6
o
C below the melting point of
paraffin wax.
When the sections are flattened, these are lifted
on to the glass slide.
For this, a clean or albuminized slide (slides
smeared with Mayers egg albumin which contain
equal amount of egg white and glycerin) is half
submerged in the water and brought into contact
with the section.
The slide is then withdrawn bringing the
flattened sections with it.
The water is drained from beneath the sections
and these are then dried in an oven at 50-60oC.
The slides are now ready for staining.
Staining of Paraffin Sections
The most common method of staining of paraffin cut
sections for routine histopathological studies in Haematoxylin
and Eosin (H & E).
The usual procedure is:
Deparaffinize the sections in 2-3 changes of xylene for 5-10
minutes each.
Place in absolute alcohol 90% alcohol, 80% alcohol and 70%
alcohol for 1-23 minutes each.
Wash in tap water.
Stain with haematoxylin for 5-10 minutes.
Wash in tap water
Differentiate in acid alcohol, 2-5 quick dips depending upon the
intensity of stain.
Check the differentiation with a microscope.
Nuclei should be distinct and the background very light or
colourless.
Wash in tap water for 2-5 minutes.
Dip in ammonia water or lithium carbonate water until sections
are bright blue.
Wash in running tap water for 15-20 minutes. This step is
important because in case the washing is inadequate, eosin will
not stain evenly.
Stain with eosin for 15 seconds to 2 minutes depending on the
concentration of eosin and depth of the counterstain desired.
Dehydrate in ascending grades of alcohol 70%,
80%, 90% and absolute, 1-2 dips each.
Clear in xylene, 2-3 changes of 2 minutes each.
Mount in D.P.X. and examine under the microscope.
Results: Nuclei blue
Cytoplasm various shades of pink
Mounting of sections
Histopathological sections which need to be
examined for any length of time or to be stored must be
mounted under a cover slip. The media which are commonly
employed for mounting are:
i) Canada Balsam
ii) D.P.X.
Canada Balsam is solid resin from the Canadian fir
tree (Abies balsamea) and is usually dissolved in xylene; a 55
per cent solution is suitable for routine use.
It is not commonly used these days because
i) it turns yellow with time
ii) sections stained with aniline dyes tend to fade and Prussian
blue is slowly bleached
iii) it dries slowly and
iv) excess mountant is not easily removed.
In most of the histopathological laboratories, Canada balsam
has been replaced by D.P.X., a synthetic resin. It preserved
most routine stains, dries quickly and the slides can be
cleaned of excess mountant simply by stripping it off after
cutting around the edge of the cover slip.
Composition of D.P.X.
Distrene 80 - 10 gm
Dibutylphthalate - 5 ml
Xylene - 35 ml
Anda mungkin juga menyukai
- Microtechniques 2023Dokumen14 halamanMicrotechniques 2023nabaa ahmedBelum ada peringkat
- Tissue ProcessingDokumen31 halamanTissue ProcessingZia Ashraf ChaudharyBelum ada peringkat
- Preparation of TissueDokumen25 halamanPreparation of TissueAhmed JawdetBelum ada peringkat
- Preparation of Histological SpecimensDokumen26 halamanPreparation of Histological SpecimensMuhammad RizkyBelum ada peringkat
- Methods of Histological Study: Fixation, Embedding, Sectioning and StainingDokumen48 halamanMethods of Histological Study: Fixation, Embedding, Sectioning and StainingAhmad QuraanBelum ada peringkat
- Histochemistry ZS (NEU2012)Dokumen82 halamanHistochemistry ZS (NEU2012)Oben UğurBelum ada peringkat
- HISTOPATHOLOGY TITLEDokumen9 halamanHISTOPATHOLOGY TITLEIRSHADBHAI BAHELIMBelum ada peringkat
- Methods Used in Histology: Tissue Preparation, Histochemistry, CytochemistryDokumen50 halamanMethods Used in Histology: Tissue Preparation, Histochemistry, Cytochemistrynav_malhiBelum ada peringkat
- Lab1 HistotechniqueDokumen26 halamanLab1 HistotechniqueGitta Lakshita AnggariniBelum ada peringkat
- TonsilasDokumen15 halamanTonsilasLuz InfanteBelum ada peringkat
- Special Staining Techniques for Carbohydrates, Nucleic Acids, Lipids and MucinsDokumen26 halamanSpecial Staining Techniques for Carbohydrates, Nucleic Acids, Lipids and MucinsAbel AlvaBelum ada peringkat
- Special Stains GuideDokumen45 halamanSpecial Stains GuideJerry TurtleBelum ada peringkat
- BloodDokumen6 halamanBloodFadhil Hussam AhmedBelum ada peringkat
- 04 HistotechniquesDokumen70 halaman04 HistotechniquesJulliene Dadole100% (1)
- 2098 Sample Preparation OpticalDokumen27 halaman2098 Sample Preparation OpticalRitesh ThakurBelum ada peringkat
- Histology 01 02Dokumen47 halamanHistology 01 02jhcjdjksahdjkhaBelum ada peringkat
- Immunology Tonsil 2Dokumen31 halamanImmunology Tonsil 2ariienndrrahanniie100% (1)
- MicrotomeDokumen32 halamanMicrotomegandhiayu100% (2)
- H&E Staining Overview: A Guide To Best Practices:, JD CT (Ascp) HTL, Bappsc, FaimsDokumen29 halamanH&E Staining Overview: A Guide To Best Practices:, JD CT (Ascp) HTL, Bappsc, FaimsAnonymous iqoU1mtBelum ada peringkat
- Histology & Its Methods MON 21Dokumen20 halamanHistology & Its Methods MON 21Ayman AlmugaddamiBelum ada peringkat
- An Introduction To Routine and Special StainingDokumen13 halamanAn Introduction To Routine and Special StainingBadiu ElenaBelum ada peringkat
- Spill Kit PresentationDokumen4 halamanSpill Kit PresentationRavipati MouryaBelum ada peringkat
- Nerve StainingDokumen26 halamanNerve Stainingtlyons1188Belum ada peringkat
- Histology: Introduction To Tissue ProcessingDokumen18 halamanHistology: Introduction To Tissue ProcessingalmutazimBelum ada peringkat
- Special Stain PPT by G.P. TiwariDokumen64 halamanSpecial Stain PPT by G.P. TiwariGulab Chand Tiwari Tiwari100% (1)
- Phase Contrast MicrosDokumen2 halamanPhase Contrast MicrosPatrick ManriqueBelum ada peringkat
- Precautions in Handling, Acceptance and Fixation of SpecimenDokumen5 halamanPrecautions in Handling, Acceptance and Fixation of SpecimenMary ChristelleBelum ada peringkat
- Fixation & Fixatives: Histology ProcedureDokumen14 halamanFixation & Fixatives: Histology ProcedureAsyeon GhaziBelum ada peringkat
- 1378968054.1856 - Histochemistry and Enzyme HistochemistryDokumen39 halaman1378968054.1856 - Histochemistry and Enzyme HistochemistryCarlos Ivan Cruz Cardenas100% (1)
- Decalcification: MR G.P. Tiwari Technologist Tata Memorial HospitalDokumen42 halamanDecalcification: MR G.P. Tiwari Technologist Tata Memorial HospitalGulab Chand Tiwari Tiwari100% (5)
- Microtome and Micro To My WordDokumen12 halamanMicrotome and Micro To My WordKaran KapoorBelum ada peringkat
- Introduction To Histology: Preparation of Tissue For Histology Dr. OkoloDokumen38 halamanIntroduction To Histology: Preparation of Tissue For Histology Dr. OkoloAbiola NerdBelum ada peringkat
- 1 Histopathologic TechniquesDokumen47 halaman1 Histopathologic TechniquesShanne Katherine MarasiganBelum ada peringkat
- Histopathology Review NotesDokumen8 halamanHistopathology Review NotesKhoreen ObisoBelum ada peringkat
- AlgaeDokumen34 halamanAlgaeAnand KumarBelum ada peringkat
- Connective Tissue StainsDokumen49 halamanConnective Tissue StainsLloyd LinBelum ada peringkat
- HE StainDokumen2 halamanHE Stainthy100% (2)
- Laser in Medicine: M.Sc. Student Shahad EsmatDokumen17 halamanLaser in Medicine: M.Sc. Student Shahad EsmatShahad AlrubieBelum ada peringkat
- General PathologyDokumen32 halamanGeneral PathologyLeul DawitBelum ada peringkat
- Introduction To Microscope and Histology (With Histopathology)Dokumen169 halamanIntroduction To Microscope and Histology (With Histopathology)Patricia Vermudo100% (1)
- Histology Lecture 1, Introduction (Slides)Dokumen10 halamanHistology Lecture 1, Introduction (Slides)Ali Al-Qudsi100% (2)
- Grossing Technique in BreastDokumen37 halamanGrossing Technique in BreastadiBelum ada peringkat
- Histological TechniquesDokumen25 halamanHistological TechniquesAnna Donato100% (1)
- HP Lec - FixationDokumen4 halamanHP Lec - FixationAaron Vincent MillomedaBelum ada peringkat
- Histological Stains 1Dokumen4 halamanHistological Stains 1Godwin HalfonBelum ada peringkat
- Introduction To HistopathologyDokumen9 halamanIntroduction To HistopathologyNur LiyanaBelum ada peringkat
- MCQ Sample QuestionsDokumen4 halamanMCQ Sample QuestionsNaveen Vachipalli100% (1)
- Tissue Specimen With Number and Request FormDokumen2 halamanTissue Specimen With Number and Request FormChristine EscolBelum ada peringkat
- Introduction To Tissue ProcessingDokumen21 halamanIntroduction To Tissue ProcessingELIEZER MAYAPITBelum ada peringkat
- Histology of Cartilage and Bone-SLIDE 21Dokumen21 halamanHistology of Cartilage and Bone-SLIDE 21bibeksd796Belum ada peringkat
- Module 19 - Routine and Special Tissue Processing in HistopathologyDokumen100 halamanModule 19 - Routine and Special Tissue Processing in Histopathologyhot silogBelum ada peringkat
- Tissue ProcessingDokumen2 halamanTissue ProcessingAbhinavCyBelum ada peringkat
- Artifacts: A Menace in HistopathologyDokumen3 halamanArtifacts: A Menace in HistopathologyMahesh SataputeBelum ada peringkat
- StainsDokumen4 halamanStainsMonique ManiwanBelum ada peringkat
- His To PathologyDokumen52 halamanHis To PathologyRathinaKumarBelum ada peringkat
- Tissue ProcessingDokumen38 halamanTissue Processingadesiyaniyanu7Belum ada peringkat
- Introduction To HistopathologyDokumen31 halamanIntroduction To HistopathologyRhea PaduaBelum ada peringkat
- Histology - Wet Lab 1Dokumen36 halamanHistology - Wet Lab 1Victoria RennaBelum ada peringkat
- RP, BVD, SF, AsfDokumen34 halamanRP, BVD, SF, AsfVickypathoBelum ada peringkat
- Equine InfluenzaDokumen126 halamanEquine InfluenzaVickypathoBelum ada peringkat
- EiaDokumen14 halamanEiaVickypathoBelum ada peringkat
- Application For The Post ofDokumen1 halamanApplication For The Post ofVickypathoBelum ada peringkat
- AbttDokumen2 halamanAbttVickypathoBelum ada peringkat
- Iqac 2013 120813Dokumen4 halamanIqac 2013 120813VickypathoBelum ada peringkat
- AbttDokumen2 halamanAbttVickypathoBelum ada peringkat
- Actinobacillosis &actinomycosisDokumen16 halamanActinobacillosis &actinomycosisVickypathoBelum ada peringkat
- Nancy Overcott, Dana Gardner Fifty Common Birds of The Upper Midwest 2006Dokumen125 halamanNancy Overcott, Dana Gardner Fifty Common Birds of The Upper Midwest 2006pavel_janda_3Belum ada peringkat
- The Dinosaur Heresies PDFDokumen480 halamanThe Dinosaur Heresies PDFDeyvid Dos Anjos100% (1)
- All About AnalogyDokumen8 halamanAll About AnalogySalman TaurusBelum ada peringkat
- Par OseDokumen6 halamanPar OseloskalahariBelum ada peringkat
- International Symposium on Asian Vertebrate Species DiversityDokumen2 halamanInternational Symposium on Asian Vertebrate Species DiversityMelisa EprianiBelum ada peringkat
- Unit 2.decodable ReadersDokumen93 halamanUnit 2.decodable ReadersTeach math CreativityBelum ada peringkat
- BIO271 - Practice MCQDokumen6 halamanBIO271 - Practice MCQEric LuongBelum ada peringkat
- Asexual Reproduction Webquest PerfectedDokumen6 halamanAsexual Reproduction Webquest PerfectedFBelum ada peringkat
- The history and spread of donkeys in AfricaDokumen9 halamanThe history and spread of donkeys in AfricaTimothy TanBelum ada peringkat
- Dentition in Mammals CC-8 PDFDokumen5 halamanDentition in Mammals CC-8 PDFtapas.manushBelum ada peringkat
- Communication in Plants PDFDokumen2 halamanCommunication in Plants PDFAngelBelum ada peringkat
- 00017Dokumen132 halaman00017Audry AriasBelum ada peringkat
- Understanding PH and BufferDokumen2 halamanUnderstanding PH and BufferRyan Carlo CondeBelum ada peringkat
- Ornamental Fishes of East Kolkata Wetland, West Bengal, IndiaDokumen3 halamanOrnamental Fishes of East Kolkata Wetland, West Bengal, IndiaDR. BIJAY KALI MAHAPATRABelum ada peringkat
- Scope AfterDokumen2 halamanScope AfterCS DeptBelum ada peringkat
- Let - S Speak! - Part 1 A1A2Dokumen5 halamanLet - S Speak! - Part 1 A1A2weronika.betlejewskaBelum ada peringkat
- Artemia SalinaDokumen4 halamanArtemia Salinadhimas06Belum ada peringkat
- McGahan CondorMSDokumen542 halamanMcGahan CondorMSDaniel BlancoBelum ada peringkat
- All Genera of The Infraorder Gryllidea of The WorldDokumen44 halamanAll Genera of The Infraorder Gryllidea of The WorldRodrigo ChangBelum ada peringkat
- Guia Virtual 2 Periodo 2 Inglés 7Dokumen3 halamanGuia Virtual 2 Periodo 2 Inglés 7ThomasBelum ada peringkat
- Anatomy and Physiology of The Spine: Google ImagesDokumen9 halamanAnatomy and Physiology of The Spine: Google Imagesraquel maniegoBelum ada peringkat
- Ammonium Persulfate (A3678) - Product Information SheetDokumen1 halamanAmmonium Persulfate (A3678) - Product Information SheetPatricio Javier Alfaro CerdaBelum ada peringkat
- For Scrap BookDokumen59 halamanFor Scrap BookDon Johnnedel Basa OsitaBelum ada peringkat
- Zoology (T & E) Model Question PaperDokumen8 halamanZoology (T & E) Model Question PaperPalanichamy MuthuBelum ada peringkat
- Questions 1 Through 10 Are Based On The Following PassageDokumen5 halamanQuestions 1 Through 10 Are Based On The Following PassageSarjitoBelum ada peringkat
- Histopath 1Dokumen3 halamanHistopath 1Rudy QuismorioBelum ada peringkat
- The Anatomy of VertebratesDokumen698 halamanThe Anatomy of VertebratesJhenys MaikerBelum ada peringkat
- 09 Chapter 2Dokumen51 halaman09 Chapter 2Anonymous OP6R1ZSBelum ada peringkat
- Alloparental Care DesignsDokumen8 halamanAlloparental Care Designsapi-488072701Belum ada peringkat
- Analysing The Praying MantisDokumen3 halamanAnalysing The Praying Mantissmallbrowndog100% (1)