Patch
Diunggah oleh
Firman oktivendraJudul Asli
Hak Cipta
Format Tersedia
Bagikan dokumen Ini
Apakah menurut Anda dokumen ini bermanfaat?
Apakah konten ini tidak pantas?
Laporkan Dokumen IniHak Cipta:
Format Tersedia
Patch
Diunggah oleh
Firman oktivendraHak Cipta:
Format Tersedia
BAB II
PEMBAHASAN
A. Definisi Sediaan Patch
Sistem penghantaran obat transdermal adalah sistem penghantaran yang
mempertahankan efektivitas pelepasan obat dalam waktu yang lama, mengurangi
intensitas administrasi dan efek samping yang terkait dengan rute pemberian oral.
Obat transdermal adalah kategori obat yang sangat luas yang didefinisikan untuk
mengantarkan obat dengan mekanisme aksi lokal atau sistemik melalui formulasi
dosis tertentu (Khan S., and Sharman T., 2023). Sediaan transdermal memiliki
banyak manfaat dibandingkan penghantaran obat secara oral, termasuk
menghindari metabolisme jalur pertama melindunginya dari kerusakan,
melindungi obat sensitif dari lingkungan gastrointestinal yang keras,
memungkinkan pelepasan obat yang berkelanjutan, dan mempertahankan
konsentrasi plasma yang lebih seragam serta meningkatkan kemungkinan
penggunaan pasien secara konsisten, dan memungkinkan pemberian obat dengan
cara interval stabil yang berkelanjutan . Untuk meningkatkan permeabilitas obat
melintasi subcutan, pendekatan pasif digunakan untuk memengaruhi interaksi
obat seperti peningkat permeasi, nanoemulsi, dan vesikel (Alkilani et.al., 2023).
Sistem penghantaran obat transdermal biasanya memberikan obat yang ditujukan
untuk orang lanjut usia (CHMP, 2014).
Transdermal patch adalah salah satu bentuk sediaan farmasi baru untuk
mengantarkan obat ke aliran darah setelah dilekatkan ke kulit. Patch dimaksudkan
untuk mendistribusikan obat dengan cara yang diatur, dimodifikasi, dan dibatasi
waktu dengan kecepatan yang telah ditentukan sebelumnya. Sistem transdermal
patch sangat efektif dan hemat biaya, dan memberikan alternatif yang baik untuk
pembuatan patch untuk molekul obat yang sangat manjur (Alkilani et.al., 2023).
Gambar: transdermal patch
Transdermal patch adalah sediaan farmasi dengan berbagai ukuran
mengandung satu atau lebih bahan aktif yang dimaksudkan untuk ditempelkan
pada kulit dan mengakomodasi bahan tersebut untuk memasuki sirkulasi sistemik
setelah melewati sawar kulit. Transdermal patch memiliki beberapa keunggulan,
antara lain kemudahan dalam penghantaran obat, pengurangan rasa sakit, dan
mudah pengobatan jika terjadi toksisitas sistemik. Sebagai bentuk sediaan, patch
transdermal terdiri dari banyak sistem, yaitu sistem perekat (menggunakan sistem
matriks (menggunakan polimer perekat), sistem matriks (menggunakan polimer
sebagai matriks/wadah obat), sistem reservoir (menggunakan polimer sebagai
wadah obat dan mengontrol laju pelepasan obat), dan sistem reservoir mikro (Yati
and Pamungkas, 2018).
Prinsip dasar penghantaran obat transdermal patch bergantung pada
kemampuan obat melewati kulit ke dalam sirkulasi sistemik. Oleh karena itu, ada
obat terbatas yang memenuhi kriteria yang dibutuhkan untuk dapat melewati kulit.
Kriteria tersebut antara lain memiliki berat molekul rendah (kurang dari 600
g/mol), memiliki kemampuan melewati epidermis, diserap oleh pembuluh darah,
dan masuk ke dalam sirkulasi. Selain itu, obat aktif harus stabil secara kimia dan
fisik. Zat aktif harus memiliki dosis harian yang rendah untuk kenyamanan pasien
dan kecenderungan perekat. Kulit harus memetabolisme obat. Hanya ada sejumlah
kecil obat transdermal patch yang berhasil dengan sifat spesifik seperti itu (Khan
S., and Sharman T., 2023).
B. Jenis-jenis transdermal patch
Terdapat dua jenis utama sistem patch transdermal tergantung pada
bagaimana zat obat didispersikan ke dalam komponen patch lainnya:
1. Sistem matriks dengan pelepasan obat berdasarkan difusi zat obat.
Sistem matrik pada patch transdermal terdiri dari 2 komponen utama, yaitu
backing layer dan matrik. Pada sistem ini, obat di dalam eksipien seperti
polimer, plasticizer, permeation enhancer dan perekat diformulasikan menjadi
satu, yang kemudian dibiarkan mengering hingga membentuk matrik.
Selanjutnya, matrik ditempelkan pada backing layer. Keuntungan dari sistem
matrik yaitu akan membentuk suatu patch yang tipis sehingga nyaman untuk
digunakan.
Gambar : Sistem matriks dari patch transdermal.
2. Sistem membran (reservoir) yang mengandung kompartemen obat cair
tertentu dan pelepasannya dikendalikan oleh membran. Dalam sistem ini,
reservoir tertanam antara lapisan backing layer dan sebuah lapisan membran.
Lapisan membran dapat berpori atau tidak berpori. Obat bisa dalam bentuk
larutan, suspensi, gel atau tersebar dalam matrik polimer padat. Polimer
hipoalergenik adesif dapat diterapkan sebagai permukaan luar membran
polimer yang kompatibel dengan obat. Sistem ini mengikuti kinetika orde ke
nol (CHMP, 2014).
Gambar: Sistem membran dari patch transdermal
C. Mekanisme Aksi Transdermal Patch
Sediaan transdermal patch merevolusi pasar adalah sediaan patch nikotin,
yang pertama kali diperkenalkan pada tahun 1991 sebagai pengobatan untuk
berhenti merokok. Sejak saat itu telah dikembangkan sejumlah patch yang
berbeda, termasuk patch testosteron untuk hipogonadisme pada pria dan patch
kombinasi estradiol dan norethindrone atau levonorgestrel untuk gejala
menopause dan produk patch lidokain dan fentanil HCl (Paudel, et.al. 2010).
Tantangan terbesar untuk keberhasilan pengiriman obat transdermal adalah
menyebarkan zat aktif melalui fungsi penghalang dari banyak lapisan kulit.
Lapisan terluar kulit, stratum korneum, adalah lapisan paling tebal yang
mengandung banyak lapisan korneosit yang mengandung banyak keratin. Selain
itu, stratum korneum terdiri dari dua daerah yang berbeda secara kimiawi yang
perlu diperhitungkan saat membuat obat transdermal patch. Ada daerah berair di
permukaan luar filamen keratin dan matriks lipid di antara filamen yang
dibutuhkan obat aktif untuk memiliki kemampuan berdifusi melalui keduanya
agar berhasil. Namun, ada kemajuan baru-baru ini dalam pengembangan metode
pengiriman tambahan untuk obat aktif melalui rute transdermal patch seperti
Microneedles patch adalah jarum yang sangat kecil dan tidak menyakitkan yang
berongga atau padat dan diisi dengan obat yang diinginkan. Microneedles
menembus stratum korneum tanpa menimbulkan sensasi nyeri. Keuntungan dari
metode ini adalah sifatnya yang tidak menyakitkan dan kemampuan untuk
menghantarkan senyawa yang memiliki berat molekul lebih tinggi (Khan S., and
Sharman T., 2023).
Stabilitas patch transdermal juga penting untuk mengantarkan obat secara
efektif. Pengemasan patch individu yang tepat sangat penting karena patch
mungkin mengandung pelarut yang mudah menguap atau zat higroskopis. Oleh
karena itu, setiap patch individu perlu disegel panas ke dalam kemasan
multilaminasi yang terdiri dari foil, kertas, dan bagian polietilen yang dapat
disegel panas. Hal ini membutuhkan langkah ekstra dalam proses pembuatannya,
dan menambah biaya ekstra, dibandingkan dengan kebanyakan tablet dan kapsul
yang dapat dikemas dengan mudah dalam botol multientry.
D. Administrasi pemberian patch transdermal
Pemberian patch transdermal harus mengikuti pemeriksaan fisik pasien
yang tepat dan pertimbangan komorbiditas terkait. Langkah-langkah berikut
adalah gambaran umum untuk pemberian patch transdermal:
Disinfektan dan pembersihan kulit yang tepat di tempat patch diterapkan
Penerapan patch di area yang diinginkan
Janji temu lanjutan untuk memastikan bahwa pasien tidak mengalami reaksi
kulit yang merugikan
Kepatuhan yang tepat terhadap pedoman patch transdermal untuk penggunaan
di masa mendatang
Pemberian patch transdermal bervariasi berdasarkan obat yang diberikan
melalui patch. Namun, langkah-langkah tertentu umumnya berlaku untuk semua
patch transdermal:
1. Pasien harus membersihkan dan mensterilkan area tempat patch akan
ditempelkan dengan air jernih, menepuk area tersebut hingga benar-benar
kering. Mereka harus menghindari penggunaan sabun, alkohol, losion, atau
minyak apa pun sebelum mengoleskan patch.
2. Mereka harus membuka kantong yang berisi patch dan mengeluarkannya dari
kantong. Kemudian kelupas kedua bagian lapisan pelindung dari bagian
belakang patch, berhati-hatilah agar tidak menyentuh sisi perekat patch.
3. Pasien kemudian harus segera menekan sisi perekat patch ke kulit mereka
dengan telapak tangan.
4. Pegang atau tekan patch dengan kuat selama minimal 30 detik, pastikan patch
menempel dengan baik pada kulit, terutama di bagian tepinya.
5. Jika patch tidak melekat dengan baik atau menjadi longgar setelah aplikasi,
mereka dapat menggunakan selotip pertolongan pertama untuk merekatkan
ujung-ujungnya ke kulit mereka. Jika patch masih gagal menempel dengan
baik pada kulit, mereka dapat mencoba menggunakan pembalut tembus
pandang tetapi tidak boleh menutupi patch dengan perban atau selotip jenis
lain.
6. Jika patch lepas sebelum saatnya melepasnya, pasien harus membuang patch
dengan benar dan memasang patch baru, mengikuti interval pemberian dosis
yang sama seperti patch asli. Dianjurkan untuk menghubungi kantor dokter
atau apoteker mereka dalam kasus seperti itu.
7. Setelah aplikasi patch berhasil, pasien harus segera mencuci tangan dengan
air.
8. Ketika saatnya mengganti patch, pasien akan melepaskan patch lama dan
memasang patch baru ke area kulit yang berbeda.
9. Setelah melepas patch, pasien harus melipatnya dengan sisi perekat menjadi
satu dan membuangnya dengan benar.
Pasien harus memilih situs yang berbeda untuk aplikasi berturut-turut
untuk menghindari iritasi kulit. Beberapa patch hanya dimaksudkan untuk dipakai
selama sebagian hari (misalnya, 12 jam), sedangkan patch lainnya dimaksudkan
untuk dipakai selama beberapa hari. Misalnya, patch fentanil dipakai selama 72
jam lalu dilepas dan dioleskan kembali.
Pasien perlu memahami bahwa patch yang dilepas masih mengandung
cukup obat untuk menimbulkan masalah jika mereka tidak membuangnya dengan
benar. Ini terutama benar jika anak-anak dapat menguasainya. Patch tidak boleh
digunakan jika rusak atau robek, karena hal ini dapat menyebabkan fenomena
"dosis dumping" di mana obat disalurkan lebih cepat dan dengan dosis yang lebih
besar dari yang dimaksudkan. Jika pasien memiliki pertanyaan tentang patch
transdermal mereka, mereka harus segera menghubungi dokter atau apoteker
mereka (Khan S., and Sharman T., 2023).
E. Kesalahan Umum Penggunaan Sediaan Patch
Produk patch harus menunjukkan tingkat tidak mengiritasi, fototoksisitas,
dan sensitisasi lebih rendah, sensitisasi kulit, dan kelengketan yang lebih baik.
Lokasi aplikasi untuk patch harus terstandarisasi dan sama dengan referensi
karena beberapa patch bekerja pada situs berbeda di wilayah yang sama. Patch
tidak harus ditempelkan secara berlebihan. Pemindahan patch kulit pemakai ke
yang bukan pemakai patch secara tidak sengaja harus dicegah dan juga risiko
terkait perekatan yang buruk harus diminimalkan dengan memastikan
karakteristik perekatan yang dapat diterima dari lekatan patch. Daya rekat patch
harus diukur sebagai persentase area yang tetap melekat pada akhir dari interval
dosis. Untuk mengevaluasi daya rekat patch, pengaruh dari faktor eksternal
(misalnya panas, krim matahari) harus dipertimbangkan (CHMP, 2014). Jadi
sangat penting untuk melepas patch dengan hati-hati dan membuangnya sesuai
petunjuk yang diberikan. Sisa obat di patch dapat menimbulkan bahaya bagi anak-
anak yang mungkin menarik patch dari sampah, dan sisa obat ini juga merupakan
sumber yang sangat diinginkan dari zat yang dikendalikan untuk pecandu. Oleh
karena itu, pengembangan pompa dosis terukur atau sistem difusi aktif mungkin
sangat bermanfaat untuk membuat produk transdermal patch dengan penggunaan
obat yang lebih efisien dan profil keamanan/ penyalahgunaan yang lebih baik
(Paudel, et.al. 2010).
F. Gejala Setelah Penggunaan Sediaan Patch
Efek samping utama yang dapat disebabkan oleh pemberian obat
transdermal patch adalah reaksi kulit. Patch transdermal adalah metode
pengiriman zat aktif yang paling umum. Patch transdermal dapat mengiritasi kulit
dan menyebabkan gatal-gatal, luka bakar, dan kemerahan pada area sekitarnya.
Selain itu, reaksi alergi dilaporkan untuk semua jenis patch pada kulit karena zat
aktif yang diberikan. Dua reaksi kulit yang paling umum adalah dermatitis kontak
iritan dan dermatitis kontak alergi, keduanya biasanya disebabkan oleh obat atau
patch, termasuk perekat dan eksipien. Jadi jelas bahwa menggunakan beberapa
patch hanya satu yang diindikasikan atau menempelkan lebih lama dari yang
diarahkan dapat menyebabkan dosis obat yang berlebihan dan bahkan overdosis
(Khan S., and Sharman T., 2023).
Faktor-faktor yang berkontribusi terhadap iritasi kulit termasuk perubahan
pH fisiologis kulit, gangguan penghalang stratum korneum (yaitu, delipidisasi,
hidrasi dan gangguan kemasan lipid stratum korneum), reaksi imunologi dan
fisiologis, proliferasi bakteri di tempat pengiriman, dan fitur kimia/farmakologis
obat. Banyak hal berkontribusi terhadap iritasi kulit termasuk bahan aktif farmasi,
untuk formulasi (termasuk peningkat permeasi kulit dan eksipien), oklusi kulit,
durasi aplikasi patch, dan jenis patch yang digunakan. Selain itu, perekat dan
membran patch, pelarut, peningkat dan obat aktif semuanya telah terbukti
berkontribusi pada pengembangan reaksi alergi (Paudel, et.al. 2010).
G. Kontraindikasi sediaan patch
Penggunaan obat transdermal patch dikontraindikasikan pada pasien,
seperti: (Khan S., and Sharman T., 2023).
1. Alergi terhadap zat aktif yang digunakan dalam patch transdermal
2. Defisiensi asam lemak esensial
3. Kulit kering
4. Psoriasis
5. Ichthyosis
6. Dermatitis atopik
7. Peningkatan paparan radiasi ultraviolet
8. neonatus prematur
9. Ibu hamil (tergantung obat yang diberikan)
H. Peningkatan Perawatan Kesehatan
Banyak masalah terkait obat mengakibatkan bahaya terkait pengobatan
dan perawatan kesehatan berkualitas rendah untuk pasien tanpa peraturan dan
teknik pemberian yang tepat. Untuk mengirimkan obat dengan benar melalui
transdermal patch, langkah-langkah berikut diperlukan:
Pemeriksaan fisik yang tepat dan memperbarui riwayat medis pasien oleh
dokter yang meresepkan
Konsultasikan dengan apoteker mengenai obat yang tepat untuk diberikan
berdasarkan gejala dan riwayat pasien
Konsultasikan dengan dokter jika ada kekhawatiran tentang patch transdermal
Jika ada keadaan darurat yang mengancam jiwa terkait dengan patch
transdermal, unit gawat darurat harus segera dihubungi.
Menurut Peraturan Pemerintah Nomor 51 tahun 2009 menyatakan bahwa
pekerjaan kefarmasian tersebut harus dilakukan oleh tenaga kesehatan yang
mempunyai keahlian dan kewenangan untuk itu peran apoteker dituntut untuk
meningkatkan pengetahuan, keterampilan, dan perilaku agar dapat melaksanakan
interaksi langsung dengan pasien. Bentuk interaksi tersebut antara lain adalah
pemberian informasi obat dan konseling kepada pasien sehingga apoteker harus
memahami dan menyadari kemungkinan terjadinya kesalahan pengobatan
(medication error) dalam proses pelayanan dan mengidentifikasi, mencegah, serta
mengatasi masalah terkait obat (drug related problems), masalah
farmakoekonomi, dan farmasi sosial (socio-pharmacoeconomy). Ini juga
mencakup penguatan poin-poin konseling pasien untuk memastikan administrasi
yang tepat dan menghubungi penulis resep jika ternyata pasien tidak dapat
melakukan administrasi mandiri dengan benar. Untuk menghindari hal tersebut,
apoteker harus menjalankan praktik sesuai standar pelayanan. Apoteker harus
mampu berkomunikasi dengan tenaga kesehatan lainnya dalam menetapkan terapi
untuk mendukung penggunaan obat yang rasional. Dalam melakukan praktik
tersebut, apoteker juga dituntut untuk melakukan monitoring penggunaan obat,
melakukan evaluasi serta mendokumentasikan segala aktivitas kegiatannya. Untuk
melaksanakan semua kegiatan ini diperlukan Standar Pelayanan Kefarmasian.
Konseling merupakan bentuk komunikasi antara apoteker dengan pasien
untuk memberikan pengetahuan mengenai obat yang dikonsumsi dengan tujuan
untuk mencegah terjadinya medication eror (pengobatan yang salah). Konseling
untuk pasien rawat jalan maupun rawat inap di semua fasilitas kesehatan dapat
dilakukan atas inisitatif Apoteker, rujukan dokter, keinginan pasien atau
keluarganya. Pemberian konseling yang efektif memerlukan kepercayaan pasien
dan/atau keluarga terhadap Apoteker. Pemberian konseling sediaan patch
bertujuan untuk mengoptimalkan hasil terapi, meminimalkan resiko obat yang
tidak diketahui (ROTD), dan meningkatkan cost-effectiveness yang pada akhirnya
meningkatkan keamanan penggunaan obat bagi pasien (patient safety) (Fajarini
H., and Ludin A., 2020). Dengan koordinasi interprofessional dan berbagi
informasi selama perawatan pasien, pengobatan transdermal patch dapat secara
signifikan menguntungkan populasi pasien yang sesuai, menghasilkan kepatuhan
pengobatan yang lebih baik dan hasil yang lebih baik.
I. Contoh Jurnal Sediaan Patch
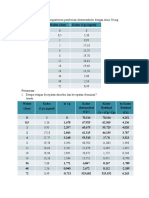
Menurut Asian Journal of Pharmaceutical, 2007, tentang studi oleoresin
sebagai penambah penetrasi untuk patch transdermal ketoprofen, menyatakan
bahwa ketoprofen merupakan obat analgesik yang bekerja sebagai antiinflamasi.
Ketoprofen dapat digunakan secara oral maupun topical. Penggunaan secara
topical dapat dilakukan melalui patch transdermal. Bentuk sediaan patch
transdermal ketoprofen lebih efektif dan lebih nyaman digunakan dibandingkan
bentuk sediaan lainnya, karena mampu menghantarkan obat ke dalam sistem
aliran darah dengan lebih baik juga efek samping yang ditimbulkan lebih ringan.
Peningkat atau promotor penetrasi adalah agen yang memiliki sifat terapeutik
sendiri tetapi dapat mengangkut penyerapan obat dari sistem penghantaran obat,
ke kulit atau penetrasi transdermal selanjutnya melalui kulit.
Contoh jurnal sediaan patch ketoprofen
Obat-obatan golongan AINS yang digunakan secara oral diketahui
menyebabkan gangguan pada gastrointestinal, sehingga kurang baik diberikan
secara peroral termasuk sediaan ketoprofen. Ketoprofen memiliki waktu eliminasi
sangat cepat, sehingga jika dikonsumsi terlalu sering akan terakumulasi di dalam
tubuh. Jika jumlahnya mencapai 300 mg, maka akan mengiritasi lambung dan
mengakibatkan pendarahan. Pemberian ketoprofen secara patch transdermal dapat
dilakukan untuk menghindari gangguan yang terjadi pada pemberian oral,
meningkatkan bioavailabilitas obat dan meningkatkan kenyamanan pasien. Sistem
penghantaran obat secara patch transdermal mampu menghantarkan obat ke
sistem aliran darah dengan efektif, tepat dosis dan dapat menghindari terjadinya
first pass effect, sehingga mengurangi toksisitas obat.
Hasil analisis keseluruhan studi, dapat disimpulkan bahwa untuk
menghantarkan obat melalui kulit, peningkat penetrasi memainkan peran penting.
Jadi untuk mengantarkan obat ke sirkulasi sistemik pada kecepatan yang telah
ditentukan oleh sistem terapi patch transdermal Ketoprofen, menggunakan
oleoresin alami sebagai peningkat penetrasi. Singkatnya, oleoresin seperti
oleoresin pala, oleo resin capsicum, oleoresin bawang putih, oleoresin ketumbar
menunjukkan daya tembus dan obat dengan fluks maksimum. Di antara oleoresin
ini capsaicin menunjukkan kinerja yang lebih baik sebagai peningkat penetrasi
(Gupta, A.M. et.al. 2007).
Gambar: Produk Patch
BAB III
KESIMPULAN
Sediaan transdermal memiliki banyak manfaat dibandingkan penghantaran
obat secara oral, termasuk menghindari metabolisme jalur pertama, melindungi
obat sensitif dari lingkungan gastrointestinal yang keras, memungkinkan
pelepasan obat yang berkelanjutan, dan mempertahankan konsentrasi plasma yang
lebih seragam. Transdermal patch adalah salah satu bentuk sediaan farmasi baru
untuk mengantarkan obat ke aliran darah setelah dilekatkan ke kulit. Pemberian
konseling sediaan transdermal patch bertujuan untuk mengoptimalkan hasil terapi,
meminimalkan resiko obat yang tidak diketahui, dan meningkatkan cost-
effectiveness yang pada akhirnya meningkatkan keamanan penggunaan obat bagi
pasien (patient safety).
Penggunaan secara topical Ketoprofen dapat dilakukan melalui patch
transdermal. Bentuk sediaan patch transdermal ketoprofen lebih efektif dan lebih
nyaman digunakan dibandingkan bentuk sediaan lainnya, karena mampu
menghantarkan obat ke dalam sistem aliran darah dengan lebih baik juga efek
samping yang ditimbulkan lebih ringan.
DAFTAR PUSTAKA
Alkilani A.Z., Musleh B., Hamed R., Swellmeen L., and Basheer H.A., 2023,
Preparation and Characterization of Patch Loaded with Clarithromycin
Nanovesicles for Transdermal Drug Delivery, J. Funct. Biometer, Vol. 14,
No.2, https://doi.org/10.3390/jfb14020057
Committee for Medicinal Products for Human Use (CHMP), 2014, Guideline on
the pharmacokinetic and clinical evaluation of modified release dosage
forms, European Medicines Agency Science Medicines Health
Fajarini H., and Ludin A., 2020, Evaluasi Pelaksanaan Konseling di Apotek Etika
Farma Brebes berdasarkan PERMENKES RI Nomor 73 Tahun 2016, Jurnal
Sains dan Kesehatan, Jurnal Sains dan Kesehatan, Vol. 2, No. 4, e-ISSN:
2407-6082
Gupta A.M., Mundhada D.R., Yeole P.G., and Pande V.V., 2007, Studies of
Oleoresins As Penetration Enchancer for Transdermal Patch of Ketoprofen,
Asian Journal of Pharmaceutics, Vol. 1, No. 1
Khan S., and Sharman T., 2023, Transdermal Medications, Treasure Island:
StatPearls
Paudel K.S., Milewski M., Swadley C.L., Brogden N.K., Ghosh P., and
Stinchcomb A.L., 2010, Challenges and opportunities in dermal/transdermal
delivery. Therapeutic Delivery, Vol. 1, No. 1, 109–
131. doi:10.4155/tde.10.16
Yati K., and Pamungkas S.T., 2018, The Formulation of Carvedilol Transdermal
Patch With Resin Gum as Rate Control, Pharmaciana, Vol. 8, No. 1, ISSN:
2477 0256
Anda mungkin juga menyukai
- Makalah IsoprenoidDokumen19 halamanMakalah IsoprenoidSry ResekiBelum ada peringkat
- OBAT DAN RUTE PEMBERIANDokumen39 halamanOBAT DAN RUTE PEMBERIANIndah C. KadullahBelum ada peringkat
- ALIPHATIC POLYESTER MANUFACTURINGDokumen12 halamanALIPHATIC POLYESTER MANUFACTURINGANita Zulhadj Damayanti100% (1)
- HORMONDokumen74 halamanHORMONCamboy D' JablezBelum ada peringkat
- JUDULDokumen22 halamanJUDULiyamBelum ada peringkat
- UJI DISOLUSIDokumen11 halamanUJI DISOLUSIMira EnmilianaBelum ada peringkat
- Kerangka Topik Penelitian - Een Husnul F - 194201416023Dokumen5 halamanKerangka Topik Penelitian - Een Husnul F - 194201416023Een HusnulfebriantiBelum ada peringkat
- SEDIAAN PARENTERAL TRAMADOLDokumen21 halamanSEDIAAN PARENTERAL TRAMADOLArum Dwi KusumaraniBelum ada peringkat
- IDENTIFIKASI ANTRAQUINON DENGAN KLTDokumen14 halamanIDENTIFIKASI ANTRAQUINON DENGAN KLTMeylania Citra Dewi100% (1)
- DIRD-DrugDokumen16 halamanDIRD-DrugfellycahyanaBelum ada peringkat
- Perhitungan Farkin-1Dokumen14 halamanPerhitungan Farkin-1alyahBelum ada peringkat
- SEDIAAN LARUTANDokumen25 halamanSEDIAAN LARUTANFaisal FebriyantoBelum ada peringkat
- Kelompok 5 - QC Salep Acyclovir 5%Dokumen19 halamanKelompok 5 - QC Salep Acyclovir 5%Sheila Pratiwi100% (1)
- Hasil Dan Pembahasan KelarutanDokumen4 halamanHasil Dan Pembahasan Kelarutansaya siapaBelum ada peringkat
- Makalah CapsaicinDokumen25 halamanMakalah CapsaicinWildan Ramadhan100% (1)
- Dipiro Pharmacotherapy Edition 8-672-690.en - IdDokumen19 halamanDipiro Pharmacotherapy Edition 8-672-690.en - IdNadifulBelum ada peringkat
- Teori Modul6 SSODokumen33 halamanTeori Modul6 SSORini AstutiBelum ada peringkat
- Kel 10 - Produksi Sediaan Emulsi Minyak Ikan - Rev 5Dokumen53 halamanKel 10 - Produksi Sediaan Emulsi Minyak Ikan - Rev 5Nida nur fadhillahBelum ada peringkat
- SPO Swelling Controlled (Topik 1 FG C)Dokumen7 halamanSPO Swelling Controlled (Topik 1 FG C)DickiRachmanBelum ada peringkat
- Program Kerja DiproDokumen7 halamanProgram Kerja DiproIta NurisariBelum ada peringkat
- TULAKDokumen14 halamanTULAKAnggrilina FitriaBelum ada peringkat
- Laporan Semsol Uji Pelepasan Gel Na DiklofenakDokumen7 halamanLaporan Semsol Uji Pelepasan Gel Na DiklofenakmonikatwBelum ada peringkat
- Pembahasan AntikonvulsanDokumen2 halamanPembahasan AntikonvulsanDani Thoppy Saputra100% (1)
- Makalah TSF FixDokumen29 halamanMakalah TSF Fixnadya puspa kusumahBelum ada peringkat
- LaporanDokumen15 halamanLaporanMarina FitrianiBelum ada peringkat
- Makalah Terpenoid Stikes GarutDokumen14 halamanMakalah Terpenoid Stikes GarutRESYA KHAIRUNNISABelum ada peringkat
- Farmakokinetika Data Urine (6)Dokumen15 halamanFarmakokinetika Data Urine (6)cory imaBelum ada peringkat
- Jurnal Meiliza 3BDokumen109 halamanJurnal Meiliza 3BMeiliza HandayaniBelum ada peringkat
- Pengambilan Darah pada Hewan UjiDokumen6 halamanPengambilan Darah pada Hewan UjiI Kadek Adi Putra Suandana 2005Belum ada peringkat
- Grafik viskositas minyak nabati vs suhuDokumen3 halamanGrafik viskositas minyak nabati vs suhuM.TAUFIQ HIDAYATBelum ada peringkat
- Uji KlinikDokumen24 halamanUji KlinikOwenBelum ada peringkat
- Model Farmakokinetik Linier dan Non LinierDokumen17 halamanModel Farmakokinetik Linier dan Non LinierLie Mei LinBelum ada peringkat
- AFI Kromatografi GasDokumen8 halamanAFI Kromatografi GasgantiraBelum ada peringkat
- Dasar Teori Dapus SterilDokumen4 halamanDasar Teori Dapus SterilPramytha WidyasiwiBelum ada peringkat
- TP Perc. 2Dokumen7 halamanTP Perc. 2hanum hasyifah fahriahBelum ada peringkat
- Obat OtonomDokumen9 halamanObat Otonomhelda ketmaBelum ada peringkat
- Kesetimbangan FaseDokumen14 halamanKesetimbangan FaseharyBelum ada peringkat
- Konsep Disolusi Editor-1Dokumen25 halamanKonsep Disolusi Editor-1Inna TakdirBelum ada peringkat
- FArmakokinetik Klinik Dan TDMDokumen23 halamanFArmakokinetik Klinik Dan TDMChandra pranataBelum ada peringkat
- Tugas Laporan Praktikum Teknologi Sediaan Steril Pertemuan 8Dokumen23 halamanTugas Laporan Praktikum Teknologi Sediaan Steril Pertemuan 8hasan maulanaBelum ada peringkat
- Isolasi Dan Standardisasi Bahan AlamDokumen10 halamanIsolasi Dan Standardisasi Bahan AlamsupatmiBelum ada peringkat
- Laporan Tba KadarDokumen6 halamanLaporan Tba KadarYuberd RomieBelum ada peringkat
- OPTIMASI METODE ANALISA OBATDokumen19 halamanOPTIMASI METODE ANALISA OBATSandy Maha PutriBelum ada peringkat
- Laporan AntioksidanDokumen13 halamanLaporan AntioksidanRatulangi RatWulBelum ada peringkat
- Resume Pertemuan 6&7Dokumen14 halamanResume Pertemuan 6&7royan abdiBelum ada peringkat
- Pemanfaatan Obat ParacetamolDokumen5 halamanPemanfaatan Obat ParacetamolAlferizal SeptianBelum ada peringkat
- Identifikasi Kualitatif Senyawa Tanaman Obat Bu AtinaDokumen25 halamanIdentifikasi Kualitatif Senyawa Tanaman Obat Bu AtinaYunitia AnjaniBelum ada peringkat
- Standarisasi Parameter Spesifik Ekstrak Etanol Daun Tapak LimanDokumen2 halamanStandarisasi Parameter Spesifik Ekstrak Etanol Daun Tapak LimanmegawatiBelum ada peringkat
- Sbo RegioselektifDokumen12 halamanSbo RegioselektifTrisnadi AfriawanBelum ada peringkat
- MODIFIKASI PROTEINDokumen16 halamanMODIFIKASI PROTEINnadia0% (1)
- Inflamasi Akut SekaliDokumen27 halamanInflamasi Akut SekaliZufar FirzaBelum ada peringkat
- P-12 SuppositoriaDokumen7 halamanP-12 SuppositoriaSyarifahBelum ada peringkat
- MentolDokumen2 halamanMentolnugrah_angrainiBelum ada peringkat
- OPTIMALKAN KELARUTANDokumen27 halamanOPTIMALKAN KELARUTANAs'adBelum ada peringkat
- Laporan MPB Ekstraksi Dan PemekatanDokumen11 halamanLaporan MPB Ekstraksi Dan Pemekatansiti laelatul kBelum ada peringkat
- KELARUTAN KAFEINDokumen29 halamanKELARUTAN KAFEINLisa JonesBelum ada peringkat
- OPTIMALKANDokumen30 halamanOPTIMALKANMf PitaBelum ada peringkat
- Outline Spo-1Dokumen71 halamanOutline Spo-1Ari DewiyantiBelum ada peringkat
- Kelompok 21 Transdermal Drug Delivery SystemDokumen34 halamanKelompok 21 Transdermal Drug Delivery SystemEJ ArtaBelum ada peringkat
- Patches 2 LNGKPDokumen15 halamanPatches 2 LNGKPNur BaytiBelum ada peringkat
- Tabel StemiDokumen1 halamanTabel StemiFirman oktivendraBelum ada peringkat
- Sarana Prasarana PenyimpananDokumen4 halamanSarana Prasarana PenyimpananFirman oktivendraBelum ada peringkat
- KKS - Kelompok 14Dokumen18 halamanKKS - Kelompok 14Firman oktivendraBelum ada peringkat
- KKS KLMPK 6 - AsmaDokumen22 halamanKKS KLMPK 6 - AsmaFirman oktivendraBelum ada peringkat
- Firman Oktivendra - O1b122108 - Manajemen FarmasiDokumen16 halamanFirman Oktivendra - O1b122108 - Manajemen FarmasiFirman oktivendraBelum ada peringkat
- Babhhyuii DotDokumen12 halamanBabhhyuii DotFirman oktivendraBelum ada peringkat
- Promosi KesehatanDokumen2 halamanPromosi KesehatanFirman oktivendraBelum ada peringkat
- Dukungan Manajemen Rumah SakitDokumen7 halamanDukungan Manajemen Rumah SakitFirman oktivendraBelum ada peringkat
- Daftar PustakaDokumen1 halamanDaftar PustakaFirman oktivendraBelum ada peringkat
- Lomba Akademik DiesNatalisDokumen1 halamanLomba Akademik DiesNatalisFirman oktivendraBelum ada peringkat
- C - O1a118139 - Firman Oktivendra - FarmakokinetikaklinikDokumen11 halamanC - O1a118139 - Firman Oktivendra - FarmakokinetikaklinikFirman oktivendraBelum ada peringkat
- C - O1a118139 - Firman OktivendraDokumen4 halamanC - O1a118139 - Firman OktivendraFirman oktivendraBelum ada peringkat
- C - O1a118139 - Firman Oktivendra - Buah TomatDokumen13 halamanC - O1a118139 - Firman Oktivendra - Buah TomatFirman oktivendraBelum ada peringkat
- KKN REGULER SEMESTER GENAP 2019/2020Dokumen1 halamanKKN REGULER SEMESTER GENAP 2019/2020Dina OktavianiBelum ada peringkat
- Kelebihan Kekurangan, Syarat, Tipe Spektro UvDokumen4 halamanKelebihan Kekurangan, Syarat, Tipe Spektro UvFirman oktivendraBelum ada peringkat
- FutsalDokumen1 halamanFutsalFirman oktivendraBelum ada peringkat
- Bagian Spektrofotometri UV-VisDokumen3 halamanBagian Spektrofotometri UV-VisFirman oktivendra100% (2)
- Proses PembakaranDokumen2 halamanProses PembakaranFirman oktivendraBelum ada peringkat
- Kosong 3Dokumen2 halamanKosong 3Firman oktivendraBelum ada peringkat
- O1a118139 Firman OktivendraDokumen7 halamanO1a118139 Firman OktivendraFirman oktivendraBelum ada peringkat
- O1a118139 Firman OktivendraDokumen8 halamanO1a118139 Firman OktivendraFirman oktivendraBelum ada peringkat
- Kegunaan Spektro UvDokumen1 halamanKegunaan Spektro UvFirman oktivendraBelum ada peringkat
- OPTIMASI RHINITIS ALERGI DAN RESPIRATORY DISTRESS SYNDROMEDokumen12 halamanOPTIMASI RHINITIS ALERGI DAN RESPIRATORY DISTRESS SYNDROMEFirman oktivendraBelum ada peringkat
- Formulasi Dan Teknologi Sediaan SterilDokumen11 halamanFormulasi Dan Teknologi Sediaan SterilFirman oktivendraBelum ada peringkat
- FT - Uas - KLSC - Firman Oktivendra - O1a118139Dokumen15 halamanFT - Uas - KLSC - Firman Oktivendra - O1a118139Firman oktivendraBelum ada peringkat
- C - O1a118139 - Firman OktivendraDokumen8 halamanC - O1a118139 - Firman OktivendraFirman oktivendraBelum ada peringkat
- Tugas IndividuDokumen20 halamanTugas IndividuFirman oktivendraBelum ada peringkat
- Kleas C - O1a1118146 - Alya Dwi Meistyka Sari - Uas BiofarmasetikaDokumen5 halamanKleas C - O1a1118146 - Alya Dwi Meistyka Sari - Uas BiofarmasetikaFirman oktivendraBelum ada peringkat
- FT - Uas - KLSC - Firman Oktivendra - O1a118139Dokumen15 halamanFT - Uas - KLSC - Firman Oktivendra - O1a118139Firman oktivendraBelum ada peringkat
- FT - Uas - KLSC - Firman Oktivendra - O1a118139Dokumen15 halamanFT - Uas - KLSC - Firman Oktivendra - O1a118139Firman oktivendraBelum ada peringkat